w w w . r e u m a t o l o g i a . c o m . b r
REVISTA
BRASILEIRA
DE
REUMATOLOGIA
Original
article
Rheumatoid
arthritis
seems
to
have
DMARD
treatment
decision
influenced
by
fibromyalgia
Rafael
Mendonc¸a
da
Silva
Chakr
a,∗,
Claiton
Brenol
a,
Aline
Ranzolin
b,
Amanda
Bernardes
a,
Ana
Paula
Dalosto
a,
Giovani
Ferrari
a,
Stephanie
Scalco
a,
Vanessa
Olszewski
a,
Charles
Kohem
a,
Odirlei
Monticielo
a,
João
Carlos
T.
Brenol
a,
Ricardo
M.
Xavier
aaUniversidadeFederaldoRioGrandedoSul(UFRGS),PortoAlegre,RS,Brazil bUniversidadeFederaldePernambuco(UFPE),Recife,PE,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory: Received18May2016 Accepted8November2016 Availableonline10February2017
Keywords: Fibromyalgia Rheumatoidarthritis Drugtherapy
a
b
s
t
r
a
c
t
Objective:TocompareDMARDuseinpatientswithandwithoutFMovertime,including overtreatmentandundertreatmentratesinbothgroups.
Methods:AprospectivecohortstudywithpatientsattendinganRAoutpatientclinicwas con-ducted.ParticipantswereconsecutivelyrecruitedbetweenMarch2006andJune2007and werefollowedthroughDecember2013.DataonDMARDuse(prevalences,dosesand esca-lationrates),DAS28,HAQandradiographicprogressionwerecomparedamongRApatients with FMandwithout FM.Mistreatmentclinicalscenarios wereallegedlyidentifiedand comparedbetweengroups.
Results:256RApatients(32withFM)werefollowedfor6.2±2.0(mean±SD)yearscomprising 2986visits.Atbaseline,RAdurationwas11.1±7.4years.DAS28andHAQweregreaterinRA withFMgroup,andwereclosertoRAwithoutFMgrouptowardstheend.RApatientswithFM usedhigherdosesoftricyclicantidepressants,leflunomideandprednisone,andlowerdoses ofmethotrexate.WhencomparedtoRApatientswithoutFM,participantswithRAandFM usedmoreoftentricyclicantidepressants,leflunomide,prednisone,continuousanalgesics andlessoftenmethotrexate.Groupspresentedsimilar7-yearbiologic-freesurvival,and radiographicprogression-freesurvivalinCoxregression.RApatientswithFMhadgreater proportionsofvisitsinmistreatmentscenarioswhencomparedtoRApatientswithoutFM (28.4vs.19.8%,p<0.001).
∗ Correspondingauthor.
E-mail:[email protected](R.M.Chakr). http://dx.doi.org/10.1016/j.rbre.2017.01.004
Conclusions: RApatientswithFMusedmoreleflunomideandprednisone,andRA mistreat-mentwasmorefrequentinFMpatients.Certainly,RApatientswithFMwillbenefitfroma personalizedT2Tstrategy,includingultrasound(whensuitable)andproperFMtreatment. ©2017ElsevierEditoraLtda.ThisisanopenaccessarticleundertheCCBY-NC-NDlicense (http://creativecommons.org/licenses/by-nc-nd/4.0/).
As
decisões
de
tratamento
com
DMARD
na
artrite
reumatoide
parecem
ser
influenciadas
pela
fibromialgia
Palavras-chave: Fibromialgia Artritereumatoide Tratamentofarmacológico
r
e
s
u
m
o
Objetivo: Compararousodefármacosantirreumáticosmodificadoresdadoenc¸a(DMARD) empacientescomesemfibromialgia(FM)aolongodotempo,incluindoastaxasde trata-mentoexcessivoesubtratamentoemambososgrupos.
Métodos: Estudodecoorte prospectivacompacientesatendidosem umambulatóriode artritereumatoide(AR).Osparticipantesforamrecrutadosconsecutivamenteentremarc¸o de2006ejunhode2007eforamseguidosatédezembrode2013.Compararam-seosdadosde usodeDMARD(prevalências,dosesetaxasdeescalonamento),28-JointDiseaseActivityScore (DAS28),HealthAssessmentQuestionnaire(HAQ)eprogressãoradiográficaentrepacientescom esemFM.Oscenáriosclínicosdetratamentosupostamenteincorretoforamidentificados ecomparadosentreosgrupos.
Resultados: Seguiram-se256pacientescomAR(32comFM)por6,2±2,0(média±DP)anos, períodoqueabrangeu2.986consultas.Noiníciodoestudo,adurac¸ãodaARerade11,1±7,4 anos.ODAS28eoHAQforammaioresnogrupoARcomFMeestavammaispróximosdo grupoARsemFMnofimdoestudo.OspacientescomARcomFMusaramdosesmaisaltas deantidepressivostricíclicos,leflunomidaeprednisonaedosesmaisbaixasde metotrex-ato.QuandocomparadoscomospacientescomARsemFM,osparticipantescomAReFM usarammaisfrequentementeantidepressivostricíclicos,leflunomida,prednisonae anal-gésicoscontínuosemenosfrequentementemetotrexato.Osgruposapresentaramsobrevida emseteanossemagentesbiológicoselivresdeprogressãoradiográficasemelhantesna regressãoCox.OspacientescomARcomFMapresentaramumamaiorproporc¸ãode con-sultasem cenáriosde tratamentosupostamenteincorretoquandocomparadoscomos pacientescomARsemFM(28,4vs.19,8%,p<0,001).
Conclusões:OspacientescomAReFMusarammaisleflunomidaeprednisonaeotratamento supostamenteincorretonaARfoimaisfrequenteempacientescomFM.Ospacientescom ARcomFMcertamentesebeneficiarãodeumaestratégiapersonalizadadetratamentopor metas(T2T),incluindoultrassonografia(quandoapropriado)econtroledaFM.
©2017ElsevierEditoraLtda.Este ´eumartigoOpenAccesssobumalicenc¸aCC BY-NC-ND(http://creativecommons.org/licenses/by-nc-nd/4.0/).
Introduction
Rheumatoidarthritis(RA)isasystemicautoimmunedisease characterizedbychronic destructive polyarthritisthat may causeseverefunctionalimpairmentanddeath.1Tostopjoint destruction and prevent worse outcomes clinicians should use disease-modifying antirheumatic drugs (DMARD) in a treat-to-target(T2T)strategy,wherelowerdiseaseactivityis pursued.1–3 Diseaseactivitylevelcanbeclinicallyestimated by28-jointdiseaseactivityscore(DAS28),ascorethatincludes objective (number of swollen joints and erythrocyte sedi-mentationrate)andsubjectiveparameters(numberoftender joints andpatient’s global healthevaluation usingavisual analoguescale).4,5
Fibromyalgia (FM) is a chronic painful condition affect-ing up to 20% ofRApatients. FM may falsely increase RA activitybyaugmentingthesubjectivecomponentsofDAS28
and,therefore,biastreatmentdecision. Bothovertreatment (DMARDescalationwhenahigherDAS28isduetoFM)and undertreatment(noDMARDescalationwhenahigherDAS28 isduetoRA)arepossible.6–10
TheprimarygoalofthisstudyistocompareDMARDuse inpatientswithandwithoutFMovertime.Also,weintend tocompareovertreatmentandundertreatmentratesinboth groups.
Patients
and
methods
AmericanCollegeofRheumatology(ACR)classification crite-riaforRA.11Everyparticipantwasalsoassessedatbaseline for the diagnosis of FM according to 1990 ACR classifica-tion criteria.12 There were two unmatched groups under observation: RA patients with FM and RA patients with-outFM.Exclusioncriteriawereconcomitantsystemiclupus erythematosus, systemicsclerosis,idiopathic inflammatory myopathies,spondyloarthropathies,hepatitisBorCvirus,or humanimmunodeficiencyvirusinfectionatbaselineor dur-ingfollow-up.Participantswerealsoexcludediftheydidnot haveFMatbaselinebutdevelopeddiffusechronicpain after-wards.
Dataofeachvisitwerecollectedinstandardizedresearch forms. DAS28 (ranges from 0.0 through 9.4; greater values represent higher disease activity) and health assessment questionnaire(HAQ;greatervaluesrepresentworsefunctional status)calculation and FM diagnosis were made by differ-entblindedexaminers.13,14Charlsoncomorbidityindex(CCI; ranges from 0 through 35;greater valuesrepresent higher comorbidity)wasalsocalculatedatbaseline.15
Hospital de Clínicas de Porto Alegre isa public tertiary institution where patients have access to all DMARD as part of a national Government-funded programme. Also, analgesics, nonsteroidal anti-inflammatory drugs (NSAID), steroids, amitriptyline, fluoxetine and cyclobenzaprine are providedbytheGovernment.
IntheRAclinic,patientsaretreatedaccordingtoastep-up
T2T approach where DMARD are escalated up to
remis-sionorlowdiseaseactivity.16Forthepurposeofthisstudy, DMARD escalation was defined as any dose increment or drugswitchtoachievetreatmenttarget,andtreatment fail-urewasdefinedasconsistentlymoderate/highdiseaseactivity afterthreemonths ofthe highesttolerateddose ofa syn-theticDMARDorsixmonthsofabiologicDMARD.According toT2T, methotrexate was the first prescribed DMARD and startedjustafterthediagnosis.Ifmethotrexatemonotherapy failed,asecondlinesyntheticDMARDtherapywasstarted. ThissecondsyntheticDMARDstepconsistedofleflunomide monotherapy,association ofmethotrexateand leflunomide or association of methotrexate, sulfasalazine and hydrox-ylchloroquine(orchloroquine).IfthesecondDMARDscheme failed,abiologicDMARD,preferablyananti-tumournecrosis factoragent(anti-TNF),wasstartedinassociationwitha syn-theticDMARD,preferablymethotrexate.Treatmentfailureto thefirstbiologicDMARDwasanindicationforbiologic switch-ing.Anotheranti-TNF,abatacept,rituximabandtocilizumab werethealternatives.Theoptionforthenextbiologictook intoaccountpatients’clinicalaspectsandpreferencesinlight oftheexistingevidence.Adalimumab,etanercept,infliximab andrituximabwereavailableforuseinthestudycentresince itsonsetin2006.Abataceptandtocilizumabbecameavailable in2010,andcertolizumabandgolimumab,in2012.Steroids couldbeusedanytimeatthelowestdosetocontrol synovi-tis.Nonsteroidalanti-inflammatorydrugs(NSAID)wereused for short periods of time to control worsening inflamma-torysymptoms.AnalgesicsNSAID(acetaminophen,dipyrone, tramadol,codeine)were used ondemandfor paincontrol. DMARDwithdrawalwasrecommendedincaseofsustained remission without radiographic progression and consisted ofgradualdose reductionor interval spacingovermonths.
Biologic DMARD should be withdrawn before synthetic
DMARD.2
Duringthestudy,patientswereroutinelyassessedforthe presenceofwidespreadpain.FMtreatmentconsistedofdrug therapy,exerciseandpsychotherapy.Drugtherapyincluded on-demand analgesicsand continuousamitriptyline, fluox-etine,cyclobenzaprine,pregabalinor duloxetine.Pregabalin andduloxetineareavailableforFMtreatmentinBrazilsince 2011.17FMpatientswereencouragedtoattendthe multidis-ciplinaryPainTreatmentclinic.
Everythreemonths,RApatientsshouldperformbloodand urinetestsfordrugsafetymonitoring.Testsfordisease activ-ity assessmentwere performed every threeto sixmonths. RadiographicmonitoringofRAdamagewasperformedonce ayear.Experiencedmusculoskeletalradiologistsnotawareof RAactivity levelorFMdiagnosisreadtheX-raysand radio-graphicprogressionwasdefinedinthepresenceofworsening orappearanceofjoint spacenarrowingortypicaljoint ero-sions in the hands or feet. Previous damage was visually consideredworse,asnomeasuringwasperformed.Absence ofradiographicprogressionwasdefinedasnoworseningor appearanceofjointspacenarrowingortypicalhandsandfeet erosionsintwoconsecutiveradiographs.After2011,patients could be referred forultrasound examinationof RA activ-ity.Thediseaseactivityassessmentbyultrasoundcomprised sevenjoints(dominantwrist,secondandthird metacarpopha-langeal, and second and fifth metatarsophalangeal joints) plus any symptomaticjoints(tender and swollen). Referral forultrasoundexaminationandDMARDescalationdecision afterwardswerebasedonfreeclinician’sjudgement.
To verify the potential role of FM in inducingRA mis-treatment, two clinical scenarios were analyzed. Scenario A comprisedvisits withcontinuous moderate/high disease activity despiteDMARDescalation(treatmentfailure) with-out radiographic progression. In other words, whenever DMARDtreatmentwasnotescalated(DAS28moderate/high) and radiographicprogression was noticed, undertreatment was present. Scenario B comprised visits with persistent moderate/high disease activity without DMARD escalation but with radiographic progression identified. Therefore, in theabsenceofradiographicprogressiondespitepersistently moderate/high DAS28, DMARD escalation was considered overtreatment.Allegedly,scenariosAandBrespectively rep-resentovertreatmentandundertreatment.
equations were used. Biologic DMARD-free survival, and radiographicprogression-freesurvivalwerecomparedusing Kaplan–MeiercurvesandtestedusingCoxproportional haz-ardsmodels.ResultsoftheCoxregressionswerepresentedas hazardratios(HR)and95%confidenceinterval(CI).Apvalue oflessthan0.050wasconsideredstatisticallysignificant.
ThisstudywasapprovedbytheResearchEthics Commit-teesofHospitaldeClínicasdePortoAlegreandUniversidade Federal do Rio Grande do Sul. All patients signed written informedconsentbeforeenteringtheoriginalcross-sectional studyin2006and2007,6andresearcherssignedadata utiliza-tionformtocomplywiththeDeclarationofHelsinki.
Results
Initially, 270 patients were recruited, but 14 participants were excluded due to diffuse pain or overlap syndrome development.Totally,256patientswerefollowedfor6.2±2.0 (mean±SD) years(Table1).They weremostly middle-aged (55.4±12.6years),caucasian(85.2%),married(50.4%),women (84.4%)with≤8yearsofschoolattendance(75.4%).Datafrom 2986visitswere analyzed. All patientsregularlyperformed handsandfeetradiographsthroughoutfollow-up.
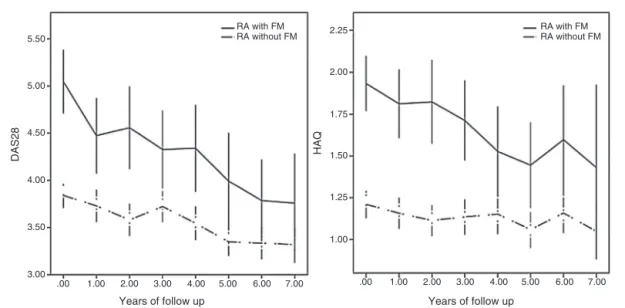
Atbaseline,FMwaspresentin12.5%ofthe participants (n=32), Sjögren’s syndrome in 4.7%, rheumatoid factor in 83.6% and joint erosions in 83.6%.Participants had RA for 11.1±7.4yearsandtheirCCIwas1.7±1.0.Overall,DAS28and HAQscoressignificantlydecreasedfrom4.1±1.9to3.5±1.4 (p<0.001)andfrom1.8±0.8to0.7±0.7(p<0.001),respectively. Initially,RApatientswithFMwereslightlyolder, predom-inantlyfemale and had greater DAS28and HAQ compared tothose withoutFM(Table1).DAS28and HAQvalueswere superiorinRAwithFMovertime(Fig.1).Attheendofthe
study, DAS28 and HAQ were superior in RA patients with FM:4.2±1.3vs.3.3±1.3,p=0.001,and 1.3±0.8vs.0.7±0.7, p<0.001, respectively. However, FM patients exhibited a greater decrease inDAS28 andHAQ valuesthroughoutthe study(Fig.1).Accordingtogeneralizedestimatingequations analyses, between-group comparisons, intra-group (time-based) comparisons and group-time interactions were all statisticallysignificant(p<0.001).Inotherwords,groups dif-feredbetweeneachother,bothofthemchangedovertimeand theydidsoindifferentways.
Considering treatment response, 37.5% and 39.7%
(p=0.809) of RA patients with and without FM, respec-tively, had moderate/high disease activity at baseline and wentdowntoremission/lowdiseaseactivityinthelastvisit. In terms of remission rates, at baseline, 0.0% of patients withFMand24.6%ofpatientswithoutFMwereinremission (p=0.002),and,bytheendofthestudy,remissionrateswere 18.8%and32.1%,respectively(p=0.124).
Duringfollowup,amitriptylinewasusedby24.2%of partic-ipantsat25.0(25.0,50.0)mg/day[median(IQR)],methotrexate by 89.5% at 20.0 (15.0, 20.0)mg/day, leflunomide by 50.8% at 20.0 (20.0, 20.0)mg/day, prednisone by79.3% at 7.5(5.0, 10.0)mg/day and biologic DMARD by 21.9% of individuals. RA patientswith FMusedhigher doses oftricyclic antide-pressants,leflunomide andprednisone, andlower dosesof methotrexate(Table2).Dosesofprednisoneprescribedbythe physicianineachvisitwerealsohigheramongRApatients with FM: 5.0 (0.0,10.0) vs. 2.5(0.0, 7.5), p<0.001. MoreRA patients withFMused tricyclicantidepressants than those withoutFM(Table2).Cyclobenzaprine,fluoxetine,pregabalin, duloxetine, sulfasalazine, chloroquine and hydroxychloro-quinewereusedbyfewerthan50.0%ofparticipantseach.No patientsusedcertolizumaborgolimumabduringthestudy. When compared to RA patients without FM, participants
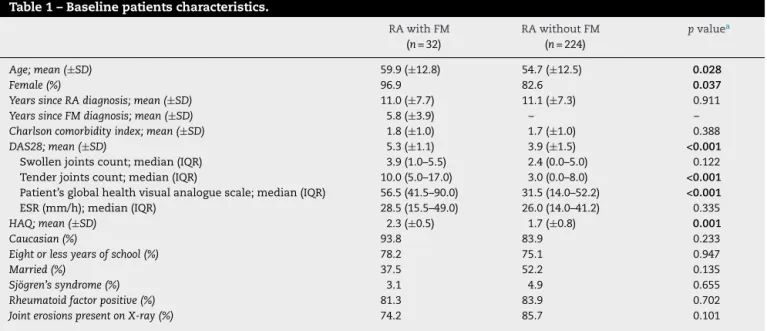
Table1–Baselinepatientscharacteristics.
RAwithFM RAwithoutFM pvaluea
(n=32) (n=224)
Age;mean(±SD) 59.9(±12.8) 54.7(±12.5) 0.028
Female(%) 96.9 82.6 0.037
YearssinceRAdiagnosis;mean(±SD) 11.0(±7.7) 11.1(±7.3) 0.911
YearssinceFMdiagnosis;mean(±SD) 5.8(±3.9) – –
Charlsoncomorbidityindex;mean(±SD) 1.8(±1.0) 1.7(±1.0) 0.388
DAS28;mean(±SD) 5.3(±1.1) 3.9(±1.5) <0.001
Swollenjointscount;median(IQR) 3.9(1.0–5.5) 2.4(0.0–5.0) 0.122
Tenderjointscount;median(IQR) 10.0(5.0–17.0) 3.0(0.0–8.0) <0.001
Patient’sglobalhealthvisualanaloguescale;median(IQR) 56.5(41.5–90.0) 31.5(14.0–52.2) <0.001
ESR(mm/h);median(IQR) 28.5(15.5–49.0) 26.0(14.0–41.2) 0.335
HAQ;mean(±SD) 2.3(±0.5) 1.7(±0.8) 0.001
Caucasian(%) 93.8 83.9 0.233
Eightorlessyearsofschool(%) 78.2 75.1 0.947
Married(%) 37.5 52.2 0.135
Sjögren’ssyndrome(%) 3.1 4.9 0.655
Rheumatoidfactorpositive(%) 81.3 83.9 0.702
JointerosionspresentonX-ray(%) 74.2 85.7 0.101
RA,rheumatoidarthritis;FM,fibromyalgia;DAS28,28-jointdiseaseactivityscore;ESR,erythrocytesedimentationrate;HAQ,healthassessment questionnaire;SD,standarddeviation;IQR,interquartilerange.
a Student’s,Mann–Whitney’sorPearson’schi-squaretestwasusedaccordingtonatureanddistributionofdata;alpha=0.050.Inbold,pvalues
5.50
DAS28
2.25
2.00
1.75
1.50
1.25
1.00 5.00
RA with FM RA without FM
HAQ
RA with FM RA without FM
4.50
4.00
3.50
3.00
.00 1.00 2.00 3.00 4.00 5.00 6.00
Years of follow up Years of follow up
7.00 .00 1.00 2.00 3.00 4.00 5.00 6.00 7.00
Fig.1–DAS28andHAQcurvesofRApatientswithandwithoutFM.DAS28,28-jointdiseaseactivityscore;HAQ,health assessmentquestionnaire;RA,rheumatoidarthritis;FM,fibromyalgia.RApatientswithFMasacontinuouslineandRA patientswithoutFMasaninterruptedline.Generalizedestimatingequations;alpha=0.050;errorbars:95%confidence interval;between-groupcomparisons,p<0.001,intra-group(time-based)comparisons,p<0.001;group-timeinteractions, p<0.001.
withRAandFMused moreoftentricyclicantidepressants, leflunomide,prednisone,continuousanalgesicsandlessoften methotrexatethroughoutthe study (Table3).Nodifference wasobservedbetweengroups regardingthe prevalencesof biologicDMARDuseandDMARDescalations(Table3).
Overall,DMARDescalationratewas1.0(0.7,1.5) DMARD escalation/patient-year.Intermsofbiologicsuse,RApatients wereonaverageunderbiologicDMARDinlessthanonevisit peryear(0.9±0.5visitunderbiologicDMARD/patient-year). DMARDescalationandbiologicsuse didnotdifferbetween groups(Tables 2and 3).Regarding ultrasoundclinic atten-dance,FMwasmorefrequentlyassociatedwithultrasound examination (6.1% vs. 2.9% of visits; p=0.047). Among RA patientswithFM,biologicsusewaslessfrequentwhen ultra-soundexaminationwasperformed:9.1%vs.66.7%ofvisits; p=0.002.
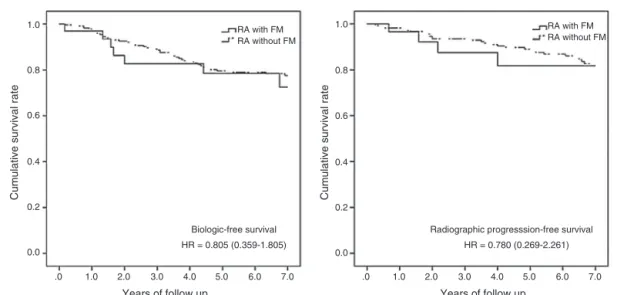
Nostatisticallysignificantdifferenceswerefoundbetween groups concerning7-year biologic-free survival, and radio-graphicprogression-freesurvivalinCoxregressionadjusted by age, sex and CCI (Fig. 2). The only factor significantly influencing7-yearbiologic-free survivalwas age,HR=0.966 (0.944–0.988).Seven-year radiographic progression-free sur-vivalwasnotinfluencedbyanyofthevariables.
ScenarioA(overtreatment)waspresentin15.3%ofallvisits andscenarioB(undertreatment)wasidentifiedin5.5%ofall visits.Overall,RApatientswithFMweremorefrequently over-andundertreated(scenariosA+B)whencomparedwiththose withoutFM(28.4vs.19.8%,p<0.001;Table3).
Discussion
To our best knowledge, this isthe first longitudinal study dedicatedto quantifytheimpact ofFMonDMARD escala-tiondecision inanRAcohort.Recently,Lage-Hansen etal.
described an increase in biologic DMARD use among RA patientswithFMinacross-sectionalstudy.18However,due toitstransversedesignnocausalitycouldbeestablished.In ourstudy,DMARDescalationwasnotaffectedbyFMpossibly duetophysiciansawarenessofitspresence.In2009,ourgroup demonstratedtheimpactofFMonDAS28inpatients attend-ingthesameclinic.6Therefore,rheumatologistswereaware ofthisinterferenceearlyoninthestudy.Also,comparedto otherdiseaseactivityscores,DAS28isparticularlypronetoFM interference,duetotheheavierweightofsubjective compo-nents(tenderjointscountandvisualanaloguescales)inits formula.14,19 By knowingDAS28characteristicsbeforehand, rheumatologistswere morecarefulinescalatingDMARDin RApatientswithFM,emphasizingobjectivemeasures,such asswollenjointscount,erythrocytesedimentationrateand C-reactiveprotein,fortreatmentdecision.10,20Inaddition,since 2010,ultrasoundhasbeen performedforsynovitis quantifi-cationinourcentre.Aspreviouslydemonstrated,synovitis onDopplermaybetterrepresentRAactivitylevelthan clin-icalindexesinpatientswithconcomitantFM.21,22Moreover, FM-inducedDAS28andHAQoverestimationsdiminished dur-ing thestudy,possiblyminimizingovertreatment.However, nodefiniteconclusioncanbedrawnfromthisobservation.
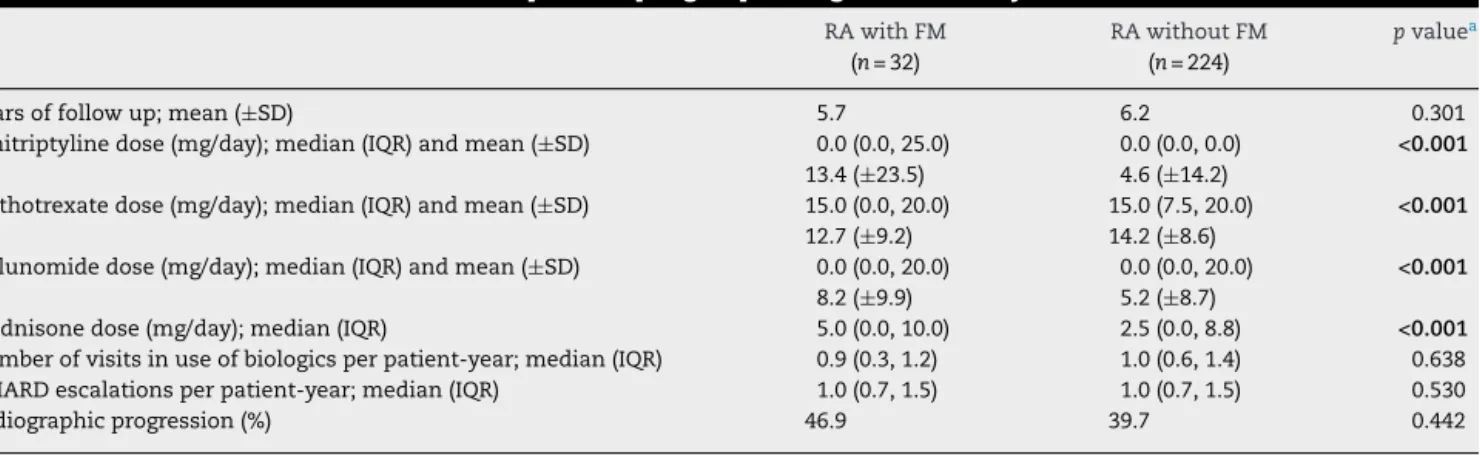
Table2–Treatment-relatedcharacteristicsofpatientspergroupthroughoutthestudy.
RAwithFM RAwithoutFM pvaluea
(n=32) (n=224)
Yearsoffollowup;mean(±SD) 5.7 6.2 0.301
Amitriptylinedose(mg/day);median(IQR)andmean(±SD) 0.0(0.0,25.0) 0.0(0.0,0.0) <0.001
13.4(±23.5) 4.6(±14.2)
Methotrexatedose(mg/day);median(IQR)andmean(±SD) 15.0(0.0,20.0) 15.0(7.5,20.0) <0.001
12.7(±9.2) 14.2(±8.6)
Leflunomidedose(mg/day);median(IQR)andmean(±SD) 0.0(0.0,20.0) 0.0(0.0,20.0) <0.001
8.2(±9.9) 5.2(±8.7)
Prednisonedose(mg/day);median(IQR) 5.0(0.0,10.0) 2.5(0.0,8.8) <0.001
Numberofvisitsinuseofbiologicsperpatient-year;median(IQR) 0.9(0.3,1.2) 1.0(0.6,1.4) 0.638
DMARDescalationsperpatient-year;median(IQR) 1.0(0.7,1.5) 1.0(0.7,1.5) 0.530
Radiographicprogression(%) 46.9 39.7 0.442
RA,rheumatoidarthritis;FM,fibromyalgia;SD,standarddeviation;IQR,interquartilerange;analgesics,acetaminophen,dipyrone,tramadol, codeine;DMARD,disease-modifyingantirheumaticdrug.
a Student’s,Mann–Whitney’sorPearson’schi-squaretestwasusedaccordingtonatureanddistributionofdata;alpha=0.050.Inbold,pvalues
withstatisticalsignificance.
Table3–Treatmentcharacteristicsofeachgroup measuredinvisitsthroughoutthestudy.
RAwithFM RAwithoutFM pvaluea
(n=373 visits)
(n=2613 visits)
Tricyclicantidepressants (%)
31.4 13.2 <0.001
Methotrexate(%) 69.4 79.8 <0.001
Leflunomide(%) 41.3 26.2 <0.001
Prednisone(%) 65.4 52.9 <0.001
Continuousanalgesics(%) 21.7 12.7 <0.001
ContinuousNSAIDs(%) 26.5 22.0 0.076
BiologicDMARD(%) 10.5 12.4 0.292
Abatacept(%) 1.1 0.9 0.773
Adalimumab(%) 1.3 2.1 0.346
Certolizumab(%) 0.0 0.0 NS
Etanercept(%) 0.0 3.2 <0.001
Golimumab(%) 0.0 0.0 NS
Infliximab(%) 5.1 3.4 0.112
Rituximab(%) 2.9 1.9 0.186
Tocilizumab(%) 0.0 0.8 0.075
DMARDescalations(%) 46.9 47.2 0.960
DMARDescalationsinthe firstyearoffollowup (%)
51.6 49.5 0.400
ScenarioA(%) 20.4 14.6 0.004
ScenarioB(%) 8.0 5.2 0.023
DMARD,disease-modifyingantirheumaticdrug;RA, rheumatoid arthritis;FM,fibromyalgia;analgesics,acetaminophen,dipyrone, tramadol,codeine;NSAID,nonsteroidalanti-inflammatorydrug; NS,notstated;scenarioA,DAS28moderate/high,DMARD,escalated and radiographic progression absent; scenario B, DAS28 mod-erate/high,DMARD,not escalatedandradiographic progression present.
a Student’s,Mann–Whitney’sorPearson’schi-squaretestwasused
accordingtonatureanddistributionofdata;alpha=0.050.Inbold,
pvalueswithstatisticalsignificance.
T2Tstudyinlong-standingRA,DAS28andHAQdiminished after 3 years (p=0.004 and p<0.001, respectively) and the final scoreswere comparable toours: DAS28=3.3±1.4and HAQ=1.1±0.4.Inthis same study,remissionwas achieved by35.3%ofpatientsinitiallyrefractorytosyntheticDMARD.24 Santos-Moreno et al. studied moderately to highly active
RA patients treated to target for 6 months and found a significantdecreasebothinDAS28andHAQ(p<0.001).Their finalvalueswere:DAS28=2.5(2.3,3.2),HAQ=0.1(0.1,0.3)and remissionrate=51%.25Inourstudy,thelongerobservational period, jointdeformitiesand chronicproliferativesynovitis seen inlong-standingdisease,aswell aslower educational levelmayhavecontributedtothefinalDAS28slightlyabove the target.8,26 Despite the greater decrease of DAS28 over timeamongFMpatients,the finalscorewashigherinthis groupbut notthe radiographicprogressionrate,suggesting thattheinterferenceofthepainfulconditioninRAactivity scoring was attenuated but still present throughout the study.
RApatientswithFMusedmoreoftentricyclic antidepres-sants, leflunomide, prednisone and continuous analgesics, but less often methotrexate than those without FM. Tri-cyclicantidepressantindicationwasnotcollectedandother conditions, such as diabetic neuropathy, could have influ-enced this difference. The greater use of the preferably second-line agent leflunomide (Table 3) may indicate an interference of the painful condition in DMARD escala-tion (methotrexatediscontinuation). Also, prednisone dose was greater among FM patients (Table 3), suggesting FM could have pushed steroid therapy forward. In the study by Andersonet al., chronic widespreadpain patients were treatedwithprednisonetoagreaterextentthanthosewith chronicregionalpainsyndromebutasimilarrateofDMARD treatment.9
Radiographicprogressionwas thesame betweengroups and comparabletootherT2Tstudies.27,28 Possibly,no joint damage difference was observed in the presence of FM becauseDMARDescalationwassimilartothosewithoutFM. Inaddition,FMinterferenceontreatmentdecisioncouldhave been attenuated by ultrasound examination. Speculatively, DMARDescalationdecisioncouldhavebecomelessbiasedby FMafterwards.21,29,30
Kaplan–Meier curves contemplate the time elapsed
1.0
0.8
0.6
0.4
0.2
Cumulative survival rate Cumulative survival rate
0.0
.0 1.0 2.0
Biologic-free survival HR = 0.805 (0.359-1.805)
Radiographic progresssion-free survival HR = 0.780 (0.269-2.261)
RA with FM RA without FM
RA with FM RA without FM
Years of follow up Years of follow up
3.0 4.0 5.0 6.0 7.0 .0 1.0 2.0 3.0 4.0 5.0 6.0 7.0
1.0
0.8
0.6
0.4
0.2
0.0
Fig.2–Kaplan–MeiercurvesofRApatientswithandwithoutFM.(A)7-yearbiologic-freesurvival;(B)7-yearradiographic progression-freesurvival.RA,rheumatoidarthritis;FM,fibromyalgia.RApatientswithFMasacontinuouslineandRA
patientswithoutFMasaninterruptedline.Coxproportionalhazardsmodelsadjustedbyage,sex,Charlsoncomorbidity
indexandprednisonedose;HR:hazardratio(95%confidenceinterval);alpha=0.050.
patients.Theseresultshavenotbeendescribedinequivalent studiesyet.
OurfindingssuggestthatRApatientswithFMcouldhave beenmorefrequentlymistreatedthan RApatientswithout FM in two arbitrary scenarios. In theory, these scenarios intendtorepresentbothover-andundertreatment frequen-cies.Nonetheless,nodefiniteconclusioncanbedrawn,since radiographicprogressionwas notquantifiedinascoreand thesescenarios require furthervalidation astrue outcome measures. Moreover, ultrasound scoring could be used in futurestudiesasacomparatorineachscenario,sinceithas beenvalidatedasanobjectivemeasureofsynovitisandasa radiographicprogressionpredictor.21,29
Ultrasound-basedsynovitisassessmentcouldhave influ-encedDMARDescalationdecision,contributingtoreducethe supposedinfluenceofFMonRAtreatment.21Alongitudinal studywithRApatientswithFMdividedintwogroups accord-ingtoultrasoundassessmentcouldaddressthisissuemore properly.Anotherimperfectionwastheexistenceofvarious DAS28examinersthroughoutthestudy,asithasbeen demon-stratedthatclinicalreliabilityofthescoreishighlydependent ontheexaminer.31SinceDAS28wasusuallyperformedbythe traineeandconfirmedbytheseniorrheumatologistandour resultsaresimilartothoseofotherreal-lifestudies,webelieve theexaminersvariationwasnotamajorbiasfortheresults.
Conclusions
In the present RA cohort, FM did not significantly impact overallDMARDescalation,andFM-inducedDAS28andHAQ overestimations diminished towards the end of the study. However,RApatients withFMusedmore leflunomideand prednisone,andRAmistreatmentseemstobemorefrequent inFMpatients. Certainly, RApatients withFM willbenefit fromapersonalizedT2Tstrategy,includingobjective synovi-tisbiomarkers,suchasultrasound,paralleltoapermanent
FMtreatmentoptimization.PersonalizedmedicineinRAisa proficuousresearchfieldandmorestudiesontheimpactof FMonRAtreatmentwouldprobablyhelpimprove decision-makingprocessinbenefitofpatientsandthesociety.
Funding
ThisworkwassupportedbyFundodeIncentivoàPesquisae Eventos(FIPE)ofHospitaldeClínicasdePortoAlegre(HCPA).
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
Acknowledgements
WewouldliketothankthestatisticianVâniaHirakata,MSc, andthecolleaguesfromtheDivisionofRheumatologyof Hos-pitaldeClínicasdePortoAlegreAndreseGasparin,MD,MSc, NicoleAndrade,MD,MScandPenélopePalominos,MD,MSc fortheirinvaluablesupportduringthestudyexecution.The funderwasnotinvolvedinwriting,studydesign,collection, analysisorinterpretationofdata.
r
e
f
e
r
e
n
c
e
s
1.ScottDL,WolfeF,HuizingaTW.Rheumatoidarthritis.Lancet. 2010;376:1094–108.
2.daMotaLM,CruzBA,BrenolCV,PereiraIA,Rezende-Fronza LS,BertoloMB,etal.2012BrazilianSocietyofRheumatology Consensusforthetreatmentofrheumatoidarthritis.Rev BrasReumatol.2012;52:152–74.
recommendationsofaninternationaltaskforce.AnnRheum Dis.2010;69:631–7.
4. SalaffiF,CarottiM,CiapettiA,GaspariniS,FilippucciE,Grassi W.Relationshipbetweentime-integrateddiseaseactivity estimatedbyDAS28-CRPandradiographicprogressionof anatomicaldamageinpatientswithearlyrheumatoid arthritis.BMCMusculoskelDisord.2011;12:120.
5. VermeerM,KuperHH,MoensHJ,Drossaers-BakkerKW,van derBijlAE,vanRielPL,etal.Sustainedbeneficialeffectsofa protocolizedtreat-to-targetstrategyinveryearlyrheumatoid arthritis:three-yearresultsoftheDutchRheumatoid ArthritisMonitoringremissioninductioncohort.Arthritis CareRes.2013;65:1219–26.
6. RanzolinA,BrenolJC,BredemeierM,GuarientiJ,RizzattiM, FeldmanD,etal.Associationofconcomitantfibromyalgia withworsediseaseactivityscorein28joints,health assessmentquestionnaire,andshortform36scoresin patientswithrheumatoidarthritis.ArthritisRheum. 2009;61:794–800.
7. KapoorSR,HiderSL,BrownfieldA,MatteyDL,PackhamJC. Fibromyalgiainpatientswithrheumatoidarthritis:drivenby depressionorjointdamage?ClinExpRheumatol.2011;29 Suppl.69:S88–91.
8. PincusT,CastrejonI,BergmanMJ,YaziciY.Treat-to-target: notassimpleasitappears.ClinExpRheumatol.2012;30 Suppl.73:S10–20.
9. AnderssonML,SvenssonB,BergmanS.Chronicwidespread paininpatientswithrheumatoidarthritisandtherelation betweenpainanddiseaseactivitymeasuresoverthefirst5 years.JRheumatol.2013;40:1977–85.
10.TonE,BakkerMF,VerstappenSM,TerBorgEJ,van
Albada-KuipersIA,SchenkY,etal.Lookbeyondthedisease activityscoreof28joints(DAS28):tenderpointsinfluencethe DAS28inpatientswithrheumatoidarthritis.JRheumatol. 2012;39:22–7.
11.ArnettFC,EdworthySM,BlochDA,McShaneDJ,FriesJF, CooperNS,etal.TheAmericanRheumatismAssociation 1987revisedcriteriafortheclassificationofrheumatoid arthritis.ArthritisRheum.1988;31:315–24.
12.WolfeF,SmytheHA,YunusMB,BennettRM,BombardierC, GoldenbergDL,etal.TheAmericanCollegeofRheumatology 1990criteriafortheclassificationoffibromyalgia:reportof theMulticenterCriteriaCommittee.ArthritisRheum. 1990;33:160–72.
13.MaskaL,AndersonJ,MichaudK.Measuresoffunctional statusandqualityoflifeinrheumatoidarthritis:Health AssessmentQuestionnaireDisabilityIndex(HAQ),Modified HealthAssessmentQuestionnaire(MHAQ),Multidimensional HealthAssessmentQuestionnaire(MDHAQ),Health
AssessmentQuestionnaireII(HAQ-II),ImprovedHealth AssessmentQuestionnaire(ImprovedHAQ),andRheumatoid ArthritisQualityofLife(RAQoL).ArthritisCareRes.2011;63 Suppl.11:S4–13.
14.AndersonJK,ZimmermanL,CaplanL,MichaudK.Measures ofrheumatoidarthritisdiseaseactivity:Patient(PtGA)and Provider(PrGA)GlobalAssessmentofDiseaseActivity, DiseaseActivityScore(DAS)andDiseaseActivityScorewith 28-JointCounts(DAS28),SimplifiedDiseaseActivityIndex (SDAI),ClinicalDiseaseActivityIndex(CDAI),PatientActivity Score(PAS)andPatientActivityScore-II(PASII),Routine AssessmentofPatientIndexData(RAPID),Rheumatoid ArthritisDiseaseActivityIndex(RADAI)andRheumatoid ArthritisDiseaseActivityIndex-5(RADAI-5),ChronicArthritis SystemicIndex(CASI),Patient-BasedDiseaseActivityScore WithESR(PDAS1)andPatient-BasedDiseaseActivityScore withoutESR(PDAS2),andMeanOverallIndexforRheumatoid Arthritis(MOI-RA).ArthritisCareRes.2011;63Suppl.
11:S14–36.
15.CharlsonME,PompeiP,AlesKL,MacKenzieCR.Anew methodofclassifyingprognosticcomorbidityinlongitudinal studies:developmentandvalidation.JChronicDis.1987;40: 373–83.
16.BrenolCV,daChakrRM,AndradeNP,ToniM,LaurindoIM, BrenolJC,etal.Dailypracticefeasibilityandeffectivenessof treatinglong-standingrheumatoidarthritistotargetwith syntheticdisease-modifyingantirheumaticdrugs:a prospectivecohortstudy.ClinRheumatol.2015;34:1781–5. 17.HeymannRE,PaivaEdosS,HelfensteinMJr,PollakDF,
MartinezJE,ProvenzaJR,etal.Brazilianconsensusonthe treatmentoffibromyalgia.RevBrasReumatol.2010;50:56–66. 18.Lage-HansenPR,ChrysidisS,Lage-HansenM,HougaardA,
EjstrupL,AmrisK.Concomitantfibromyalgiainrheumatoid arthritisisassociatedwiththemorefrequentuseof biologicaltherapy:across-sectionalstudy.ScandJ Rheumatol.2015:1–4[Epubaheadofprint].
19.CouryF,RossatA,TebibA,LetroublonMC,GagnardA,Fantino B,etal.Rheumatoidarthritisandfibromyalgia:afrequent unrelatedassociationcomplicatingdiseasemanagement.J Rheumatol.2009;36:58–62.
20.HobbsKF,CohenMD.Rheumatoidarthritisdisease measurement:anewoldidea.Rheumatology(United Kingdom).2012;51Suppl.6:vi21–7.
21.ChakrR,BrenolJC,BeharM,Mendonc¸aJA,KohemC, MonticieloO,etal.Isultrasoundabettertargetthanclinical diseaseactivityscoresinrheumatoidarthritiswith fibromyalgia?Acase–controlstudy.PLOSONE. 2015;10:e0118620.
22.DaleJ,PurvesD,McConnachieA,PorterD,McInnesIB. Tighteningup:musculoskeletalultrasoundcouldfurther individualisetreatmentdecisionsinearlyrheumatoid arthritispatientstreatedbyastep-upDMARDescalation regimen.ArthritisRheum.2012;64:S1129.
23.SokkaT,HetlandML,MakinenH,KautiainenH,
Horslev-PetersenK,LuukkainenRK,etal.Remissionand rheumatoidarthritis:dataonpatientsreceivingusualcarein twenty-fourcountries.ArthritisRheum.2008;58:2642–51. 24.MohammedRH,KewanHH,BukhariM.Assessmentofthe
treat-to-targetstrategyinpatientswithrefractory rheumatoidarthritis.Aprospectivestudyonefficacyand safetyinaSaudipopulation.ZRheumatol.2014;73:746–53. 25.Santos-MorenoPI,delaHoz-ValleJ,VillarrealL,PalominoA,
SanchezG,CastroC.Treatmentofrheumatoidarthritiswith methotrexatealoneandincombinationwithother
conventionalDMARDsusingtheT2Tstrategy.Acohortstudy. ClinRheumatol.2015;34:215–20.
26.SokkaT,KautiainenH,PincusT,TolozaS,daRochaCastelar PinheiroG,LazovskisJ,etal.Disparitiesinrheumatoid arthritisdiseaseactivityaccordingtogrossdomesticproduct in25countriesintheQUEST-RAdatabase.AnnRheumDis. 2009;68:1666–72.
27.SugiharaT,IshizakiT,HosoyaT,IgaS,YokoyamaW,HiranoF, etal.Structuralandfunctionaloutcomesofatherapeutic strategytargetinglowdiseaseactivityinpatientswith elderly-onsetrheumatoidarthritis:aprospectivecohort study(CRANE).Rheumatology(Oxford).2015;54:798–807. 28.VermeerM,KuperHH,HoekstraM,HaagsmaCJ,Posthumus
MD,BrusHL,etal.Implementationofatreat-to-target strategyinveryearlyrheumatoidarthritis:resultsofthe DutchRheumatoidArthritisMonitoringremissioninduction cohortstudy.ArthritisRheum.2011;63:2865–72.
30.Funck-BrentanoT,GandjbakhchF,EtchepareF,Jousse-Joulin S,MiquelA,CytevalC,etal.Predictionofradiographic damageinearlyarthritisbysonographicerosionsandpower Dopplersignal:alongitudinalobservationalstudy.Arthritis CareRes.2013;65:896–902.