www.jped.com.br
ORIGINAL
ARTICLE
Association
of
neopterin
as
a
marker
of
immune
system
activation
and
juvenile
rheumatoid
arthritis
activity
夽
Mones
M.
Abu
Shady
a,
Hanan
A.
Fathy
b,
Alaa
Ali
a,
Eman
R.
Youness
c,∗,
Gihan
A.
Fathy
aaChildHealthDepartment,MedicalDivision,NationalResearchCentre,Cairo,Egypt
bResearchDepartment,NationalCentreforRadiationResearchandTechnology,AtomicEnergyAuthority,Cairo,Egypt cMedicalBiochemistryDivision,NationalResearchCentre,Cairo,Egypt
Received13March2014;accepted10September2014 Availableonline26February2015
KEYWORDS
MCP-1; TNF-␣;
Rheumatoidarthritis
Abstract
Objective: Toevaluateneopterinplasmaconcentrationsinpatientswithactivejuvenile idio-pathicarthritis(JIA)andcorrelatethemwithdiseaseactivity.
Methods: Sixty patients diagnosed as activeJIA, as well as another 60 apparently healthy age- and gender-matched children as controls, were recruited from the Pediatrics Allergy andImmunologyClinic, AinShams University. Diseaseactivitywas assessed bythe Juvenile ArthritisDiseaseActivityScore27(JADAS-27).Laboratoryinvestigationswereperformedforall patients,includingdeterminationofhemoglobinconcentration(Hgb),erythrocyte sedimenta-tionrate(ESR),andC-reactiveprotein.Serumconcentrationsoftumornecrosisfactor-alpha (TNF-␣),interleukin-6(IL-6),monocytechemoattractantprotein-1(MCP-1),andneopterinwere measured.
Results: SignificantdifferenceswerefoundbetweenJIApatientsandcontrolswithregardto themeanlevelsofHgb,ESR,TNF-␣,IL-6,andMCP-1(p<0.05).Astatisticallysignificanthigher meanlevelserumneopterin concentration(p<0.05)wasfound inJIApatients(20.43±8.73 nmol/L)thanincontrols(6.88±2.87nmol/L)(p<0.05).Positivesignificantcorrelationswere detectedbetweenserumneopterinandESR,TNF-␣,IL-6,MCP-1,andJADAS-27(p<0.05).No correlation was found between serum neopterin andCRP (p>0.05).Multiple linear regres-sionanalysisshowedthatJADAS-27andESRwerethemainvariablesassociatedwithserum neopterininJIApatients(p<0.05).
Conclusion: TheelevationofplasmaneopterinconcentrationsinearlyJIApatientsmayindicate stimulationofimmuneresponse.Serumneopterincanbeusedasasensitivemarkerforassaying backgroundinflammationanddiseaseactivityscoreinJIApatients.
©2015SociedadeBrasileiradePediatria.PublishedbyElsevierEditoraLtda.Allrightsreserved.
夽
Pleasecitethisarticleas:AbuShadyMM,FathyHA,AliA,YounessER,FathyGA.Associationofneopterinasamarkerofimmunesystem activationandjuvenilerheumatoidarthritisactivity.JPediatr(RioJ).2015;91:352---7.
∗Correspondingauthor.
E-mail:[email protected](E.R.Youness).
http://dx.doi.org/10.1016/j.jped.2014.09.007
PALAVRAS-CHAVE
MCP-1; FNT-␣;
Artritereumatoide
Associac¸ãodeneopterinacomomarcadordeativac¸ãodosistemaimunológicoe
atividadedaartritereumatoidejuvenil
Resumo
Objetivo: avaliar as concentrac¸ões plasmáticas de neopterina em pacientes com artrite idiopáticajuvenil(AIJ)ativaecorrelacioná-lascomaatividadedadoenc¸a.
Métodos: Sessenta pacientes diagnosticados com AIJ ativa, bem como outras 60 crianc¸as aparentementesaudáveiscomamesmaidadeesexonogrupodecontrole,foramrecrutados daclínicadeAlergiaeImunologiaInfantildaUniversidadeAinShams.Aatividadedadoenc¸afoi avaliadapeloEscoredeAtividadedaDoenc¸adaArtriteJuvenilem27Articulac¸ões(JADAS-27). Foramrealizadasinvestigac¸õeslaboratoriaisemtodosospacientes,incluindoadeterminac¸ão daconcentrac¸ãodehemoglobinas,ataxadesedimentac¸ãodeeritrócitoseaproteínaC-reativa. Forammensuradasasconcentrac¸õesséricasdofatordenecrosetumoralalfa,interleucina-6e proteínaquimiotáticademonócitos-1eneopterina.
Resultados: Foiencontradaumadiferenc¸asignificativaentreospacientescomAIJeos con-trolesquantoàsmédiasdeHb,TSE,FNT-␣,IL-6eMCP-1(p<0,05).Foiencontradoumnível estatísticaesignificativamentemaiordeconcentrac¸ãomédiadeneopterinasérica(p<0,05) em pacientescomAIJ(valormédio de20,43±8,73nmol/L)queem controles(valormédio de6,88±2,87nmol/L)(p<0,05).Foramdetectadascorrelac¸õespositivassignificativasentrea neopterinaséricaeaTSE,FNT-␣,IL-6,MCP-1eJADAS-27(p<0,05).Nãofoiencontrada nen-huma correlac¸ãoentreaneopterinaséricaeaPCR (p>0,05).A análisederegressão linear múltiplamostrouqueoJADAS-27eaTSEforamasprincipaisvariáveisassociadasàneopterina séricaempacientescomAIJ(p<0,05).
Conclusão: A elevac¸ão das concentrac¸ões plasmáticasde neopterina em pacientescomAIJ precocepodeindicarumestímuloderespostaimune.Aneopterinaséricapodeserusadacomo umindicadorsensívelparaanalisarohistóricodeinflamac¸õeseoescoredeatividadedadoenc¸a empacientescomAIJ.
©2015SociedadeBrasileiradePediatria.PublicadoporElsevierEditoraLtda.Todososdireitos reservados.
Introduction
Juvenileidiopathicarthritis(JIA)isgenerallyconsidered a clinical syndrome involving several disease subsets, with a number of inflammatory flows, leading to an eventual commonpathwayinwhichpersistentsynovialinflammation andassociateddamage toarticular cartilageand underly-ing bone arepresent.1 One main inflammatory processin the pathophysiology of the JIAconsists of overproduction of tumor necrosis factor that leads to overproduction of many cytokines such as interleukin-6, which causes per-sistent inflammationand jointdestruction.2---4 The disease arises in a genetically susceptible individual due to envi-ronmentalfactors.5Moreover,ithasbeenproposedthatan antigen-drivenautoimmuneprocessmediatesthe inflamma-torypathologyinsomecasesofarthritis(e.g.,oligoarthritis, polyarthritis).Incontrast,therearenosignsof lymphocyte-mediated,antigen-specificimmuneresponsesinindividuals with systemic onset disease. Recent investigations in the pathophysiology of systemic onset disease have indicated that this disorder is due to an uncontrolled activation of theinnate immunesystem.6Regardless ofthe differences intheunderlyingpathogenesisofthevarioustypes ofJIA, pro-inflammatory cytokinesareconsistently overproduced andarerelatedtotheclinicalmanifestationsinalltypesof JIA.7
The International League of Associations for Rheuma-tology (ILAR) classification system divides JIA into seven clinical subgroups.8 The few population based estimates available indicate that the prevalence of JIA is approxi-mately one totwo per 1,000 children, andthe incidence is11to14newcasesper100,000children.9
Neopterin, a pyrazino-pyrimidine compound, is syn-thesized by monocytes and macrophages in response to interferon-␥ (IFN-␥) produced by activatedT-cells. It is a markerofcellularimmuneresponse,andlevelsareelevated inconditionsofT-cellormacrophagesactivation,including autoimmunediseasessuchassystemiclupuserythematosus andrheumatoidarthritis.10,11
Thecurrent studywasundertakentoassessthe associ-ationof plasma levelof neopterin with inflammatoryand diseaseactivityinJIApatients.
Subjects
and
methods
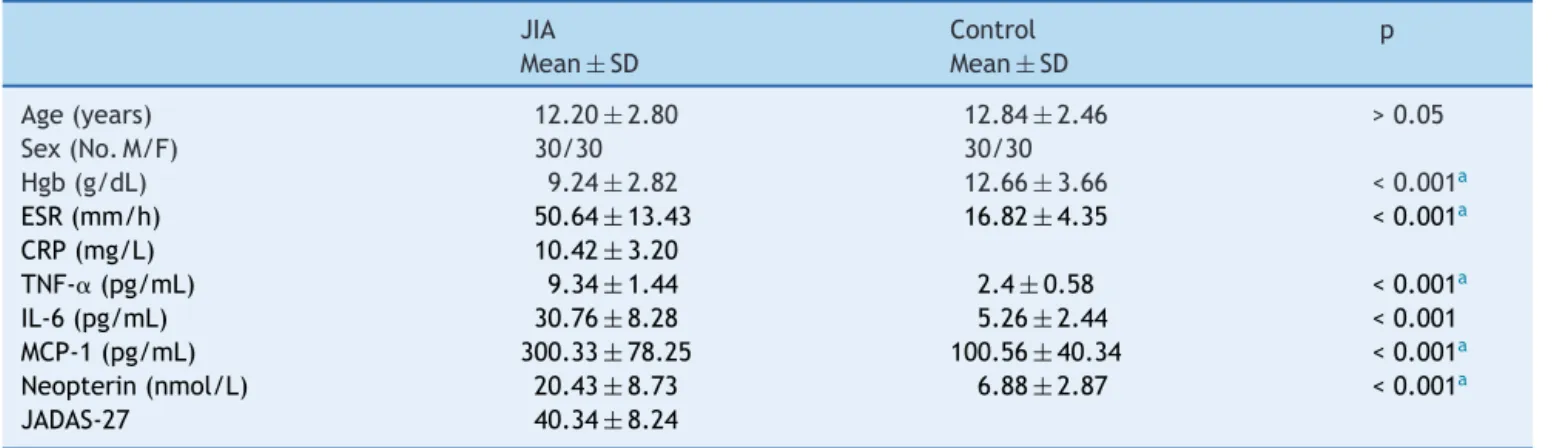
Table1 Demographic,biochemical,anddiseasecharacteristicsofallsubjects.
JIA Control p
Mean±SD Mean±SD
Age(years) 12.20±2.80 12.84±2.46 >0.05
Sex(No.M/F) 30/30 30/30
Hgb(g/dL) 9.24±2.82 12.66±3.66 <0.001a
ESR(mm/h) 50.64±13.43 16.82±4.35 <0.001a
CRP(mg/L) 10.42±3.20
TNF-␣(pg/mL) 9.34±1.44 2.4±0.58 <0.001a
IL-6(pg/mL) 30.76±8.28 5.26±2.44 <0.001 MCP-1(pg/mL) 300.33±78.25 100.56±40.34 <0.001a
Neopterin(nmol/L) 20.43±8.73 6.88±2.87 <0.001a
JADAS-27 40.34±8.24
JIA,juvenileidiopathicarthritis;Hgb,hemoglobinconcentration;ESR,erythrocytesedimentationrate;CRP,C-reactiveprotein;
TNF-␣,tumornecrosisfactor-alpha;IL-6,interleukin-6;MCP-1,monocytechemoattractantprotein-1;JADAS-27,JuvenileArthritisDisease ActivityScore-27.
ap<0.001ishighlysignificant.
corresponding documents were approved by Ethical and ResearchCommitteeof theNationalResearchCenter.Age ranged from 5 to 15 years with a mean age 12.20±2.8 years.PatientswereeligibleiftheymettheEdmonton Inter-national Leagueof Associations for Rheumatology criteria (second revision) for a diagnosis of JIA.8 Disease activ-ity was measured using a validated score, the JADAS-27 (Juvenile ArthritisDiseaseActivity Score-27).12 This score includesfourmeasures:physicianglobalassessmentof dis-easeactivityusingavisualanalogscale(VAS),parentglobal assessmentofchild’swell-beingdeterminedbyaVAS,count of joints with active disease (evaluating 27 joints), and erythrocytesedimentationrate(ESR).ESRisnormalizedto ascorerangingfrom0to10,by theformula(ESR-20)/10. JADAS-27iscalculatedasthesimplelinearsumofthescores ofitsfourcomponents,whichyieldsatotalscoreof0-57, withhigherscores associated withworse disease activity. Thirty-eightpatients(63.3%)hadpolyarticularonsetJIA,15 patients(25%)hadpauciarticularonset,andsevenpatients (11.7%)hadsystemiconset.
All patients had full history takenand were subjected to clinical examination. Laboratory investigations were performed for all patients, including determination of hemoglobin concentration (Hgb in g/dL), ESR in mm/h by Westergren method, and C-reactive protein (CRP in mg/L) detection by the latex agglutination slide test. Serumconcentrationsoftumornecrosisfactor-alpha (TNF-␣), interleukin-6 (IL-6), and monocyte chemoattractant protein-1 (MCP-1) were measured by multiplex enzyme-linked immunosorbent assay (ELISA) (Millipore®, Merck KgaA,Darmstadt,Germany).
Assay
of
serum
neopterin
(nmol/L)
Serumneopterin levels wereexamined usingan ELISA kit (Neopterin ELISA,IBL, Hamburg, Germany). The assay is based on the basic principle of competition between a peroxide-conjugatedandnon-conjugatedantigenforafixed number of antibody binding sites. The peroxidase conju-gated,antigen-antibodycomplexesbindtothewellsofthe microfilterstrips,whicharecoatedwithagoodanti-rabbit
antibody.Unboundedantigenis thenremovedbywashing. Afterthesubstratereaction,theopticaldensityismeasured at450nm.Astandardcurveisplottedandneopterin concen-tration inthe sample isdetermined byinterpolationfrom thestandardcurve.
Statistical
analysis
StatisticalanalysiswasperformedusingStatisticalPackage for Social Sciences (SPSS) version 16 for Windows (SPSS Inc.,Chicago,IL,USA).Continuousdatawereexpressedas mean±standarddeviationandwerecomparedbyStudent’s
t-test.Pearson’scorrelationanalysiswasconductedto eval-uate the association between continuous exposure and continuous covariates. Multiple linear regression analysis wasperformedtoidentifytheinfluenceof multiple varia-bles(age,sex,ESR,CRP,TNF-␣,IL-6,MCP-1,andJADAS-27) on a dependent variable (serum neopterin). p<0.05 was consideredasstatisticallysignificant.
Results
The clinical characteristics of patients with JIA and con-trolsubjectsareshowninTable1.Nosignificantdifference wasdetected between patients and controls asregard to theirmeanages(p>0.05).However,significantdifferences werefoundbetweenbothgroupswithregardtothemeans of theother variables(Hgb,ESR, TNF-␣,IL-6, MCP-1,and neopterin;p<0.05).
Demographicandbiochemicalcharacteristicwere com-paredbetweenmalesandfemalesinthepatientsgroupin
Table2.MeanJADAS-27washigherinfemalesthaninmales andthe differencewassignificant (p<0.05),while no sig-nificant differences weredetected asregard totheother variables.
Table2 Demographic,biochemical,anddiseasecharacteristicsinmaleandfemaleJIApatients.
Males Females p
Mean±SD Mean±SD
Age(years) 12.15±3.25 11.96±2.76 >0.05
Hgb(g/dL) 9.54±3.36 9.16±2.49 >0.05
ESR(mm/h) 48.54±11.63 51.47±10.33 >0.05
CRP(mg/L) 9.72±2.47 12.47±3.43
TNF-␣(pg/mL) 8.24±1.46 10.04±1.37 >0.05
IL-6(pg/mL) 29.46±6.46 31.46±4.67 >0.05
MCP-1(pg/mL) 295.33±78.25 304.74±38.23 >0.05
Neopterin(nmol/L) 19.84±7.49 20.16±2.42 >0.05
JADAS-27 38.56±8.46 49.38±10.36 <0.05a
JIA,juvenileidiopathicarthritis;Hgb,hemoglobinconcentration;ESR,erythrocytesedimentationrate;CRP,C-reactiveprotein;
TNF-␣,tumornecrosisfactor-alpha;IL-6,interleukin-6;MCP-1,monocytechemoattractantprotein-1;JADAS-27,JuvenileArthritisDisease ActivityScore-27.
a p<0.05issignificant.
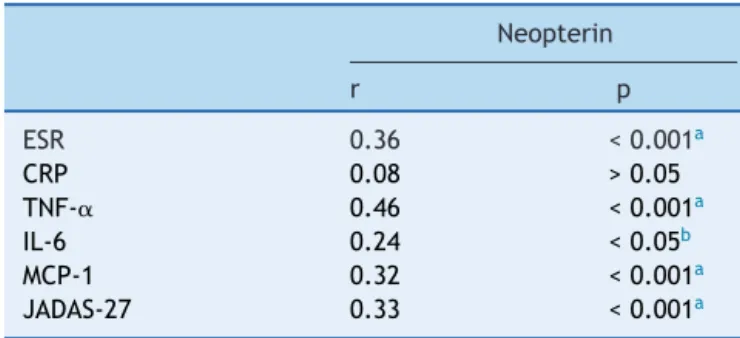
Table 3 Correlations between neopterin, inflammatory markers,anddiseaseactivityinJIApatients.
Neopterin
r p
ESR 0.36 <0.001a
CRP 0.08 >0.05 TNF-␣ 0.46 <0.001a
IL-6 0.24 <0.05b
MCP-1 0.32 <0.001a
JADAS-27 0.33 <0.001a JIA, juvenile idiopathic arthritis; ESR, erythrocyte sedimen-tation rate; CRP, C-reactive protein; TNF-␣, tumor necrosis factor-alpha; IL-6, interleukin-6; MCP-1, monocyte chemoat-tractantprotein-1;JADAS-27,JuvenileArthritisDiseaseActivity Score-27.
a p<0.001ishighlysignificant. b p<0.05issignificant.
MCP-1, and JADAS-27(p<0.05). No significant correlation wasfoundbetweenserumneopterinandCRP(p>0.05).
Multiplelinearregressionanalysisfortheassociationof the different variables with serum neopterin is shown in
Table 4.After adjustmentfor age, sex, andinflammatory markers,JADAS-27andESRwerethemainpredictorsforthe highneopterinlevelsseeninpatientswithJIA(p<0.05).The effectsofTNF-␣,IL-6,andMCP-1wereattenuatedbythe adjustmentofallthevariables.
Discussion
Thepresentstudyshowedahighlevelofserumneopterin, as a marker of macrophage activation, in patients with JIA.NeopterinandESRaresignificantpredictorsfordisease activityinsuchpatients.
Previous studies of neopterin in rheumatoid arthri-tis (RA) have been performed to define the relationship between neopterin and disease activity. Generally, they foundthatneopterincorrelatedwithdiseaseactivityinRA
Table4 Multiplelinearregression analysisfor neopterin anddifferentindependentvariables.
Variables Neopterin(r2=0.458)
Standardizedcoefficient t p
Age 0.065 0.710 >0.05
Sex -0.018 -0.203 >0.05
ESR 0.517 4.934 <0.001a
CRP 0.230 1.934 >0.05 TNF-␣ 0.164 1.842 >0.05 IL-6 0.315 1.872 >0.05 MCP-1 0.232 1.723 >0.05 JADAS-27 0.534 4.862 <0.001a JIA, juvenile idiopathic arthritis; ESR, erythrocyte sedimen-tation rate; CRP, C-reactive protein; TNF-␣, tumor necrosis factor-alpha; IL-6, interleukin-6; MCP-1, monocyte chemoat-tractantprotein-1;JADAS-27,JuvenileArthritisDiseaseActivity Score-27.
a p<0.001ishighlysignificant.
anddecreasedwithtreatment.11,13,14Incontrasttothe cur-rentstudy,apreviousstudyinJapandetectedsignificantly increasedneopterininSLE(SystemicLupusErythematosus) patients(p<0.001)butnotinRApatients.However,patients withRAhadagreaterconcentrationofneopterininsynovial fluid.15
kynurenine/tryptophan (r=0.486, p<0.05), erythrocyte sedimentation rate (r=0.472, p<0.05), and RF(r=0.478, p<0.05)inagroupofrheumatoidarthritispatients,which arein concordance withthe results of thepresent study. However,contrarytothepresent results,theyfound that neopterin and TNF-␣ levels did not showstatistically sig-nificant differences between patient and control groups. Thedifferencemaybeduetotheagerangeintheirstudy, asit was adult group. In 2013, D’agostino et al.18 evalu-atedneopterinplasmaconcentrationsinpatientswithearly Rheumatoid Arthritis (RA) and correlated them with dis-easeactivity.In agreementwiththepresent results, they detected a statistically significant elevation of neopterin mean concentration in early RA patients: mean value of 8.92±0.93 [3.94-28.3] nmol/L (p<0.001). Pearson prod-uct moment correlation suggested a correlation between neopterinconcentrations andDAS-28(r=0.208,p=0.065). Similar results were obtained by Arshadi et al.,19 who detected significantly higher level of neopterin in RA patients compared to healthy controls. Moreover, plasma neopterinlevelwasincreasedinpatientswithactivedisease andalsowascorrelatedwithdisease activity parameters. The previous results are in concordance withthe present study.Butincontrastwiththepresentstudy,theyfounda higherneopterin level in male RApatients versusfemale patients and a significant correlation of plasma level of neopterinwithagein boththeRAandcontrol group.The differenceintheresultsmaybeattributedtotheadultage groupof their study comparedto theyoung age group in the present study. Also Fagerer etal., in 2013,20 studied theinvolvementofspecificchemokinesintheinflammatory processofpatientswithRAandcardiovasculardiseasewith activatedproduction ofthepteridine neopterin.In agree-ment with the current study, they found high neopterin levelsin RApatientsandsignificantly elevated concentra-tionsof neopterin in patients withRA pluscardiovascular disease(CVD)comparedtoRAwithoutCVD(p<0.03).
Tothebestoftheauthors’knowledge,fewstudieshave includedbothneopterinasapro-inflammatorymarkerand JIA.TwostudiesfoundhighserumconcentrationsofIL-10, IL-18,IL-6,andneopterininpatientswithsystemicJIA com-plicated by macrophage activation syndrome. They found thatIL-10andIL-18remainedelevatedintheinactive dis-ease,whileotherpro-inflammatorycytokinesnormalizedin theinactivephaseofthedisease.21,22In2014,Brunneretal. 23evaluatedtryptophanasbiomarkerinJIAandcorrelated
itwithneopterininserumandsynovialfluid.Theydetected thatserumtryptophanshowednorelevantdifferenceinJIA patients vs. controls in contrast to neopterin, which was higherinpatientsthancontrols.
The presentstudy hasseverallimitations.It was cross-sectionalindesign,andthereforecausalinferencescannot be drawn. Also, the effects of the type of therapy on neopterinlevelswerenotfollowed,sotheiruseasamarker for therapy efficacy cannot be evaluated. The relatively smallsamplesizecouldberesponsibleforinadequate sta-tistical power. Larger sample sizes would have increased numberswithineachJIAcategory,allowingforgreater pre-cision inassessing the pro-inflammatorymarkers between categories.
In conclusion, the present study emphasized that macrophage activation, reflected by increased serum
neopterin, is a sensitive marker for assaying background inflammationanddiseaseactivityscoreinJIApatients.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
References
1.PhelanJD,ThompsonSD.Genomicprogressinpediatric arthri-tis: recent work and future goals. Curr Opin Rheumatol. 2006;18:482---9.
2.vanderHelm-vanMilAH,HuizingaTW.Advancesinthegenetics ofrheumatoidarthritispointtosubclassificationintodistinct diseasesubsets.ArthritisResTher.2008;10:205.
3.vanOosterhout M, Bajema I,LevarhtEW, Toes RE,Huizinga TW, van Laar JM. Differences in synovial tissue infiltrates betweenanti-cycliccitrullinatedpeptide-positiverheumatoid arthritisandanti-cycliccitrullinatedpeptide-negative rheuma-toidarthritis.ArthritisRheum.2008;58:53---60.
4.ScottDL,WolfeF,HuizingaTW.Rheumatoidarthritis.Lancet. 2010;376:1094---108.
5.PrakkenBJ,Albani S.Usingbiologyofdisease tounderstand and guide therapy of JIA. Best Pract Res Clin Rheumatol. 2009;23:599---608.
6.AdamsA,LehmanTJ.Updateonthepathogenesisand treat-mentofsystemiconsetjuvenilerheumatoidarthritis.CurrOpin Rheumatol.2005;17:612---6.
7.Hahn YS, Kim JG. Pathogenesis and clinical manifesta-tions of juvenile rheumatoid arthritis. Korean J Pediatr. 2010;53:921---30.
8.PettyRE,SouthwoodTR,MannersP,BaumJ,GlassDN, Gold-enberg J, et al. International League of Associations for Rheumatologyclassificationofjuvenileidiopathicarthritis: sec-ondrevision,Edmonton,2001.JRheumatol.2004;31:390---2. 9.GabrielSE,Michaud K. Epidemiological studiesinincidence,
prevalence,mortality, andcomorbidityoftherheumatic dis-eases.ArthritisResTher.2009;11:229.
10.HoffmannG,WirleitnerB,FuchsD.Potentialroleofimmune systemactivation-associated productionofneopterin deriva-tivesinhumans.InflammRes.2003;52:313---21.
11.SchroecksnadelK,FrickB,KaserS,WirleitnerB,Ledochowski M,MurE,etal.Moderatehyperhomocysteinaemiaandimmune activationinpatientswithrheumatoidarthritis.ClinChimActa. 2003;338:157---64.
12.ConsolaroA,RupertoN,BazsoA,PistorioA,Magni-ManzoniS, FilocamoG,etal.Developmentandvalidationofacomposite diseaseactivityscoreforjuvenileidiopathicarthritis.Arthritis Rheum.2009;61:658---66.
13.Beckham JC, Caldwell DS, Peterson BL, Pippen AM, Currie MS,Keefe FJ,etal. Diseaseseverity inrheumatoidarthritis: relationships of plasma tumor necrosis factor-alpha, soluble interleukin2-receptor,soluble CD4/CD8ratio,neopterin, and fibrinD-dimertotraditionalseverityandfunctionalmeasures. JClinImmunol.1992;12:353---61.
14.NasonovEL,SamsonovM.Iu,TilzG, FuchsD.Neopterin: new immunologicalmarkerofautoimmunerheumaticdisease.Klin Med(Mosk).2000;78:43---6.
15.HagiharaM,NagatsuT,OhhashiM,MiuraT.Concentrationsof neopterinandbiopterininserumfrompatientswithrheumatoid arthritisorsystemiclupuserythematosusandinsynovialfluid from patientswith rheumatoidor osteoarthritis.ClinChem. 1990;36:705---6.
systemiclupuserythematosusandrheumatoidarthritis. Arthri-tisCareRes(Hoboken).2011;63:535---41.
17.OzkanY,MeteG,Sepici-Dincel A,SepiciV,SimsekB. Trypto-phandegradationandneopterinlevelsintreatedrheumatoid arthritispatients.ClinRheumatol.2012;31:29---34.
18.D’agostinoLE,VentimigliaF,Verna JA,ColinaAdeL, Aguirre Y,ArturiA,etal. CorrelationbetweenDAS-28and neopterin asabiochemicalmarkerofimmunesystemactivationinearly rheumatoidarthritis.Autoimmunity.2013;46:44---9.
19.ArshadiD,NikbinB,ShakibaY,KianiA,JamshidiAR,Boroushaki MT.Plasmalevelofneopterinasamarkerofdiseaseactivityin treatedrheumatoidarthritispatients:associationwithgender, diseaseactivityandanti-CCPantibody.IntImmunopharmacol. 2013;17:763---7.
20.Fagerer N, Arnold M, Günther B, Werner K. Expression of neopterinandchemokinesinrheumatoidarthritisand cardio-vasculardisease.Pteridines.2013;22:7---12.
21.Shimizu M,Yachie A. Compensated inflammationinsystemic juvenile idiopathic arthritis: role of alternatively activated macrophages.Cytokine.2012;60:226---32.
22.ShimizuM,YokoyamaT,YamadaK,KanedaH,WadaH,Wada T, etal. Distinctcytokine profilesofsystemic-onsetjuvenile idiopathicarthritis-associatedmacrophageactivationsyndrome withparticular emphasison therole of interleukin-18 inits pathogenesis.Rheumatology(Oxford).2010;49:1645---53. 23.Brunner J,Giner T, WeissG, Fuchs D.Biomarkers of
ERRATUM
Erratum
on
‘‘Association
of
neopterin
as
a
marker
of
immune
system
activation
and
juvenile
rheumatoid
arthritis
activity’’
夽
Errata
de
‘‘Associac
¸ão
de
neopterina
como
marcador
de
ativac
¸ão
do
sistema
imunológico
e
atividade
da
artrite
reumatoide
juvenil’’
Mones
M.
Abu
Shady
a,
Hanan
A.
Fathy
b,
Alaa
Ali
a,
Eman
R.
Youness
c,∗,
Gihan
A.
Fathy
aaChildHealthDepartment,MedicalDivision,NationalResearchCentre,Cairo,Egypt
bResearchDepartment,NationalCentreforRadiationResearchandTechnology,AtomicEnergyAuthority,Cairo,Egypt
cMedicalBiochemistryDivision,NationalResearchCentre,Cairo,Egypt
Intheaffiliationoftheauthorsoftheoriginalarticle‘‘Associationofneopterin asamarkerofimmunesystemactivation andjuvenilerheumatoidarthritisactivity’’(JPediatr(RioJ).2015;91(4):352-357),whereitreads:
MonesM.AbuShadya,HananA.Fathyb,AlaaAlia,EmanR.Younessc,GihanA.Fathya
aChildHealthDepartment,MedicalDivision,NationalResearchCentre,Cairo,Egypt
bHealthResearchDepartment,NationalResearchCentre,Cairo,Egypt
cMedicalBiochemistryDivision,NationalCenterforRadiationandTechnology,Cairo,Egypt
itshouldread
MonesM.AbuShadya,HananA.Fathyb,AlaaAlia,EmanR.Younessc,GihanA.Fathya
aChildHealthDepartment,MedicalDivision,NationalResearchCentre,Cairo,Egypt
bResearchDepartment,NationalCentreforRadiationResearchandTechnology,AtomicEnergyAuthority,Cairo,Egypt