230 Rev. odonto ciênc. 2010;25(3):230-233
Received: February 27, 2010 Accepted: June 11, 2010
Conflict of Interest Statement: The authors state that there are no financial and personal conflicts of interest that could have inappropriately influenced their work.
Copyright: © 2010 Zastrow et al.; licensee EDIPUCRS. This is an Open Access article distributed under the terms of the Creative Commons Attribution-Noncommercial-No Derivative Works 3.0 Unported License.
Original Article
The relationship between nasopharyngeal
tonsil size and laboratory markers in
children infected with HIV
Relação entre tamanho da tonsila faríngea e marcadores
laboratoriais em crianças infectadas por HIV
Michella Dinah Zastrow a
Liliane Janete Grando b
Aroldo Prohmann de Carvalho c
Inês Beatriz da Silva Rath b
Maria Cristina Calvo d
a Discipline of Dental Radiology and Stomatology, University of South of Santa Catarina State, Tubarão, SC, Brazil
b Department and Stomatology, Federal University of Santa Catarina, Florianópolis, SC, Brazil c Department of Pediatric Infectology, Joana de Gusmão Hospital, Florianópolis, SC, Brazil d Department of Public Health, Federal University of Santa Catarina, Florianópolis, SC, Brazil
Correspondence: Liliane Janete Grando Departamento de Patologia, Centro de Ciências da Saúde Campus Universitário, Bairro Trindade Universidade Federal de Santa Catarina Florianópolis, SC – Brasil
88040-900
E-mail: [email protected]
Abstract
Purpose: The enlargement of nasopharyngeal tonsils, which leads to nasal obstruction and subsequent mouth breathing, can be caused by the presence of HIV. The aim of this research was to study nasopharyngeal tonsil sizes in HIV-infected children ranging from 6 to 13 years and to relate these findings to CD4+T-cell counts and viral loads.
Methods: Sixty children with HIV (mean age: 9 years and 8 months), infected by vertical transmission, had the sizes of their nasopharynx measured using lateral cephalometric radiographs, specifically focusing on the anatomical areas occupied by the nasopharyngeal tonsils. The children’s medical records were analyzed to assess information about TCD4+ lymphocyte count (%) and viral loads (log10).
Results: The mean value for the nasopharyngeal tonsil size percentage was 70.37%±14.07%. All of the children showed moderate or accentuated hypertrophy of nasopharyngeal tonsils. The average percentage of CD4+ T-cells among the 60 HIV-infected children was 35.01%±10.76%, whereas the mean value for the viral load was 3.35±1.08 log10.
Conclusion: There was no association between the size percentages of the nasopharyngeal tonsils (calculated against overall nasopharynx sizes) and CD4+ T-cell percentage (r=-0.0136;
P=0.298) or the viral load log10 (r=-0.033; P=0.805).
Key words: HIV; pediatric AIDS; adenoids; lateral cephalometric radiograph
Resumo
Objetivo: A presença do HIV na tonsila faríngea pode causar seu aumento de volume, ocasionando obstrução nasal e, consequentemente, respiração bucal. O objetivo deste trabalho foi avaliar o tamanho da tonsila faríngea em crianças infectadas pelo HIV por transmissão vertical, de 6 a 13 anos de idade, relacionando-a com a contagem de células T-CD4+ e carga viral (log10).
Metodologia: Sessenta crianças HIV positivo (idade média: 9 anos e 8 meses), infectadas por transmissão vertical, tiveram o tamanho da tonsila faríngea mensurado na radiografia cefalométrica lateral da face. Os prontuários das crianças foram analisados para buscar os dados de contagem de linfócitos T-CD4+ (%) e carga viral (log10).
Resultados: A média do tamanho da tonsila faríngea foi de 70,37%±14,07%. As crianças apresentaram hipertrofia da tonsila faríngea considerada moderada ou acentuada. A porcentagem média de linfócitos T-CD4+ nas 60 crianças foi de 35,01%±10,76% e a média da carga viral foi de 3,35±1,08 log10.
Conclusão: Não foi comprovada relação entre o tamanho da tonsila faríngea com a porcentagem de linfócitos T-CD4+ (r=-0,0136; P=0,298) e com a carga viral (r=-0,033;
P=0,805).
Rev. odonto ciênc. 2010;25(3):230-233 231
Zastrow et al.
Introduction
The nasopharyngeal lymphoid tissue contributes to the chronic replication of HIV-1 (1). HIV-1-infected cells were found in the lymphoepitelium of the nasopharyngeal tonsil invaginations. These tissues serve as a reservoir and a vehicle for virus dissemination.A child’s nasopharyngeal tonsil has a greater number of lymphocytes than adult tonsils (2). The nasopharyngeal tonsillar tissue, a secondary lymphoid
tissue, reveals the stage of immunodeiciency of
HIV-infected patients and can also provide additional information about the evolution and response of the affected patients to their treatment (3).A large increase in the amount of nasopharyngeal tonsillar tissue can be found in magnetic resonance images of HIV-infected adults, especially when they are compared to those of healthy people (4). A study of lateral radiographs of the nasopharynx in HIV-infected and non-infected children using the adenoid-nasopharyngeal (AN) ratio showed a correlation between the increase in tonsillar tissue and the different stages of the disease (5). The increase of the nasopharyngeal tonsil size due to the
presence of HIV may reduce nasal low, obstruct the upper
respiratory tract and lead to mixed or orally-substituted breathing patterns. During examination of breathing
function, patients’ lips and jaw postures are veriied and an
overall assessment is conducted to determine lip compe- tence (6).Furthermore, oral breathing may cause breathing complications such as pneumonia, which is considered one of the main causes of mortality in HIV-infected children (7-9).
The aim of this research was to study nasopharyngeal tonsil sizes in HIV-infected children ranging from 6 to 13 years in
age to relate these indings to CD4+ T-cell counts (%) and
viral loads – VL (log10).
Methodology
This research was approved by the Institutional Committee
for Ethics in Research with Human Beings. A descriptive transversal studywith 60 children with HIV who were infected by vertical transmission (ages 6 to 14 years old) was conducted in the Infectology Section of Joana de Gusmão
Children Hospital (HIJG) in Florianópolis, Santa Catarina, Brazil. Children with past history of tonsillectomies, cleft
lips or cleft palates were excluded. After an interview was conducted and the procedures to be accomplished were explained, all children participated in the research under the free informed consent of their parents and/or legal representatives.
Clinical diagnosis
A pediatrician and a dentist were responsible for the clinical examination of the children. When necessary, patients were submitted for a radiographic examination. The clinical
indication for a radiographic evaluation involved patients presenting with mouth breathing (6,9) and was based on the following criteria:
a) Alterations of behavior during the daytime: uneasiness, bad humor, impatience, disturbances, lack of concentration in daily activities, hyperactivity, aggressiveness, fatigue and sleepiness during the day, all of which may be suggestive of mouth breathing.
b) Collected data showing occurrences of allergic rhinitis,
sinusitis, bronchitis, constant colds, tonsillitis and reports on increases in the size of the nasopharyngeal tonsils.
c) Collected data showing nocturnal behaviors, such as night
mouth breathing, snoring, sleeping position, enuresis and excessive slobbering on the pillow.
Methods of radiographic analysis
The children were submitted for a lateral cephalometric radiographic examination (J. Morita Veraview®, Kyoto,
Japan) at the dental radiographic service. The radiographs were obtained following the recommended patterns and were automatically processed (Processor REVELL®, São Paulo,
SP, Brazil).
Two examiners, specialists in dentomaxillofacial radiology, were alocated to perform the evaluation of the radiographic
images (agreement value of 95% and an intra-examiner error value of 5%). The radiographs were numbered and masked
by another professional before analysis.
Pictures of anatomical structures of interest were transferred to an acetate paper with the aid of masks, allowing the subsequent
identiication of reference points and the construction of
lines and planes to analyze the nasopharyngeal space (10). After the manual tracing was done, the radiographs were
scanned (Scanner HP Scanjet 4C/T®, São Paulo, Brazil),
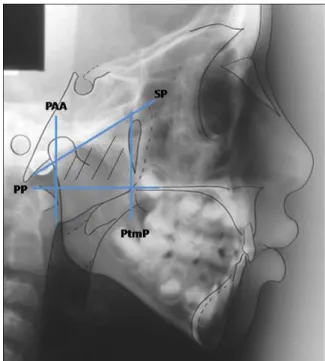
and the images were transferred to a cephalometric analysis software. To identify the degree of obstruction of the aerial tract in the lateral view and to determine the nasopharyngeal space occupied by the nasopharyngeal tonsil, a measurement of the adenoid percentage was utilized, according to previous studies (11-13). The total nasopharyngeal size was obtained through the calculation of a trapezoid area. The four sides of the trapezoid are represented by four planes: the Palate
Plane (PP), which goes through the anterior nasal spine
(ANS) and posterior nasal spine (PNS); the Sphenoid Plane
(SP) that goes through the basio point (Ba) and the sphenoid bone adjacent plane; the plane PAA that goes through the most anterior point of the Atlas Vertebra (AA); and the
Pterygomaxillary Plane (PtmP), a space perpendicular to
the palate area that goes through the PPtm/PNS (Fig. 1). The nasopharyngeal plane was determined mathematically using the nasopharyngeal depth, the nasopharyngeal height, and the angle formed between the sphenoid and the palate planes, as shown in Figure 1. The aerial portion was measured using a polar planimetric compensation, whereas the plane occupied by the nasopharyngeal tonsil was obtained by subtracting the aerial portion from the total nasopharyngeal area. A
modiied software (Radiocef, developed by Radiomemory®,
232 Rev. odonto ciênc. 2010;25(3):230-233
Nasopharyngeal tonsil size in children infected with HIV
The results of the VL (log10) and TCD4+ lymphocytes
counts (%) are shown in Table 1. The average percentage of TCD4+ lymphocytes was 35.01% ± 10.76 and the VL algorithm was 3.25 ± 1.08.
Pearson’s correlation showed no correlation between the
TCD4+ lymphocyte count and the percentage of obstruction
of the nasopharyngeal area by the nasopharyngeal tonsil (r=0.136; P=0.298). Similarly, no correlation was found
between the VL log10 and the percentage of obstruction of
the nasopharynx by the nasopharyngeal tonsil (r=0.033;
P=0.805).
Fifty eight children (96.7% of the children in the studied
sample) were under some type of therapeutic plan. The great majority utilized an association of Zidovudin (AZT),
Didanosine (ddI) and Sulphametoxazol + Trimetoprime (SMX + TMT or Bactrim). Two children lacked any
clinical indication to utilize a therapeutic plan, because they
presented with non-detectable VL counts and adequate CD4+
lymphocyte counts.
Discussion
The dentist plays an important role in the treatment of HIV-infected patients, which includes the maintenance of a good oral health and assistance in improving their overall quality of life. It is essential that the dentist be able to recognize all of the manifestations of HIV/AIDS that can affect patients (14).
An increase in the size of the nasopharyngeal tonsil can result in an alteration of breathing dynamics, which makes
breathing through the nose more dificult, causes a decrease in nasal air low, and results in patients’ adoption of temporary or deinitive substitute breathing methods, such as through
the mouth (1,9,15-21).
Almost 91.7% of the children in this study showed an
obstructed nasopharyngeal space by the nasopharyngeal
tonsil of greater than 50%, which corresponds to a
moderate or increased obstruction. Nasal obstruction and mouth breathing, as discussed in Nishimura & Suzuki (22) and Kobayashi et al. (23), can cause snoring, nocturnal slobbering, hypotonicity of the upper lip, ogival-shaped hard palates, narrow nostrils, and tension of the mentalis muscles.
HIV-infected children present with a greater occurrence of upper respiratory tract infections and repetitive colds that can, despite being temporary, also be responsible for nasal obstruction and may contribute to changes in the assessment
of nasal air low (19).Yousem et al. (4)and Benito et al. (5) detected an increase in nasopharyngeal tonsil sizes in HIV-infected patients.
Fig. 1. Planes that define the trapezoid for calculation of the
nasopharyngeal area: palate plane (PP), sphenoid plane (SP), plane going through AA (PAA) and pterygomaxillary plane (PtmP).
space (≤ 50%) was considered to be normal sized; a tonsil
obstructing between 50% and 75% of the nasopharyngeal
space was considered moderate sized; and a tonsil obstructing
more than 75% of such space was considered enlarged (8).
Data from medical records
Based on an analysis of the children’s records, medicinal intake values and both absolute and relative counts of
TCD4+ lymphocytes and VL (log10) were obtained. The results obtained were submitted to descriptive statistical analyses involving a chi-squared test (t), and Pearson’s correlation.
Results
In the total sample consisting of 60 HIV-infected children,
33 were female (55%) and 27 were male (45%). Their ages
ranged from 5 years and 1 month to 13 years and 7 months
(average age: 9 years and 8 months). According to radio-graphic analyses, 5 children (8.3%) presented normal
per-centage values for their nasopharyngeal areas obstructed by
nasopharyngeal tonsils (below 50%), 34 (56.7%) presented moderate obstruction of the nasopharynx (from 50% to 75%), and 21 (35%) presented increased obstruction (above 75%).
Table 1. Descriptive analysis of age,
percentage of nasopharyngeal area obstructed by nasopharyngeal tonsil (PT), TCD4+ lymphocytes count and VL log10 in HIV infected children.
Characteristics Minimum Maximum Average Pattern deviation
AGE 6.10 13.60 9.69 1.92
% of PT Occupation 29.07 99.45 70.37 14.07
Viral Load log10 1.90 5.36 3.25 1.08
Rev. odonto ciênc. 2010;25(3):230-233 233
Zastrow et al.
In the present study, no correlation was seen between the percentage of obstruction of the nasopharyngeal area by the
nasopharyngeal tonsil and TCD4+ lymphocyte counts or VL counts. These indings are in agreement with the results
obtained by Yousem et al. (4).
With regard to the clinical stage of HIV, as well as to the level
of immunodeiciency, it was found that the children in the
present study were well controlled with the utilization of anti-
retroviral medicine and presented an average TCD4+ lym-phocyte count of 35% and an average VL log10 of 3.25%. These
values are considered ideal clinical presentations without
symptoms, which speciically involve a relative TCD4+ lymphocyte count above 25% and a VL log10 below 2.00 (24).
All but two (3.3%) children were taking medicines and
showing a positive treatment acceptance. Furthermore, the increase in the size of the nasopharyngeal tonsil may be considered a physiological function of their ages. Therefore, the size increase of the nasopharyngeal tonsil may simply be a response of the immune system to the HIV infection stimulus, as sometimes happens with other
diseases. The results may be a relection of the positive
treatment acceptance, represented here by the responsibility of the caretaker and the good conditions of the children who comprised the sample.
Considering the role of the nasopharyngeal tonsil as a
lymphoid tissue and a reservoir site for HIV, the clinical and radiographic evaluation of the tonsil can be utilized as a clinical criterion for the evaluation of disease progression. This evaluation constitutes a possible means of an early
identiication of the infection in patients during clinically
latent periods. Although this study does not show a correlation between the increased nasopharyngeal tonsil sizes with
the TCD4+ lymphocyte and VL log10 counts, the authors believe that radiographic analyses of the nasopharyngeal tonsils may represent an effective criterion for the evaluation of disease progression, because it provides easy access,
suficient acceptance by the patients, and simple technical
execution.
Conclusions
The study indings reveal the following conclusions: • HIV-infected children presented moderate-to-large
increases in their nasopharyngeal tonsil sizes.
• No correlation was established between the increased sizes of the nasopharyngeal tonsils and the TCD4+ lymphocytes (%) or VL (log10) counts.
References
1. Foxen EHM. Conceitos básicos em otorrinolaringología. 4th ed. São Paulo: Andrei; 1982.
2. Wenig BM, Thompson LD, Frankel SS, Burke AP, Abbondanzo SL, Sesterhenn I et al. Lymphoid changes of the nasopharyngeal and palatine tonsils that are indicative of human immunodeficiency virus infection. A clinicopathologic study of 12 cases. Am J Surg Pathol 1996;20:572-87.
3. Lopez-Gonzalez MA, Lucas M, Sanchez B, Mata F, Delgado F. Adenoidal and tonsillar lymphocyte subsets in AIDS children. Int J Pediatr Otorhinolaryngol 1998;45:215-22.
4. Yousem DM, Loevner LA, Tobey JD, Geckle RJ, Bilker WB, Chalian AA. Adenoidal width and HIV factors. AJNR Am J Neuroradiol 1997;18:1721-5.
5. Benito MB, Sampelayo TH, Gurbindo D, Sánchez-Ramón S, Gómez EM, Muñoz-Fernández MA. Adenoidal tissue mass as a clinical guide of disease evolution in vertically HIV-1 infected children. Int J Pediatr Otorhinolaryngol 1999;51:145-55.
6. Marchesan IQ. Fundamentos em Fonoaudiologia: aspectos clí- nicos da motricidade oral. Rio de Janeiro: Guanabara-Koogan; 1998.
7. Legott PJ. Oral manifestations in pediatric HIV infection. In: Greenspan JS, Greenspan D. Oral manifestations of HIV infection. Chicago: Quintessence, 1995. p. 234-9.
8. Ministério da Saúde. Aprenda sobre HIV e aids. [Acessed on 2006 Jan 23]. Available at http://www.aids.gov.br
9. Carvalho GD. SOS respirador bucal: uma visão funcional e clínica da amamentação. São Paulo: Lovise; 2003.
10. Freitas A, Rosa JE, Souza IF. Radiologia odontológica. 4th ed. São Paulo: Artes Médicas; 1998.
11. Handelman CS, Osborne G. Growth of the nasopharynx and adenoid development from one to eighteen years. Angle Orthod 1976;46:243-59.
12. Poole MN, Engel GA, Chaconas SJ. Nasopharyngeal cephalometrics. Oral Surg Oral Pathol 1980;49:266-77.
13. Rickets RM, Bench RW, Gugino CF, Hilgers JJ, Schulhof RJ. Técnica bioprogressiva de Ricketts. Buenos Aires: Panamericana; 1998 14. Correa EMC, Andrade ED. Tratamento odontológico em pacientes
HIV/AIDS. Rev Odonto Ciênc 2005;20:281-9.
15. Cassano P, Gelardi M, Cassano M, Fiorella ML, Fiorella R. Adenoid tissue rhinopharyngeal obstruction grading based on fiberendoscopic findings: a novel approach to therapeutic management. Int J Pediatr Otorhinolaryngol 2003;67:1303-9.
16. Donnely LF, Casper KA, Chen B. Correlation on cine MR imaging of size of adenoid and palatine tonsils with degree of upper airway motion in asymptomatic sedated children. AJR Am J Roentgenol 2002;179:503-8.
17. Frankel SS, Tenner-Racz K, Racz P, Wenig BM, Hansen CH, Heffner D et al. Active replication of HIV-1 at the lymphoepithelial surface of the tonsil. Am J Pathol 1997;151:89-96.
18. Haapaniemi JJ. Adenoids in school-aged children. J Laryngol Otol 1995;109:196-202.
19. Hungria H. Otorrinolaringologia. 8th. Rio de Janeiro: Guanabara-Koogan, 2000.
20. Steele CH, Fairchild RC, Ricketts RM. Forum on the tonsil and adenoid problem in orthodontics. Am J Orthod 1968;54: 485-507.
21. Valera FCP, Travitzki LVV, Mattar SEM, Matsumoto MAN, Elias AM, Anselmo-Lima WT. Muscular, functional and orthodontic changes in pre school children with enlarged adenoids and tonsils. Int J Pediatr Otorhinolaryngol 2003;67:761-70.
22. Nishimura T, Suzuki K. Anatomy of oral respiration: morphology of the oral cavity and pharynx. Act Otolaryngol 2003;Suppl:25-8. 23. Kobayashi M, Sakaida H, Yuta A, Takeuchi K, Shimizu T, Majima
Y. Therapeutic results of respiratory disturbance during sleep in children. Nippon Jibiink. Gakkai Kaiho 2003;106:815-22. 24. Centers for Disease Control and Prevention. Revised classification