BASIC RESEARCH
IN VIVO OBSERVATION OF MESENTERIC
LEUKOCYTE-ENDOTHELIAL INTERACTIONS AFTER
CECAL LIGATION/PUNCTURE AND SURGICAL
SEPSIS SOURCE CONTROL
Naomi K. Nakagawa, José Jukemura, Priscila Aikawa, Rafael A. Nogueira, Luiz F. Poli-de-Figueiredo and Paulina Sannomiya
Nakagawa NK; Jukemura J, Aikawa P, Nogueira RA, Poli-de-Figueiredo LF, and Sannomiya P. In vivo observation of mesenteric leukocyte-endothelial interactions after cecal ligation/puncture and surgical sepsis source control. Clinics. 2007;62(3):321-6.
PURPOSE: Cecal ligation and puncture (CLP) has been used as a useful model for the induction of polymicrobial sepsis. Necrotic tissue resection and peritoneal lavage (REL) are the surgical procedures for controlling perforated appendicitis. The aim of this study was to evaluate leukocyte-endothelial interactions in the rat mesentery in vivo after CLP and REL.
METHODS: Thirty-seven male Wistar rats (250-300 g) underwent laparotomy and were randomly assigned to the following groups: 1) SHAM; 2) CLP: animals submitted to CLP, 3) CLP+REL: animals submitted to CLP and REL. Mesenteric leukocyte-endothelial interactions were studied by intravital microscopy assessed once in each animal (3-5 postcapillary venules, 15-25 µm diameter) 24 hours after intervention. Follow-up was performed in all animals; this included analysis of glycemia, lactate, hematocrit, white blood cell count as well as a functional score that was the sum of scoring on the following parameters: alertness, mobility, piloerection, diarrhea, encrusted eyes, and dirty nose and tail.
RESULTS: None of the animals showed significant changes in body weight (265 ± 20 g) or in hematocrit levels (46% ± 2%) during the experimental protocol. Compared to SHAM animals, CLP animals showed an increased number of rolling (2x), adherent, and migrating leukocytes (7x) in the mesenteric microcirculation, an increase in blood glucose (136 ± 8 mg/dL), lactate (3.58 ± 0.94 mmol/L), white cell count (23,570 ± 4,991 cells/mm3) and functional alterations (score 11 ± 1), characterized by impaired
alertness and mobility, and presence of piloerection, diarrhea, encrusted eyes, and dirty nose and tail. The REL procedure normalized the number of rolling, adherent, and migrated leukocytes in the mesentery; glycemia; lactate; and white blood cell count. The REL procedure also improved the functional score (7 ± 1).
CONCLUSION: Local and systemic inflammation was induced by CLP, while REL completely overcame the inflammatory process.
KEYWORDS: Intravital microscopy. Microcirculation. Mesentery. Peritonitis. Sepsis.
INTRODUCTION
Sepsis is a syndrome involving the systemic host response to an inflammatory or infectious stimulus. It is often asso-ciated with tissue injury that may lead to multiple organ fail-ure.1 Sepsis is still one of the leading causes of infectious
death.2–4 The introduction of antibiotics more than 50 years
ago and improvement in hemodynamic support resulted in a decline of sepsis-induced mortality.3-5 However, the
suc-cessful treatment of sepsis in critical care and emergency settings remains a relevant medical challenge.
Extensive experimental and clinical data have been amassed to determine the mechanism(s) of sepsis-induced multiple organ dysfunction and failure. Laparotomy compli-cated by sepsis is a common clinical presentation of sepsis. The rationale for selecting cecal ligation and puncture (CLP) as a septic model is that CLP induces effects analogous to a Research Division, Heart Institute, InCor, and LIM11, University of Sao
Paulo School of Medicine- São Paulo/SP, Brazil Email: [email protected]
perforated appendicitis.3,6,7 The infectious milieu promotes
microbial growth or impairs host defenses, which can be quickly modified by surgical source control.3,5 Therefore,
sur-gical removal of the septic focus has been applied as a key component of success in therapy of ongoing sepsis.
In animal models of sepsis, microcirculatory dysfunction is seen prior to the onset of shock.8–9 The host defense responses
to sepsis may promote a generalized increase in leukocyte re-cruitment and accumulation in the tissues, which may lead to subsequent endothelial damage, leaky capillaries, and organ dysfunction and failure. Recruitment and accumulation of leukocytes in tissues involves a series of leukocyte-endothelial interactions in postcapillary venules mediated by the expres-sion of adheexpres-sion molecules on the vascular endothelium and leukocytes. Rolling is the first step of leukocyte recruitment, mediated by E-, P-, and L-selectins. This early type of leukocyte-endothelial interaction is a prerequisite for adhesion of leukocytes to the endothelium mediated by the interaction ofβ2 integrins (CD11/CD18) on leukocytes and intercellular adhesion molecule (ICAM)-1 on the endothelial cell. Leukocytes eventually transmigrate into the interstitial compart-ment and towards the site of injury.10–12
The aim of the current study was to evaluate in vivo leukocyte-endothelial interactions in rat mesenteric micro-circulation by intravital microscopy in a septic state and after surgical source control.
MATERIALS AND METHODS
Experimental Protocol
This study was approved by our institutional Ethics Com-mittee and performed according to National Institutes of Health Guidelines on the experimental use of animals. Thirty-seven male Wistar rats (weighing 250-300 g) were fasted over-night, allowed water ad libitum, and housed in standard-care facilities before the experiments. A 12-hour light/dark cycle with ambient temperature control was employed. The animals were randomly assigned to 3 groups: 1) SHAM: animals were submitted solely to laparotomy; 2) CLP: animals underwent cecal ligation/puncture, 3) CLP+REL: animals were submit-ted to cecal ligation/puncture and to necrotic cecal resection/ peritoneal lavage, 24 hours apart. Mesenteric microcirculation was evaluated in 19 animals (SHAM, n = 6; CLP, n = 7; and CLP+REL, n = 6). Mortality rate was evaluated in 18 ani-mals (SHAM, n = 6; CLP, n = 6; and CLP+REL, n = 6).
Laparotomy
All animals underwent anesthesia by means of an in-traperitoneal sodium pentobarbital injection (50 mg/kg)
followed by a 2-cm abdominal midline incision. The cecum was exposed and returned to the abdomen, which was closed in 2 layers with 5.0 suture (Ethicon, NJ, USA). Ani-mals were kept warm for 1 hour at 37oC and returned to
their cages with free access to food and water.
Cecal Ligation and Puncture (CLP)
Animals underwent anesthesia by means of an intraperi-toneal injection of sodium pentobarbital (50 mg/kg) followed by laparotomy. The cecum was exposed, isolated just distal to the ileocecal valve to avoid intestinal obstruction, and punc-tured twice with a 22-gauge needle. The puncpunc-tured cecum was squeezed to expel a small amount of fecal material through the holes. The bowel was returned to the abdomen, which was closed in 2 layers with 5.0 suture (Ethicon, NJ, USA). Ani-mals were kept warm for 1 hour at 37oC and returned to their
cages with free access to food and water.
Necrotic Tissue Resection and Peritoneal Lavage (REL)
Animals were anesthetized with intraperitoneal sodium pentobarbital (50 mg/kg), and the midline abdomen inci-sion was opened. The necrotic cecum was resected, and the bowel was returned into the abdomen. Subsequently, peri-toneal lavage with 40 mL of warmed isotonic saline solu-tion (0.9% NaCl) was performed. The midline incision was closed in 2 layers with 5.0 suture (Ethicon, NJ, USA). The animals were kept warm- for 1 hour at 37oC and returned
to their cages with free access to food and water.
Intravital Microscopy of the Mesentery
Intravital microscopy of the mesentery was performed once in each animal, 24 hours after intervention. Briefly, the animals were anesthetized with an intraperitoneal sodium pentobarbi-tal injection (50 mg/kg). After an abdominal midline incision, the distal ileum and its accompanying mesentery were exposed for in vivo microscopic examination of the microcirculation. The animals were maintained on a specially designed stage warmed by circulating water kept at 37oC. The stage has a
trans-parent platform on which the tissue to be transilluminated was placed. The mesentery was continuously perfused during the study period with a warmed (37oC) Ringer-Locke solution (154
mM NaCl, 5.6 mM KCl, 2 mM CaCl2.2 H2O, 6 mM NaHCO3, 5 mM glucose, pH 7.20-7.40), saturated with a mixture of gases (95% N2and 5% CO2). This procedure kept the microcircula-tory characteristics unchanged throughout the intravital micro-scopic analysis.11 After 10 minutes of stabilization, the
(TK-C1380U, JVC Co, Tokyo, Japan) was incorporated with a triocular microscope (Axioplan 2, Carl Zeiss Co, MünchHallbergmoos, Germany) to facilitate the observation of the en-larged image (425x) on a microcomputer monitor (SyncMaster 753DFX, Samsung, Manaus, Brazil). Analyses of leukocyte-en-dothelium interactions were performed on-line, by using im-age analysis software (Axiovision 4.1, Carl Zeiss Co, München-Hallbergmoos, Germany) with an incorporated module for in-teractive measurements and time-lapse analysis. Images were stored, enabling off-line playback analysis. After intravital microscopy, animals were sacrificed with an overdose of so-dium pentobarbital.
Rolling Leukocytes
Rolling leukocytes were defined as white blood cells that move at a velocity significantly slower than that of erythrocytes in a given microvessel.11,12 The number of
roll-ing leukocytes was presented as the mean number of cells passing a designated line perpendicular to the venular axis per 10 minutes. A given section of the vascular bed was tested only once, and 3 to 5 microvessels were selected from each animal.
Adherent Leukocytes
A leukocyte was considered to be adherent to the venu-lar endothelium if remained stationary for >30 seconds.13– 15Adherent cells were counted during a 10 minute-period
in a 100 µm segment of the vessel, with 3 to 5 microvessels being selected from each animal.
Migrated Leukocytes
The number of leukocytes accumulating in the connec-tive tissue adjacent to the chosen postcapillary venule was determined13 in a standard area of 5,000 µm2; 3 to 5
dif-ferent fields were evaluated for each microvessel, and 3 to 5 microvessels were selected from each animal.
White Blood Cell Count, Hematocrit, Glycemia, and Blood Lactate
Blood samples were collected from the rats’ tails. Hematocrit was measured by using microcapillary tube cen-trifugation. White blood cells were counted in Neubauer chambers. Glycemia was determined with a blood glucose monitor (Advantage®, Roche Diagnostics Co., IN, USA).
Blood samples were collected from the abdominal aorta for lactate measurement, using a blood gas analyzer (Radiom-eter ABL 555, Radiom(Radiom-eter Medical, Kopenhagen, Denmark).
Functional Score
Animals were followed up at 24-hours after interventions. Functional features were scored as follows: alertness (grade: 1, normal; 2, low attention; 3, very low attention), mobility (grade: 1, normal; 2, low motion; 3, motionless), piloerection (grade: 0, none; 1, mild piloerection; 2, moderate piloerection; 3, severe piloerection), diarrhea (grade: 0, none; 1, moderate; 2, severe diarrhea), encrusted eyes, dirty nose and tail (grade: 0, none; 1, one place; 2, two places; 3, three places; 4, four places). The sum of these scores determined the overall functional score, which ranged from 2 (normal condition) to 15 (the most severe condition).
Statistical Analysis
The data are expressed as mean ± standard deviation of the mean (SD). Comparisons among groups were per-formed with Kruskal-Wallis analysis, and differences were tested by Dunn’s test. All statistics were calculated using a standard computer software package (SigmaStat v4.1/ Jandel Scientific, Corte Madera, CA, USA). Values of P
less than .05 were considered statistically significant.
RESULTS
Rats submitted to CLP exhibited decreased alertness and mobility, presence of piloerection, diarrhea, encrusted eyes, and dirty nose and tail, which resulted in a functional score of 11, compared to the normal condition of SHAM rats (func-tional score, 2; Table 1). Debris around the injured tissue and ascitis were also observed in the CLP group. In addition, CLP induced increases in glycemia (136 ± 8 mg/dL, P < .05), blood
lactate (3.58 ± 0.94 mmol/L, P< .05) and total white blood
cell counts (23,571 ± 4,991 cells/mm3,P < .05) compared to
SHAM (Table 2). There were no significant changes in body weight, or hematocrit. When submitted to surgical source con-trol, CLP+REL animals showed an improvement in functional condition (score 7 ± 1). Glycemia (97 ± 9 mg/dL), and lac-tate (1.70 ± 0.27 mmol/L). White blood cell count (14,850 ± 1,459 cells/mm3) were reduced to values attained in SHAM
rats. Additional experiments performed to evaluate mortality rate showed that all CLP animals (n = 6) died after day 5, whereas all SHAM (n = 6) and CLP+REL animals (n = 6) survived up to day 30.
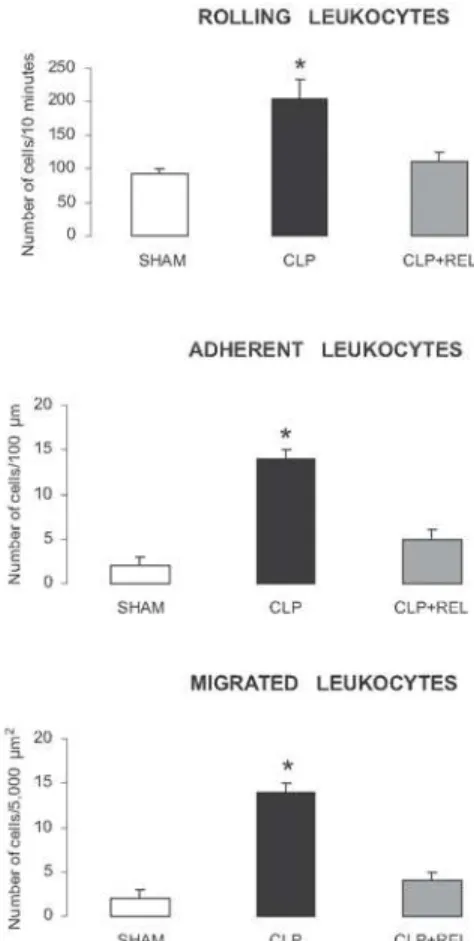
The number of rolling, adherent, and migrated leukocytes were markedly increased in rats submitted to CLP (P < .05). Rolling increased twofold, and adhesion
and migration sevenfold compared with SHAM rats (Fig-ure 2). Surgical source control reduced the number of roll-ing (110 ± 16 cells/10 minutes), adherent (5 ± 1 cells/100 µm venule length), and migrated leukocytes (4 ± 1 cells/ 5,000 µm2) to values observed in SHAM rats.
DISCUSSION
Although intra-abdominal infection has been recognized as a common disease, the mortality and morbidity for com-plicated cases remains high. Consequently, understanding of the pathophysiology including local, regional, and systemic host defense alterations is required to focus future efforts in the management of this disease. The current study evaluated systemic alterations after cecal ligation/puncture and surgical source control; leukocyte-endothelial interactions in me-senteric postcapillary venules - through intravital microscopy. The CLP procedure produces definite metabolic, immuno-logic, and physiologic alterations such as hyperglycemia; leukocytosis; increased number of rolling, adherent, and mi-grated leukocytes in the mesentery; decreased alertness and mobility; and presence of intense piloerection, diarrhea,
en-crusted eyes and dirty nose and tail. Surgical source control completely overcame the inflammatory process in the mesen-tery by reducing leukocyte rolling, adhesion, and migration in parallel with systemic functional improvement.
Neutrophils may exert damaging effects through several mechanisms. After activation, these cells generate and release toxic oxygen metabolites, numerous proteases, and phospholi-pase products, all of which may result in vasomotor changes, endothelial injury, and loss of vascular integrity.16
Leukocyte-endothelial interactions are mediated by a variety of glycoproteins expressed on the surface of leukocytes and en-dothelial cells. The accumulation of leukocytes in inflamed tissues is preceded by leukocyte rolling and adhesion to the vascular endothelium.10,17 Leukocytes roll along the walls of
postcapillary venules mediated by the selectin family of ad-hesion molecules. 12Leukocytes become firmly adherent to the
vascular wall by the interaction between ICAM-1 on endothe-lial cells with β2 integrins (CD11/CD18) on leukocytes. The CLP procedure partially destroys the normal barrier of the gastrointestinal tract.18 Previous studies in rats using
intravi-Figure 1 - Representative photomicrographs of rat mesenteric microcirculation from SHAM (SHAM-operated), CLP (cecal ligation/ puncture), and CLP+REL (cecal ligation/puncture and necrotic tissue resection/peritoneal lavage) animals. Increased number of adherent and migrated leukocytes are observed in the CLP in comparison with SHAM and CLP+REL groups. Final magnification: 850x.
Table 1 - Functional features 24 hours after intervention
SHAM CLP CLP+REL
(n = 12) (n = 13) (n = 12)
Alertness 1 ± 0 2 ± 1 1 ± 0
Mobility 1 ± 0 2 ± 1 1 ± 0
Piloerection 0 ± 0 2 ± 1 2 ± 1
Diarrhea 0 ± 0 2 ± 0 0 ± 0
Encrusted eyes /Dirty nose and tail 0 ± 0 3 ± 1 2 ± 1
Functional score 2 ± 0 11 ± 1* 7 ± 1
Kruskal-Wallis analysis and Dunn´s test, *P< .05 vs other groups. Functional
score was the sum of the scores for the following parameters: alertness (1, normal; 2, low attention; 3, very low attention); mobility (1, normal; 2, low motion; 3, motionless); piloerection (0, none; 1, mild piloerection; 2, moderate piloerection; 3, severe piloerection); diarrhea (0, none; 1, moderate diarrhea; 2, severe diarrhea); encrusted eyes, dirty nose and tail (0, none; 1, one place; 2, two places; 3, three places; 4, four places). Functional scores ranged from 2 (normal condition) to 15 (the most severe condition).
Figure 2 - Number of rolling leukocytes/10 minutes, adherent leukocytes/ 100 µm venule length, and migrated leukocytes/5,000 µm2in rat mesenteric
tal microscopy have demonstrated that sepsis induces micro-circulatory derangements in the ileal mucosa19 and in other
organs not directly affected by the septic process, such as the extensor digitorum longus muscle20 and cremaster muscle.21
Normotensive sepsis induces absence of capillary flow22 and
alters leukocyte-endothelium interactions.19,23,24Smalley et al24
found that CLP significantly increases leukocyte adherence and infiltration into the upper gastrointestinal tract 4 hours after intervention. Because any sham procedure performed in ani-mal models of sepsis may result in a low degree of inflam-mation, we included a SHAM group. These animals presented results similar to those of naive animals (data not presented).
In the present study, increased number of rolling, adher-ent, and migrated leukocytes were observed in vivo in the mesentery of rats submitted to CLP. The increased recruitment of leukocytes to the site of injury represents a mechanism trig-gered by the host to produce an efficient defense against in-vading pathogens. However, if sepsis is not controlled, it may lead to multiple organ failure. Mortality in the CLP group was observed after the 5th day, but not in SHAM and CLP+REL
animals, which survived up to the 30th day.
An adequate surgical source control of the septic focus should be used to control the immune suppressive response to the ongoing sepsis. Therapeutic strategies such as drain-age of infected fluids, debridement of infected soft tissues,
and removal of necrotic tissues may play a crucial role in avoiding the development and progression of multiple or-gan failure.3,5 In the present report, REL normalized
leukocyte-endothelium interactions in the mesentery as in-dicated by the decreasing numbers of rolling, adherent, and migrated leukocytes in comparison to CLP. Surgical source control was effective in reducing inflammation after CLP, and in improving functional parameters, which were con-sidered to indicate the resolution of the induced sepsis.
In conclusion, the current experimental rat model al-lowed in vivo observations of leukocyte-endothelium in-teractions in the mesentery by intravital microscopy in par-allel with documenting functional alterations after cecal ligation/puncture and surgical source control.
ACKNOWLEDGMENTS
We wish to thank Dr. Sandra Regina Farsky, Dr. Maria Aparecida de Oliveira and Dr. Rita do Carmo de Jesus for technical assistance. The plate for intravital microscopic study of the rat mesentery was developed by Dr. Nakagawa, Dr. Simão Bacht and Dr. Idágene Cestari, and built by Bioengeering Division of Heart Institute (InCor) University of Sao Paulo Medical School. This work was supported by grant of PRONEX, FAPESP and CNPq 505319/2004-7.
Table 2 - General characteristics of study groups
SHAM CLP CLP+REL
(n = 6) (n = 7) (n = 6)
Body weight change/48 hours (g) 10 ± 5 -20 ± 10 10 ± 10
Glycemia (mg/dL) 101 ± 6 136 ± 8* 97 ± 9
Hematocrit (%) 46 ± 1 45 ± 1 46 ± 1
Blood lactate (mmol/L) 1.74 ± 0.48 3.58 ± 0.94* 1.70 ± 0.27
White blood cell count/mm3 11,825 ± 1,478 23,571 ± 4,991* 14,850 ± 1,459
Kruskal-Wallis analysis and Dunn´s test, *P < .05 vs other groups. General characteristics were similar among SHAM, CLP and CLP+REL groups, except
for glycemia, blood lactate, and white blood cell count, which increased in the CLP group. Data are presented as mean values ± SD.
RESUMO
Nakagawa NK; Jukemura J, Aikawa P, Nogueira RA, Poli-de-Figueiredo LF and Sannomiya P. Avaliação in vivo da interação leucócito-endotélio mesentérico após ligadura e punção cecal e remoção cirúrgica do foco sééptico. Clinics. 2007;62(3):321-6.
OBJETIVO: O procedimento de ligadura cecal e perfuração (CLP) tem sido usado como um modelo útil de indução de sepse polimicrobiana. A ressecção do tecido necrosado e lavagem peritoneal (REL) são procedimentos cirúrgicos freqüentemente utilizados para controlar uma
apendicite perfurada. O objetivo desse estudo foi avaliar
in vivo as interações leucócito-endotélio no mesentério de
ratos após a CLP e REL.
todos os animais, incluindo glicemia, lactato, hematócrito, número total de células brancas e um escore funcional, o qual foi considerado como a somatória dos seguintes parâmetros: estado de alerta, mobilidade, piloereção, diarréia, olhos encrustados, e nariz e cauda sujos.
RESULTADOS: Os animais não apresentaram alterações significantes no peso (265 ± 20 g) e hematócrito (46 ± 2%) ao longo do estudo. Comparados ao SHAM, os animais CLP apresentaram aumento no número de leucócitos em rolamento (2x), aderidos (7x) e migrados (7x) na microcirculação mesentérica, aumentos da glicemia (136 ± 8 mg/dL), lactato (3,58 ± 0,94 mmol/L), leucocitose (23.570
± 4.991 células/mm3) e alterações clínicas (escore 11±1),
caracterizadas por comprometimento do estado de alerta e mobilidade, e presença de piloereção, diarréia, olhos encrustados, nariz e cauda sujos. REL normalizou o número de leucócitos em rolamento, aderidos e migrados no mesentério, a glicemia, o lactato e o número de leucócitos circulantes. REL também melhorou o escore clínico (7 ± 1).
CONCLUSÃO: A CLP induziu inflamação local e sistêmica. A REL resolveu, por completo, o processo inflamatório.
UNITERMOS:Microcirculação. Mesentério. Microscopia intravital. Peritonite. Sepse.
REFERENCES
1. Ayala A, Song GY, Chung CS, Redmond KM, Chaudry IH. Immune depression in polymicrobial sepsis: the role of necrotic (injured) tissue and endotoxin. Crit Care Med. 2000;28:2949-55.
2. Rocha-e-Silva M, Poli-de-Figueiredo LF. Small volume hypertonic resuscitation of circulatory shock. Clinics. 2005;60:159-72. 3. Chaudry IH. Sepsis: lessons learned in the last century and future
directions. Arch Surg. 1999;134:922-9.
4. Vincent JL: International Sepsis Forum. Hemodynamic support in septic shock. Intens Care Med. 2001;27:S80-S92.
5. Marshall JC, Maier RV, Jimenez M, Dellinger EP. Source control in the management of severe sepsis and septic shock: an evidence-based review. Crit Care Med. 2004;32:S513-526.
6. Wang P, Ba ZF, Zhou M, Tait SM, Chaudry IH. Pentoxifylline restores cardiac output and tissue perfusion after trauma-hemorrhage and decreases susceptibility to sepsis. Surgery. 1993;114:352-9. 7. Coimbra R, Hoyt DB, Junger WG, Angle N, Wolf P, Loomis W, et al.
Hypertonic saline resuscitation decreases susceptibility to sepsis after hemorrhagic shock. J Trauma. 1997;42:602-7.
8. Lagoa C, Poli de Figueiredo LF, Cruz Jr RJ, Silva E, Rocha e Silva M. Effects of volume resuscitation on splanchnic perfusion in canine model of severe sepsis induced by live E. coli infusion. Critical Care.
2004;8:221-8.
9. Rahal L, Garrido AG, Cruz RJ Jr, Rocha E Silva M, Poli-de-Figueiredo LF. Systemic and regional hemodynamic effects of enalaprilat infusion in experimental normotensive sepsis. Braz J Med Biol Res. 2006;39:1205-15. 10. Panes J, Granger D. Leukocyte-endothelial cell interactions: molecular mechanisms and implications in gastrointestinal disease. Gastroenterol. 1998;114:1166-72.
11. Granger DN, Kubes P. The microcirculation and inflammation: modulation of leukocyte-endothelial cell adhesion. J Leukoc Biol. 1994;55:662-75.
12. Kishimoto TK, Rothlein R. Integrins, ICAMs, and selectins: role and regulation of adhesion molecules in neutrophil recruitment to inflammatory sites. Adv Pharmacol. 1994;25:117-169.
13. Nakagawa NK, Nogueira RA, Correia CJ, Shiwa SR, Cruz JW, Poli de Figueiredo LF, et al. Leukocyte-endothelium interactions after hemorrhagic shock/reperfusion and cecal ligation/puncture: an intravital microscopic study in rat mesentery. Shock. 2006; 26:180-6. 14. Kubes P, Heit B, Van Marle G, Johnston JB, Knight D, Khan A, et al. In
vivo impairment of neutrophil recruitment during lentivirus infection. J Immunol. 2003;171:4801-8.
15. Yada-Langui MM, Anjos-Valotta EA, Sannomiya P, Rocha e Silva M, Coimbra R. Resuscitation affects microcirculatory polymorphonuclear leukocyte behavior after hemorrhagic shock: role of hypertonic saline and pentoxifylline. Exp Biol Med. 2004;229:684-93.
16. Marshall JC. Neutrophils in the pathogenesis of sepsis. Crit Care Med. 2005;33:S502-505.
17. Childs EW, Wood JG, Smalley DM, Hunter FA, Cheung LY. Leukocyte adherence and sequestration following hemorrhagic shock and total ischemia in rats. Shock. 1999;11:428-36.
18. Wichterman KA, Baue AE, Chaudry IH. Sepsis and septic shock: a review of laboratory models and a proposal. J Surg Res. 1980;29:189-201. 19. Madorin WS, Martin CM, Sibbald WJ. Dopexamine attenuates flow
motion in ileal mucosal arterioles in normotensive sepsis. Crit Care Med. 1999;27:394-400.
20. Piper RD, Pitt-Hyde ML, Anderson LA, Sibbald WJ, Potter RF. Leukocyte activation and flow behavior in rat skeletal muscle in sepsis. Am J Respir Crit Care Med. 1998;157:129-34.
21. Ferri LE, Pascual J, Seely AJE, Giannias B, Christou NV. Intra-abdominal sepsis attenuates local inflammation-mediated increases in microvascular permeability at remote sites in mice in vivo. Surgery. 2004;135:187-95. 22. Lam C, Tyml K, Martin C, Sibbald W. Microvascular perfusion is impaired in a rat model of normotensive sepsis. J Clin Invest. 1994;94:2077-83.
23. Swartz DE, Seely AJE, Ferri L, Giannias B, Christou NV. Decreased systemic polymorphonuclear neutrophil (PMN) rolling without increased PMN adhesion in peritonitis at remote sites. Arch Surg. 2000;135:959-66.