www.bjorl.org
Brazilian
Journal
of
OTORHINOLARYNGOLOGY
ORIGINAL
ARTICLE
Evaluation
of
vestibular
evoked
myogenic
potentials
(VEMP)
and
electrocochleography
for
the
diagnosis
of
Ménière’s
disease
夽
Pauliana
Lamounier
a,b,
Thiago
Silva
Almeida
de
Souza
c,
Debora
Aparecida
Gobbo
b,
Fayez
Bahmad
Jr.
a,d,∗aUniversidadedeBrasília(UNB),CiênciasdaSaúde,Brasília,DF,Brazil
bCentrodeReabilitac¸ãoeReadaptac¸ãoDr.HenriqueSantillo(CRER-GO),Goiânia,GO,Brazil
cPontifíciaUniversidadeCatólicadeGoiás(PUC-GO),Goiânia,GO,Brazil
dInstitutoBrasiliensedeOtorrinolaringologia,Brasília,DF,Brazil
Received19October2015;accepted14April2016 Availableonline2June2016
KEYWORDS
Ménière’sdisease; Electrocochleography; Vestibularevoked myogenicpotential
Abstract
Introduction:Ménière’sdisease(MD)isaninnereardisordercharacterizedbyepisodicvertigo, tinnitus,earfullness,andfluctuatinghearing.Itsdiagnosiscanbeespeciallydifficultincases wherevestibular symptomsarepresentinisolation(vestibularMD).The definitivediagnosis ismadehistologicallyandcanonlybeperformedpost-mortem,afteranalysisofthetemporal bone.Endolymphatichydropsisahistopathologicalfindingofthediseaseandoccursmoreoften inthecochleaandsaccule,followedbytheutricleandsemicircularcanals.Vestibularevoked myogenicpotentials(VEMP)emergedasthemethodofassessmentofvestibularfunctionin1994. Untilthen,therewasnouniquewayofassessingsaccularfunctionandtheinferiorvestibular nerve.Giventhatthesacculeisresponsibleformostcasesofseverehydrops,VEMPappearsas anewtooltoassistinthediagnosisofMD.
Objective:To evaluate the sensitivity and specificity of VEMP and electrocochleography (EcochG)inthediagnosisofdefiniteMDcomparedwithclinicaldiagnosis.
Methods:Thestudyincludes12patients(24ears)diagnosedwithdefiniteMDdefinedaccording totheclinicalcriteriaproposedbytheAmericanAcademyofOtolaryngology---HeadandNeck Surgery(AAO-HNS)in1995,aswellas12healthyvolunteersallocatedtothecontrolgroup(24 ears).A clinicaldiagnosis bytheAAO-HNScriteria wasconsideredasthegold standard.All
夽 Pleasecitethisarticleas:LamounierP,deSouzaTS,GobboDA,BahmadJr.F.Evaluationofvestibularevokedmyogenicpotentials(VEMP) andelectrocochleographyforthediagnosisofMénière’sdisease.BrazJOtorhinolaryngol.2017;83:394---403.
∗Correspondingauthor.
E-mail:[email protected](F.BahmadJr.).
PeerReviewundertheresponsibilityofAssociac¸aoBrasileiradeOtorrinolaringologiaeCirurgiaCervico-Facial.
http://dx.doi.org/10.1016/j.bjorl.2016.04.021
patientsunderwentanotoneurologicalexamination,includingpuretoneandspeech audiome-try,VEMP,andextratympanicEcochG.Thesensitivityandspecificitytodetectthepresenceor absenceofdiseasewerecalculated,aswellastheir95%confidenceintervals.Thereliabilityof VEMPandEcochGinbothearswasassessedusingthekappaindex.
Results:In bothtestsandinbothears,theabilitytodiagnosehealthycaseswashigh,with specificityrangingfrom84.6%to100%.Moreover,theabilityoftheteststodiagnosethedisease variedfromlowtomoderatesensitivity,withvaluesrangingfrom37.5%to63.6%.Theagreement ofbothtestsintherightear,measuredbythekappacoefficient,wasequalto0.54(95%CI: 0.20---0.89),indicatingamoderateagreement.Intheleftear,thatagreementwasequalto0.07 (95%CI:−0.33to0.46),indicatingaweakcorrelationbetweenthetests.Thesensitivityofthe VEMPfortherightearwas63.6%andfortheleftear,62.5%.ThesensitivityofEcochGforthe rightearwas63.6%and37.5%fortheleftear.
Conclusion: Thespecificityofbothtestswashigh,andthesensitivityofVEMPwashigherthan thatofEcochG.
© 2017 Associac¸˜ao Brasileira de Otorrinolaringologia e Cirurgia C´ervico-Facial. Published by Elsevier Editora Ltda. This is an open access article under the CC BY license (http:// creativecommons.org/licenses/by/4.0/).
PALAVRAS-CHAVE
Doenc¸adeMénière; Eletrococleografia; Potencialevocado miogênicovestibular
Avaliac¸ãodospotenciaisevocadosmiogênicosvestibulares(VEMP)e eletrococleografianodiagnósticodadoenc¸adeMénière
Resumo
Introduc¸ão: A Doenc¸a de Ménière (DM) é uma doenc¸a daorelha interna caracterizada por vertigemepisódica,zumbido,plenitudeaural,eaudic¸ãoflutuante.Seudiagnósticopodeser especialmentedifícilnoscasosemqueossintomasvestibularesestãopresentesisoladamente (DMvestibular).Odiagnósticodecertezaéhistológicoesomentepodeserrealizadonopost mortem,apósanálisedoossotemporal.Ahidropisiaendolinfáticaéumachadohistopatológico dadoenc¸aeocorremaisfrequentementenacócleaesáculo,seguidospeloutrículoecanais semicirculares.Ospotenciaisevocadosmiogênicosvestibulares(VEMP)surgiramcomométodo deavaliac¸ãodafunc¸ãovestibulardesde1994.Atéentãonãohaviaumamaneiraexclusivade avaliac¸ãodafunc¸ãosacular edonervovestibular inferioresendoosáculoresponsávelpor grandepartedoscasosdehidropisiasevera,oVEMPaparececomoumanovaferramentapara auxiliarnodiagnósticodaDM.
Objetivo: AvaliarasensibilidadeeespecificidadedoVEMPedaEletrococleografia(ECochG)no diagnósticodaDMemcomparac¸ãocomodiagnósticoclínico.
Método: :Foramselecionados12 pacientes(24 orelhas)comdiagnósticodeDMdefinidade acordocomoscritériosclínicospropostospelaAmericanAcademyofOtolaryngology-Headand NeckSurgery1995(AAO-HNS)e12voluntáriossaudáveisalocadosnogrupocontrole(24orelhas). Considerou-seodiagnósticoclínicopelaAAO-HNScomopadrãoouro.Todosospacientesforam submetidos aexame otoneurológico,incluindoaudiometriatonal evocal, VEMP e eletroco-cleografiaextratimpânica.Asensibilidadeeespecificidadeparadetectarapresenc¸aouausência dedoenc¸aforamcalculadaseosrespectivosintervalosdeconfianc¸ade95%obtidos.A confia-bilidadedostestesdediagnósticoVEMPeeletrococleografiaemambasasorelhasfoiavaliada peloíndicekappa.
Resultados: Em ambosos testes eem ambasas orelhas,acapacidade para diagnosticaros casos saudáveis éalta,aespecificidade variandode84,6%---100%. Alémdisso, acapacidade dostestesparaodiagnósticodadoenc¸avariadebaixaamoderadasensibilidade,comvalores de37,5%---63,6%.Aconcordânciadosdoistestesnaorelhadireita,medidapelocoeficientede kappafoiiguala0,54;com95%IC(0,20---0,89),indicandoumaconcordânciamoderada.Para aorelhaesquerdaessaconcordânciafoiiguala0,07com95%IC(-0,33---0,46),indicandouma concordânciafracaentreostestes.AsensibilidadedoVEMPparaaorelhadireitafoide63,6%e paraaorelhaesquerda,de62,5%.AsensibilidadedaECochGparaaorelhadireitafoide63,6% e37,5%paraaorelhaesquerda.
Conclusão:AespecificidadedeambosostestesfoialtaeasensibilidadedoVEMPfoimaiorque adaeletrococleografia.
Introduction
Ménière’s disease (MD) is an inner ear disorder
charac-terized by episodic vertigo, tinnitus, ear fullness, and
fluctuatinghearing.Thedefinitivediagnosisismade
histo-logically, and can only be performed post-mortem, after
analysis of the temporal bone. In 1995, the American
AcademyofOtolaryngology---HeadandNeckSurgery
(AAO-HNS)1 developed diagnosticcriteria thatare widely used.
Recently,theBáránySocietydevelopedanewguidelinefor
the diagnosis of MD; after an evolutionary understanding
of MD and vestibular migraine, the most common
differ-ential diagnosis, the need to update these criteria was
highlighted.2
For a long time, it was believed that endolymphatic
hydrops would be the histopathological substrate of the
disease;thisoccursmoreofteninthecochleaandsaccule,
followedbytheutricleandsemicircularchannels.3,4Recent
studies have indicated that hydrops is a finding of the
MD,together withthe symptoms, sinceit alone does not
explainalltheclinicalfeatures,includingtheprogressionof
hearinglossandthefrequencyofvertigoattacks.According
to the criteria of the AAO-HNS, individuals with two or
more spontaneous episodes of vertigo, lasting ≥20min,
with documented hearing loss in at least one occasion
and tinnitus or ear fullness are clinically classified as
havingdefiniteMD.The diseaseiscalled probablewhena
definedepisodeofvertigo inthepresenceof documented
sensorineural hearing loss in at least one occasion, ear
fullness, or tinnitus. MD is also classified as possible in
the presence of Ménière-type episodic vertigo without
documented hearing loss or when there is sensorineural
hearingloss,fixed or floating,associatedwithimbalance,
withoutadefinedepisodeofdizziness.1
According to the criteria of the Bárány Society, MD
is classified as definite or probable. In definite MD, the
patientshouldhavehadtwoormorespontaneousepisodes
of vertigo, each lasting from 20min to 12h; documented
mild to moderate sensorineural hearing loss; aural
symp-toms(hearing, tinnitus,andfullness) inthe affected ear;
and exclusion of other vestibular disorders that explain
the symptoms. In probable MD, the patient should have
had two or more episodes of vertigo or loss of
bal-ance, each lasting from 20min to 24h; floating aural
symptoms(hearing, tinnitus, or fullness) in that ear;and
exclusion of other vestibular disorders that explain the
symptoms.2
The cause of hydrops is still unknown, and most
theoriesarebasedonthechange inproduction or
resorp-tion of endolymph. In 1982, Schuknecht5 postulated that
hydrops causes rupture of Reissner’s membrane, allowing
theendolymphaticfluid,richin potassium,tocontactthe
perilymph andreach the surfaceof the ciliated cells and
thevestibulocochlearnerve,causinghearinglossandvertigo
attacks.Somebelievethateventhedistensionofthebasilar
membranebytheendolymphatichydropsmayalreadylead
todegenerationof ciliated cells andtherefore their
mal-function,causing adecreasein theaction potential(AP).6
Anatomicalandvascularabnormalitiesarepossiblyrelated
toitsetiopathogenesis.
In 1989, Rauch et al. found histological evidence of
endolymphatichydropsin13outof13casesofpatientswith
MD,butareviewofmedicalrecordsofsixoutof19
tempo-ralboneswithendolymphatichydropsrevealednosignsor
symptomsofMD.Theyobservedthatmanyinnerearshave
endolymphatichydropswithoutclinicalsyndrome
manifes-tation. Some suggest that endolymphatic hydropsmay be
anepiphenomenonofthepathophysiologicalmechanismof
MD.7
The hypothesis of genetic predisposition is widely
accepted,sinceapositivefamilyhistoryispresentinmany
patients with MD. Research shows that the disease could
result from mutations in the short arm of chromosome
6, where the histocompatibility antigen (HLA) is located;
such mutations would be synergistic to the development
ofMD.Approximately7%ofpatientswithfamilialMDhave
an autosomaldominantmodel with60% penetrance anda
geneticpatternofanticipation, inwhichthenext
genera-tionwiththedisease willpresentmore intensesymptoms
with earlier onset.6,8 Genetic studies in families suggest
that the genetic mechanism associated with MD is
com-plexandthatthereismorethanonegeneinvolvedinmost
cases.6,8,9
Asotherinnereardisorders,MDhasalsobeenregarded
as an autoimmune disease, as evidenced by a
rela-tionship with circulating immune complexes, suggesting
deposition in the endolymphatic sac.6,9 The autoimmune
theory has also been strengthened by recent evidence
of anti-endolymphatic sac auto-antibodies in the serum
of patients with MD. Inhalant and food allergies have
been associated with MD and, in many cases, allergic
patientsimprovedtheirsymptomsafterspecificantiallergic
therapy.6,9,10
Electrocochleography(EcochG) hasbeen usedforyears
in thediagnosis ofendolymphatic hydropsinthe cochlea.
Itsclinicalapplication,however,isstillcontroversialamong
otorhinolaryngologistsbecauseofitsvariablesensitivity,as
incasesofhearinglossduetodiseaseprogression,patients
mayexperienceareductionintheamplitudeofAPdueto
lossofauditorynervefibers.11,12
Vestibularevokedmyogenicpotentials(VEMP)emerged
as a method to assess vestibular function in 1994, when
Colebatch andHalmagyi reportedsurface potentialin the
sternocleidomastoid (SCM) muscle in response to clicks
through high-intensity air conduction (100dB),
access-ing the sacculo-collic reflex.13,14 These potentials assess
the saccular and inferior vestibular nerve function, and
are absent or decreased by 30---54% in patients with
MD. The exam is easy to perform, does not cause
dis-comfort to patient, and does not vary with hearing
loss.13---15 VEMP can be obtained by air and bone
conduc-tionandgalvanicstimulation,usingtoneburstorclicksas
stimulus.14---16
ThediagnosisofMDcanbedifficult,especiallyincases
where vestibularsymptomsarepresent inisolation
(vesti-bular MD).17 Until 1994, there was no unique method to
assess saccular and inferior vestibularnerve function.17,18
Asthesacculeisthesecondmostprevalentaffectedsiteof
endolymphatichydrops,representingmostformsofsevere
hydrops,VEMPappearsasanauxiliarytoolinthediagnosis
ofMD.18,19
Thisstudyaimedtoassessthesensitivityandspecificity
ofVEMPandEcochGinthediagnosis ofMD,aswell asthe
Methods
This was a clinical, prospective trial, which selected 12
patients (24 ears) diagnosed with definite MD according
tothe clinical criteriaproposed by theAAO-HNS in1995,
of whom seven were females and five were male, aged
between 33 and63 yearswitha mean of48.41 years; 12
healthyindividuals,sex-andage-matched,wereallocated
tothecontrolgroup(24ears).Theexclusioncriteriawere
impossibilityofcervicalrotationandmiddleorexternalear
disease. The two researchers who conducted the
assess-mentswereunawareofthegrouptowhichthepatienthad
beenallocated.
The study was approved by the Research Ethics
Committee under opinion No. 10668613.2.0000.0030. All
participantssignedaninformedconsentforinclusioninthe
study.
ClinicaldiagnosisbytheAAO-HNS1995criteriawas
con-sidered as the gold standard, and all patients underwent
otorhinolaryngological and otoneurological examinations,
including pure tone and speech audiometry, VEMP, and
extratympanicEcochG.
Clinical history was taken and a physical examination
wasconducted;thedifferentialdiagnosiswasexcludedand
theclinicaldiagnosisofMDaccordingtotheAAO-HNS1995
criteriawasconfirmed.
Tonalandspeechaudiometry/impedance
The examination was performed in all patients, with the
followinggoals:
• DiagnosehearinglosswichispartofthedefiniteMD
diag-nosticcriteriabytheAAO-HNS;
• Discardcaseswithmiddleeardiseases;and
• Discard cases with conductive loss that alters VEMP
parameters.
VEMP
TherecordingdevicewastheVivoSonicIntegrity,with
pro-grammingforevokedpotentials,usingaspecificprotocolfor
performingtheVEMP.Myogenicpotentialsarepickedupby
electrodesplaced onthe patient’sSCMmuscle(ipsilateral
tothesoundstimulus),thereferenceelectrode(negative)
isplacedontopoftheSCM;theactiveelectrode(positive),
onthesternum;andtheground,ontheforehead.Patients
wereinstructedtoturntheirheadintheoppositedirection
tothesoundstimulus,sothattherewasacontractionofthe
SCMmuscle(Fig.1).
Stimulusandresponseacquisitionparameters
150toneburststimuliwereused,atafrequencyof500Hz,
withtherateof7.1stimuli/s,stimuliintensityof95dBHL,
highpassfiltersof30Hzandlow-passfiltersof1000Hz,
pre-sentedthroughER-A3earphones.Therecordingsweremade
ina30-mswindow.
Thefollowinganalysiscriteriawereconsidered:presence
or absenceofreproduciblewaves and interauralresponse
asymmetryindexfortheamplitude.
Figure1 VEMPperformedontheright.
The presence of reproducible waves and interaural
responseasymmetryindexfortheamplitudeequaltoorless
than34%characterizednormalVEMP.
Inturn,theabsenceofreproduciblewavesand/or
inter-auralresponseasymmetryindexfortheamplitudegreater
than34%characterizedalteredVEMP.
EcochG
TherecordingdevicewastheVivoSonicIntegrityand
TIP-trode electrodes were inserted in the external auditory
meatus. The ear canal was cleaned with abrasive paste.
Negativeandpositiveelectrodeswereconnectedtothe
Tip-Trodeearphone; thenegativeelectrode inthe stimulated
ear,the positive in the contralateral ear,and the ground
electrodeontheforehead(Fig.2).
Stimulusandresponseacquisitionparameters
Click stimuli were utilized (2---4kHz), at a rate of
11.3stimulus/s; intensity of 99dB HL, high-pass filters of
30Hz,andlow-passfiltersof2400Hz.Therecordingswere
madewitha5-mswindow.
AnSP/PAratiogreaterthan50%wasconsideredaltered.
Statisticalanalysis
Inordertovalidatebothdiagnostictests(VEMPandEcochG),
theclinicaldiagnosiswasconsideredasthegoldstandard.
Bothears were classified as withor without the disease,
and as test instrument, the positive or negative result
Figure2 EcochGperformedintherightear.
the presence or absence of the disease were calculated,
aswell as their respective 95% confidence intervals. The
reliability of diagnostic tests in both ears was assessed
by kappa, using the scale proposed by Landis and Kock,
which classifies the agreement as: (≤0, poor; 0.10---0.19,
weak;0.20---0.39,regular;0.40---0.59,moderate;0.60---0.79,
substantial;0.80---0.99,almostperfect;1,perfect).The
pro-portionsof positiveand negativeresultsof thediagnostic
testswerecomparedusingMcNemar’stest.Fordata
analy-sis,SAS9.3wasused.
Thesignificancelevelwassetatp<0.05.
Results
At theVEMP, themean p13latency for thecontrol group
was 15.93ms, with standard deviation (SD) of 0.85ms.
Themean n23 latencyfor the controlgroupwas22.80ms
(SD=1.16ms). The meanasymmetry indexfor thecontrol
group was 16.22 (SD=15.58). At the EcochG, the mean
SP/APratioforthecontrolgroupwas24.39%(SD=11.61).All
Specificity measures
Right Left
VEMP
ECochG
Ear 100
90 80 70 60 50 40 30 20 10 0
%
Figure3 VEMP andECochGspecificityinthe rightandleft ears.
Sensitivity measures
VEMP
ECochG 100
90 80 70 60 50
%
40 30 20 10 0
Right Left
Ear
Figure4 VEMP andECochGsensitivityinthe rightandleft ears.
controlgrouppatientshadpuretoneandspeechaudiometry
andimpedancewithinnormallimits.
Inthestudygroup,sixcasesofbilateralMD,fivecases
ofunilateralMDintherightear,andonecaseofMDinthe
left earwere identified, totaling 11 right ears and seven
left ears. Regarding VEMP, 14 ears presented absence of
wavesandtenearsshowedthepresenceofbiphasic
wave-form;bothearsshowedthepresenceofwavesinonlythree
patients,thusenablingcalculationoftheasymmetryindex.
The mean asymmetryindex for thesepatients was11.22.
ThemeanSP/APforthecasegroupwas47.23.
Tables1 and2show theindividualresults ofthe study
groupontherightandleftears,respectively.Tables3and4
showtheindividualresultsofthecontrolgroupontheright
andleftears.
Inbothtestsandbothears,theability todiagnose the
healthycaseswashigh:thespecificityrangedfrom84.6%to
100%(Fig.3).Moreover,theabilityofthetesttodiagnose
thediseasevariedfromlowtomoderate,whicha
sensitiv-ityof37.5---63.6%(Fig.4).Table5showsthesensitivityand
specificityofbothtestsinbothears.
The agreement of both exams in the right ear,
mea-sured by the kappa coefficient, was equal to 0.54 (95%
CI: 0.20---0.89), indicating a moderate agreement. In the
leftear,thatagreementwasequalto0.07(95% CI:−0.33
to0.46), indicating a weakcorrelation betweenthe tests
(Table6).
Fortherightear,theproportionofpositiveandnegative
test VEMP results (37.5---62.5%) did notdiffer significantly
from the proportion of positive and negative results in
EcochG (33.3---66.7%; p=0.6547; Fig. 5). Forthe left ear,
Table1 Individualresultsoftherightearinthecasegroup.
Casegrouprightear Impedance
Patient ClinicalDT VEMP EcochG Audiometry
1 Yes Normal Normal ModerateSNHL,ascending Acurve
2 Yes Altered Altered MildSNHL,flat Acurve
3 No Altered Normal Normal Acurve
4 Yes Altered Altered ModerateSNHL,flat Acurve
5 Yes Altered Altered Mildconductivehearingloss Acurve
6 Yes Normal Altered ModerateSNHL,descending Acurve
7 Yes Normal Altered MildSNHL,flat Acurve
8 Yes Normal Normal MildSNHL,flat Acurve
9 Yes Altered Normal ModerateSNHL,descending Acurve
10 Yes Altered Altered ModerateSNHL,descending Acurve
11 Yes Altered Altered MildSNHL,invertedU Acurve
12 Yes Altered Normal MildSNHL,descending Acurve
Table2 Individualresultsoftheleftearinthecasegroup.
Casegroupleftear Impedance
Patient ClinicalDT VEMP EcochG Audiometry
1 Yes Normal Altered MildSNHL,flat Acurve
2 No Normal Normal Normal Acurve
3 Yes Altered Normal MildSNHL,flat Acurve
4 Yes Normal Altered ModerateSNHL,flat Acurve
5 No Altered Normal Normal Acurve
6 No Normal Normal Normal Acurve
7 No Normal Normal Normal Acurve
8 Yes Altered Normal ModerateSNHL,ascending Acurve
9 Yes Altered Normal ModerateSNHL,descending Acurve
10 Yes Altered Normal ModerateSNHL,descending Acurve
11 Yes Altered Altered ModerateSNHL,flat Acurve
12 Yes Normal Normal MildSNHL,descending Acurve
Table3 Individualresultsoftherightearinthecontrolgroup.
Controlgrouprightear Impedance
Patient ClinicalDT VEMP EcochG Audiometry
13 No Normal Normal Normal Acurve
14 No Normal Normal Normal Acurve
15 No Normal Normal Normal Acurve
16 No Normal Normal Normal Acurve
17 No Normal Normal Normal Acurve
18 No Normal Normal Normal Acurve
19 No Normal Normal Normal Acurve
20 No Normal Normal Normal Acurve
21 No Normal Normal Normal Acurve
22 No Altered Altered Normal Acurve
23 No Normal Normal Normal Acurve
Table4 Individualresultsoftheleftearinthecontrolgroup.
Controlgroupleftear Impedance
Patient ClinicalDT VEMP EcochG Audiometry
13 No Normal Normal Normal Acurve
14 No Normal Normal Normal Acurve
15 No Normal Normal Normal Acurve
16 No Normal Normal Normal Acurve
17 No Normal Normal Normal Acurve
18 No Normal Normal Normal Acurve
19 No Normal Normal Normal Acurve
20 No Normal Normal Normal Acurve
21 No Normal Normal Normal Acurve
22 No Normal Normal Normal Acurve
23 No Normal Normal Normal Acurve
24 No Normal Normal Normal Acurve
Table5 Sensitivityandspecificityvalues,bytypeoftestinbothears.
Ear Diagnostictest Clinicalexamination
Diseased Healthy
n %(95%CI) n %(95%CI)
Right
VEMP
Positive 7 63.6(30.8---89.1) 2
---Negative 4 --- 11 84.6(54.6---98.1)
EcochG
Positive 7 63.6(30.8---89.1) 1
---Negative 4 --- 12 92.3(64.0---99.8)
Left
VEMP
Positive 5 62.5(24.5---91.5) 1
---Negative 3 --- 15 93.7(69.8---99.8)
EcochG
Positive 3 37.5(8.5---75.5) 0
---Negative 5 --- 16 100.0
Table6 Agreementbetweendiagnostictests.
Ear VEMP EcochG Kappa(95%CI)
Positive Negative Total
Right
0.54(0.20to0.89)
Positive 6 3 9(37.5)
Negative 2 13 15(62.5)
Total 8(33.3) 16(66.7) 24(100.0)
Left
0.07(−0.33to0.46)
Positive 1 5 6(25.0)
Negative 2 16 18(75.0)
Percentage of positive and negative results by exam in the right ear
100 90 80 % 70 60 50 40 30 20 10 0 Positive Negative Result VEMP ECochG
Figure5 Percentageofpositiveandnegativeresultsbyexam intherightear.
Percentage of positive and negative results by exam in the left ear
% 100 90 80 70 60 50 40 30 20 10 0 Positive Result VEMP ECochG Negative
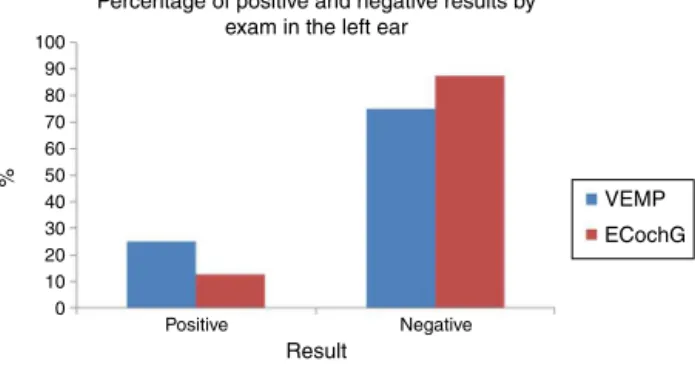
Figure6 Percentageofpositiveandnegativeresultsbyexam intheleftear.
(25.0---75.0%) did notdiffer significantly in the proportion of positive and negative results in EcochG (12.5---87.5%; p=0.2568;Fig.6).
Discussion
12patientswithdefiniteMDandcontrolswereevaluatedby
audiometryandimpedance,VEMP,andEcochG.
In the present study,VEMP wasperformed in a sitting
position,whichaccordingtopreviousstudiesistheposition
thatprovidesbetteractivationoftheSCM.20---22 Toneburst
stimuliwereused,sincestimuliwithfrequenciesnear500Hz
have higher responseamplitudes.23 The large variationin
responseamplitudecausedbydifferentdegreesofmuscle
contractureobtainedforeachindividualjustifiesthe
analy-sisoftheVEMPresponsesthroughtheinterauralasymmetry
index.Basedonareviewoftheliterature,valuesabove34%
wereconsideredasaltered.22,24However,abiphasicp13-n23
wavewasobservedintenears(41.66%),whereasonlythree
patientsshowedthepresenceofwavesinbothears,
allow-ingforthecalculationoftheasymmetryindex.In1999,Seo
etal.18 observedbiphasicwaves in72%ofcasesofMDand
Waele,17in45.7%ofcases.
14ears(58.33%)showednowavesat VEMP.Thelackof
response,aswellasasymmetryindexgreaterthan34%,
sug-gest endolymphatic hydrops.17,18,24 Jariengprasert et al.25
observedthat theabsenceofwaves andasymmetryindex
alterationsweremoresignificantthanP1andN1latencies
oramplitudemeasuresintheidentificationofsaccular
dys-functioninMD.
Dependingontheseverityofhydrops,somepatientsmay
presentirreversibledegenerationofthesensoryepithelium
of the saccular macula, with absence of waves. In 1999,
Waele17alsoattributedthelackofVEMPresponseinpatients
withMDtoinsufficientcontractionduringtheexamination,
unrecognizedvestibularpathology,orsaccule
hyposensitiv-ityduetotheagingofthesaccularmacula intheelderly.
Thisineffectivecontractioncan beavoidedbyusing
elec-tromyographytomonitorthedegreeofmusclecontraction,
whichwasnotpossibleinthepresentstudy,sincethe
equip-mentuseddidnothavethisfeature.
TheEcochGusedinthisstudywasextratympanic,which,
according to the literature review, is an effective and
non-invasivemethodofmeasuringcochlearhydrops.26 The
presenceofSPandAPwasobservedinallpatients.
The responsesshoweda lowtomoderate sensitivityof
VEMPandEcochGinthediagnosisofMDinrelationto
clin-icaldiagnosis.ThesensitivityoftheVEMPfortherightear
was63.6%andfortheleftear,62.5%.Itwasslightlyhigher
thanthat found in theliterature, which ranged from40%
to54%.16,17,27ThesensitivityofEcochGfortherightearwas
63.6%andfortheleftear,37.5%;theliteraturevaluesrange
from57%to71%.26
The fluctuating course of the disease complicates the
interpretationofelectrophysiologicaltests.Themain
ques-tion regarding the use of diagnostic tests in MD pertains
totheir sensitivity.Egamietal.14 observed that,although
the VEMP sensitivity was not high, it wascomparable to
thecalorictest,providingadditionalinformationtoidentify
vestibularabnormalitiesinMD.
Thehighspecificityofbothtestswasconsistentwith
find-ingsintheliterature,suggestingtheirhighaccuracyinruling
out the presence of disease, assisting especially in cases
wherethereisdifferentialdiagnosis.14,26
RegardingtheVEMP,twoasymptomaticearshadchanged
VEMP.Thisfindingis describedintheliteratureasaresult
ofoccultsaccularhydropsorofbinauralinteractionsinthe
otolith-cervical reflex arc of VEMP. Similar findings were
retrieved in the literature.15---18 In 2006, Lin et al. found
that27%ofpatientswithunilateralMDshowedhigh
thresh-olds in the asymptomatic ear, demonstrating that VEMP
canbealtered evenbeforetheappearance of theclassic
symptoms of the disease, and that saccular hydrops may
precedethesymptomsofbilateralMD.19,28,29In2000,
Con-lonandGibson30alsoreportedabnormalclinicalfindingsin
asymptomatic ears. There are also individual differences
in the degree of muscle tone and contraction,28,31 which
theauthorsofthepresentstudytriedtoavoidbysex-and
age-matchingthecontrolgroupandbyusingtheinteraural
asymmetryindexasaparameter.
The abilityto predictthe presence ofabnormalities in
anasymptomaticearis oneofthegreatfeaturesofVEMP.
Knowingwhether the patient has the disease in the
con-tralateralear,forexample,aidsinmakingadecisionabout
ablativeproceduresinthediseasedear.28
Seven ears with MD had normal VEMP. These ears
may have saccular macula free from hydrops, presenting
cochlearhydrops.In1987,OkunoandSando3examined26
temporalbonesofpatientswithMDanddemonstratedthat
endolymphatichydropswasobservedmostfrequentlyinthe
cochlea,followedbythesaccule,utricle,andsemicircular
ofseverehydropsrefractorytomedicaltreatmentmaybe
locatedinthesaccule.16
RegardingEcochG,noasymptomaticearhadaltered
val-ues, and nine ears with MD had normal values. Of these
ears,fivehadmoderatesensorineuralhearinglossandfour
had mild sensorineural hearing loss. Audiometric
thresh-olds around 50dB undermine the analysis of hydrops by
EcochG,32 and are therefore a hypothesis for the normal
EcochG observed in five patients with moderate hearing
loss.Theliteraturealsoreportsthatpatientswithhydrops
inotolith organs may have disease-freecochlea, which is
anotherhypothesis thatcouldapplytopatients withmild
hearingloss.AmajoradvantageofVEMPisthatthe
saccu-larmaculaissensitivetosoundevenaftertotaldestruction
ofthecochlea,andthusitcanbeperformedinindividuals
withgoodorbadhearingacuity.14,17,19
Thesamplesizewasonelimitationofthepresentstudy.
Definite MD, with document hearing loss, it is not highly
prevalent,hinderingtheselectionofpatientsforthestudy.
Forthesamereason,itwasnotpossibletocomparepatients
withthesame diseaseduration.Earswithlongerduration
ofsymptomsshowhigherabnormalitiesintheSP/APratio,
andoncetheSPrises,itpersistsforlongperiods26,33;thatis,
evenintheperiodbetweenattackstheEcochGcan
demon-stratecochlearhydrops.
CervicalVEMPbyairconductionmaybeincreasedinthe
earlystages ofMD,perhaps duetothe pressureof
saccu-larhydropsagainstthestapesfootplate,increasingsaccular
sensitivitytointensesound.16Itsmeasurementcanbe
vari-able, with a tendency to disappear with the progression
of thedisease, aswell as duringthe 24h post-crisis,and
mayreappearafter48horwiththeuseofdrugstoreduce
endolymphatichydrops.2,11,13,34
Theagreementmeasuredbythekappacoefficient
eval-uates the similar results between both exams. The low
correlationbetweenthediagnostictestswasexpected,as
theyevaluatedifferentstructures.Furthermore,theyhave
differentsensitivitiesaccordingtothestageofthedisease;
Vemp is more altered in the symptomatic period, unlike
Ecochg,thatevenintheperiodbetweenattackscan
demon-stratecochlearhydrops.Theagreementfortheleftearwas
low,andlowerthanthatfortherightear,probablybecause
thesizeoftheleftearsamplewassmallerthanthatofthe
rightear.McNemar’stestassessedtheproportionofpositive
andnegativeresultsofVEMPandEcochG,whichwerenot
statisticallydifferentfromeachother,bothintherightand
leftears.
In the control group, the subjects were sex- and
age-matched. However,further studies could pairthe control
group for hearing loss similar tothose of patients in the
casegroupinsteadofselectingcontrolswithnormal
hear-ing, since the hearing thresholds are determining in the
sensitivityofEcochG.
The possibility of assessing saccular hydrops througha
noninvasive,easytoperformmethod,suchastheVEMP,aids
theotorhinolaryngologistindiseasemanagement.In2001,
MurosfushireportedthattheprolongationoflatencyinVEMP
suggestsretrolabyrinthine injury,facilitatingtheexclusion
ofdifferentialdiagnoses.25
Severalfactors corroboratethe aid ofadditionaltests,
such asVEMP and EcochG, in the diagnosis of MD.
Possi-bleMDcasesinwhichthereisnodocumentedhearingloss,
the heterogeneity of thedisease, andthe involvement of
variouslabyrinthinestructuresoftencomplicatethe
diagno-sis.VEMPandEcochGarecomplementarytests,sincethey
assessdifferentlabyrinthinestructures,andshouldbe
incor-poratedintoroutineotoneurologicalexaminations,aidingin
theidentificationofhydropslocationandthepossibilityof
asymptomaticearinvolvement.
Theassessmentofcochlearfunctionwithaudiometryand
EcochG,of thesacculethroughcervicalVEMP, ofthe
utri-cle throughocular VEMP, ofthe lateralsemicircular canal
through caloric tests, and of all the semicircular canals
throughvHITdemonstratestheadvancementofresearchin
thevestibulardiagnosis.Newpathsareopentothe
discov-eryofthepathophysiologicalmechanism ofadiseasethat
wasfirstdescribedoveracenturyagoandstillhasnodefined
treatmentprotocol.
Conclusion
Thespecificityofbothtestswashigh,andthesensitivityof
VEMPwashigherthanthatofEcochG.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
References
1.CommitteeonHearingandEquilibriumguidelinesforthe diag-nosisandevaluationoftherapyinMeniere’sdisease.American Academy ofOtolaryngology Head and Neck Foundation, Inc. OtolaryngolHeadNeckSurg.1995;113:181---5.
2.Escamez JAL, Carey J, Chung WH, Goebel JA, Magnusson M, Mandala M, et al. Diagnostic criteria for Menière’s dis-ease.Consensusdocument oftheBárány Society,TheJapan Societyfor Equilibrium Research, theEuropean Academy of Otology and Neurotology (EAONO), the American Academy ofOtolaryngology-HeadandNeckSurgery(AAO-HNS)andthe KoreanBalanceSociety.ActaOtorrinolaringolEsp.2016. 3.Okuno T, Sando I. Localization, frequency and severity of
endolymphatichydropsandthepathologyofthelabyrinthine membrane in Méniére’s disease. Ann Otol Rhinol Laryngol. 1987;96:438---45.
4.Taylor RL, Wijewardene AA, Gibson WP, Black DA, Halmagy GM,WelgampolaMS.Thevestibularevokedpotentialprofileof Ménière’sdisease.ClinNeurophisiol.2011;122:1256---63. 5.SchuknechtHF.Ménière’sdisease,pathogenesisandpathology.
AmJOtolaryngol.1982;3:349---52.
6.PiroddaA,BrandoliniC,ChiaraRaimondiM,GaetanoGF, Mod-ugnoGC,BorghiC.Meniere’sdisease:updateoftheoriesanda proposalofexplanation.ActaClinBelg.2010:65---73.
7.RauchSD,MerchantSN,ThendingerBA.Meniere’ssyndromeand endolymphatichydrops.Double-blindtemporalbonestudy.Ann OtolRhinolLaryngol.1989;98:873---83.
8.Khorsandi MT, Amoli MM, Borghei H, Emami H, Amiri P, AmirzargarA, etal. Associationsbetween HLA-C allelesand definite Meniere’s disease. Iran J Allergy Asthma Immunol. 2011;10:119---22. Available at: http://ijaai.tums.ac.ir/index. php/ijaai/article/view/337
10.Merchant SN, Adams JC, Nadol JB Jr. Pathophysiology of Ménière’ssyndrome: are symptoms causedbyendolimphatic hydrops?OtolNeurotol.2005;26:74---81.
11.MargolisRH,RieksD,FournierEM,LevineSE.Tympanic elec-trocochleography for diagnosis of Ménière’s disease. Arch OtolaryngolHeadNeckSurg.1995;121:44---5.
12.Nguyen LT, Harris JP, Nguyen QT. Clinicalutility of electro-cochleographyinthediagnosisandmanagementofMénière’s disease:AOSandANSmembershipsurveydata.OtolNeurotol. 2010;31:455---9.
13.RosengrenSM,WelgampolaMS,ColebatchJG.Vestibularevoked myogenic potentials: Past, present and future. Clin Neuro-physiol.2010;121:636---51.
14.Egami N, Ushio M, Yamasoba T, Yamaguchi T, Murofushi T, IwasakiS.Thediagnosticvalueofvestibularevokedmyogenic potentials in patients with Ménière’s disease. J Vestib Res. 2013;23:249---57.
15.RibeiroS,AlmeidaRR,CaovillaHH,Gananc¸aMM.Dospotenciais evocadosmiogênicosvestibularesnasorelhascomprometidae assintomáticanaDoenc¸adeMénièreunilateral.RevBras Oto-rrinolaringol.2005;71:60---6.
16.YoungYH,HuangTW,ChengPW.AssessingthestageofMénière’s disease using vestibular evoked myogenic potentials. Arch OtolaryngolHeadandNeckSurg.2003;129:815---8.
17.Waele C, Huy PT, Diard JP, Freyss G, Vidal PP. Saccu-lar dysfunction in Ménière’s disease. Am J Otol. 1999;20: 223---32.
18.SeoT, YoshidaK, ShibanoA, SakagamiM.A possiblecaseof saccularendlymphatichydrops.JOtorhinolaryngolRelatSpec. 1999;61:215---8.
19.LinMY,TimmerFC,OrielBS,ZhouG,GuinanJJ,KujawaSG, etal.Vestibularevokedmyogenicpotentials(PEMV)candetect asymptomatic saccular hydrops. Laryngoscope. 2006;116: 987---92.
20.Colebach JG, Halmagyi GM. Vestibular evoked potentials in humanneckmusclesbeforeandafterunilateralvestibular deaf-ferentation.Neurology.1992;42:1635---6.
21.Colebach JG, Halmagyi GM, Skuse NF. Myogenic potentials generated by a click-evoked vestibulocollic reflex. J Neurol NeurosurgPsychiatry.1994;57:190---7.
22.MurofushiT,HalmagyiGM,YavorRA,ColebatchJG.Absent vesti-bularevokedmyogenicpotentialsinvestibular neurolabyrinthi-tis:anindicatorofinferiorvestibularnerveinvolvement?Arch OtolaryngolHeadNeckSurg.1996;122:845---8.
23.Oliveira AC, Colafêmina JF, Menezes PL. Potencial evocado miogênicovestibularabaixasfrequênciasdeestimulac¸ão.Braz JOtorhinolaryngol.2011;77:706---10.
24.Murofushi T, Shimizu K, Takegoshi H, Cheng P-W.Diagnostic valueofprolongedlatenciesinthevestibularevokedmyogenic potential.ArchOtolaryngol.2001;127:1069---72.
25.Jariengprasert C, Tiensuwan M, Euasirirattanapaisan K. A comparison of vestibular evoked myogenic potential (VEMP) betweendefiniteMeniere’sdiseasepatientsandnormalhealthy adults.JMedAssoc.2013;96:1563---8.
26.LamounierP,GobboDA,deSouzaTS,deOliveiraCA,Bahmad FJr.ElectrocochleographyforMénière’sdisease:isitreliable. BrazJOtorhinolaryngol.2014;80:527---32.
27.YoungYH,HuangTW,ChengPW.AssessingthestageofMénière’s disease using vestibular evoked myogenic potentials. Arch OtolaryngolHeadNeckSurg.2003;129:815---8.
28.Adams ME, Heidenreich KD, Kileny PR. Audiovestibular test-inginpatientswithMénière’sdisease.OtolaryngolClinNAm. 2010;43:995---1009.
29.KingmaCM, Wit HP.Asymmetricvestibular evoked myogenic potentialsin unilateralMéniérepatients.EurArch Otorhino-laryngol.2011;268:57---61.
30.ConlonBJ,GibsonWP.Electrocochleographyinthediagnosisof Meniere’sdisease.ActaOtolaryngol.2000;120:480---3. 31.CalR,BahmadF.Potencialevocadomiogênicovestibular:uma
visãogeral.BrazJOtorhinolaryngol.2009;75:456---62. 32.SoaresLCA,ConegundesLSO,FukudaC,MunhozMSL.Da
eletro-cocleografiatranstimpânicaempacientescomesemhydrops endolinfático e limiares auditivos iguais ou maiores que 50 decibéis.RevBrasOtorrinolaringol.2003;69:74---82.
33.Kim HH, Kumar A, Battista RA, Wiet RJ. Electrocochleogra-phy in patients with Ménière’s disease. Am J Otolaryngol. 2005;26:128---31.