REVISTA
PAULISTA
DE
PEDIATRIA
www.rpped.com.br
ORIGINAL
ARTICLE
Acute
effect
of
vigorous
aerobic
exercise
on
the
inhibitory
control
in
adolescents
Rodrigo
Alberto
Vieira
Browne
a,∗,
Eduardo
Caldas
Costa
a,
Marcelo
Magalhães
Sales
b,
André
Igor
Fonteles
a,
José
Fernando
Vila
Nova
de
Moraes
c,
Jônatas
de
Franc
¸a
Barros
aaUniversidadeFederaldoRioGrandedoNorte(UFRN),Natal,RN,Brazil bUniversidadeCatólicadeBrasília(UCB),Brasília,DF,Brazil
cUniversidadeFederaldoValedoSãoFrancisco(Univasf),Petrolina,PE,Brazil
Received4April2015;accepted9August2015 Availableonline3February2016
KEYWORDS
Sports;
Physicaleducation andtraining; Cognition;
Executivefunction; Puberty
Abstract
Objective: Toassesstheacuteeffectofvigorousaerobicexerciseontheinhibitorycontrolin adolescents.
Methods: Controlled, randomized study with crossover design. Twenty pubertal individuals underwenttwo30-minutesessions:(1)aerobicexercisesessionperformedbetween65%and 75%ofheartratereserve,dividedinto5minofwarm-up,20minatthetargetintensityand5min ofcooldown;and(2)controlsessionwatchingacartoon.Beforeandafterthesessions,the computerizedStrooptest---TestinpacsTMwasappliedtoevaluatetheinhibitorycontrol.Reaction
time(ms)anderrors(n)wererecorded.
Results: Thecontrolsessionreactiontimeshowednosignificantdifference.Ontheotherhand, thereactiontimeoftheexercisesessiondecreasedaftertheintervention(p<0.001).The num-beroferrorsmadeattheexercisesessionwerelowerthaninthecontrolsession(p=0.011). Additionally,therewasapositiveassociationbetweenreactiontime()oftheexercisesession andage(r2=0.404,p=0.003).
Conclusions: Vigorousaerobicexerciseseemstopromoteacuteimprovementintheinhibitory controlinadolescents.Theeffectofexerciseontheinhibitorycontrolperformancewas asso-ciatedwithage,showingthatitwasreducedatolderageranges.
©2015SociedadedePediatriadeSãoPaulo.PublishedbyElsevierEditoraLtda.Thisisanopen accessarticleundertheCCBYlicense(https://creativecommons.org/licenses/by/4.0/).
∗Correspondingauthor.
E-mail:[email protected](R.A.V.Browne). http://dx.doi.org/10.1016/j.rppede.2016.01.005
PALAVRAS-CHAVE
Esporte;
Educac¸ãofísicae treinamento; Cognic¸ão;
Func¸ãoexecutiva; Puberdade
Efeitoagudodoexercícioaeróbiovigorososobreocontroleinibitórioem adolescentes
Resumo
Objetivo: Verificaroefeitoagudodoexercícioaeróbiovigorososobreocontroleinibitórioem adolescentes.
Métodos: Estudocontroladoerandomizadocomdelineamentocruzado.Vintepúberesforam submetidosaduassessõesde30minutos:1)sessãoexercícioaeróbiofeitoentre65---75%da fre-quênciacardíacadereserva,comcincominutosparaaquecimento,20minutosnaintensidade alvoecincominutosdevoltaàcalma;e2)sessãocontroleassistindoadesenhoanimado. Pre-viamenteeapósassessões,otestedeStroopcomputadorizado(Testinpacs®)foiaplicadopara
avaliarocontroleinibitório.Otempodereac¸ão(ms)eoserroscometidos(n)foramregistrados.
Resultados: Otempodereac¸ãodasessãocontrolenãoapresentoudiferenc¸asignificativa.Por outro lado,o tempo dereac¸ãoda sessãoexercício diminuiu apósaintervenc¸ão(p<0,001). Oserroscometidosnasessãoexercícioforammenoresdoquenasessãocontrole(p=0,011). Adicionalmente,houveassociac¸ãopositivadotempodereac¸ão()dasessãoexercíciocoma idade(r2=0,404;p=0,003).
Conclusões: Oexercícioaeróbiovigorosoparecepromovermelhoriaagudanocontroleinibitório emadolescentes.Oefeitodoexercíciosobreodesempenhodocontroleinibitóriofoiassociado àidadeedemonstrouserreduzidoemfaixasetáriasmaisaltas.
©2015SociedadedePediatriadeSãoPaulo.PublicadoporElsevierEditoraLtda.Esteéumartigo OpenAccesssobalicençaCCBY(https://creativecommons.org/licenses/by/4.0/deed.pt).
Introduction
Executivecontrol(or executivefunctions) referstohigher cognitiveprocessesthatmanagethecontrolofother,more basic cognitive functions and direct the ideal behavior toachievegoal-orientedbehaviors.1 Ingeneral,executive
control is subdivided into inhibitory control (IC),working
memoryandcognitiveflexibility.1The ICisconsidered the
maindomainofexecutivecontrolandadeterminantof
aca-demic success, asit controls attention, behavior, thought
and/or emotion to override a strong internal
predisposi-tion or external attraction and adapt itself to conflicting
situations.1
School activities constitute a model of environmental
requestregardingtheautonomyandcontrolofattentional,
organizationandplanningfunctions,whichrequiresan
effi-cient performance of the IC.2 Evidence suggests that the
development of IC skills during childhood promotes an
increasedcapacity for success in the development of the
theoryofmind---facilitatesthinkingandlearning3---aswell
asa betterperformance in counterfactual4 reasoningand
strategic5 tasks. The ICalso has been strongly associated
withintelligencelevel6andschoolperformance.7
Bothfrontal regions,cortical andsubcortical,subserve
theexecutive control.8 The prefrontalcortex(PFC) is the
onethat playsa keyrole.8Increasedbrain activityof the
PFCwasobservedduringthetaskmakingoftheIC(Stroop
Test).9 The PFC comprises from a quarter to a third of
the cerebral cortex and contains rich reciprocal
connec-tionswithitself,withother corticalareasandsubcortical
and limbic regions.10 The performance of executive
con-troldevelopsfromearlychildhood,throughoutadolescence,
to adulthood,11 concurrently with the neuroanatomical,
functional12 andbrainbloodperfusion13 changes,including
thePFCregions.
Physical exercise has been considered an important
environmentalfactorfor neurodevelopment,14 topromote
cognitiveand brain health,15 aswell as for a better
per-formance of the executive and school control.7 A single
sessionofaerobicexerciseshasbeenshowntoimprovethe
efficiencyof theIC inchildren16 and young adults,17---19 in
contrasttowhatwasobservedinadolescentsafter20minof
aerobicexerciseperformedonacycleergometerat60%of
maximumheartrate(HRmax).20Cognitiveperformanceafter
acuteexerciseseemstobedependentontheintensity.21In
themeta-analysisofChangetal.,21thestudiesthatusedlow
intensity,<50%ofHRmax,hadanegativeeffectoncognitive
performance.Ontheotherhand,instudieswithintensities
>64%ofHRmax,theeffectswerepositive.
Apossiblephysiologicalhypothesisthatmayexplainthe
acuteeffectofexerciseintensityontheICisincreased
cere-bralbloodflowgeneratedbyexerciseeffort,whichcanhave
an impactonpost-exercise cognitiveperformance.17---19 In
thestudy byYanagisawaetal.,19 therewasan increasein
cerebralbloodflow(↑oxygenatedhemoglobin)inthePFC
andimprovedperformanceintheStrooptestinyoungadults
after 10min of aerobic exercise at 50% of the peak
oxy-genconsumption(peakVO2).Thesameeffectwasobserved
insimilarexperimentscarriedoutwithyoungadultsafter
20minofexercisebetween60%and70%ofHRmax18andafter
15minofexerciseat40%ofmaximumload,respectively,17
andinchildrenafter20minofaerobicexercisebetween65%
and75%ofHRmax.16
However, there is still a gap of knowledge whether
a vigorous aerobic exercise session can improve the IC
in adolescents, which may be important, as their PFC is
still undergoing the maturation phase. It is a period of
structural,functional12andbloodperfusion13changes.The
studyhypothesisisthattheuseofaprescriptionofaerobic
confoundingfactorsassociatedwithcognitiveimprovement
byacuteexercise,mayfavorthephysiologicalbrain
mecha-nismsinducedbyexerciseandinfluencethepost-exerciseIC
performance,asshowninchildren16 andyoungadults.17---19
Therefore,theaimofthisstudywastoinvestigatetheacute
effectofvigorousaerobicexerciseontheICinadolescents.
Theintensity andvolumeof theexercisewere prescribed
accordingtovigorousexerciserecommendationsfor
adoles-centsfromtheWorldHealthOrganization22andaccordingto
thebesteffectobtainedbymeta-analysisofChangetal.21
Other control factors, such as time of the exercise and
thetimeintervaltoapplythepost-exercisecognitivetest,
werealsosupportedbythemeta-analysisofChangetal.21
Method
This was a controlled, randomized study with crossover design,carried out onthe seaside,in the municipality of Icapuí(CE). The acute effectof theexercise prescription protocolwastestedintwosessions,withaminimumof48h ofinterval,namely: (1) vigorousaerobicexercisesession; and(2)controlsessionwatchinganage-appropriatecartoon. Halfoftheparticipants,randomly,firstreceivedthe exper-imental treatment and then the control, while the other half firstreceivedthe controland after theexperimental treatment.Finally, the assessmentof ICperformance was performedbeforeandafterthesessions.
Sample size was calculatedusing the statistical power (1−ˇ), with analysis of variance being used in the main study outcome (Split Plot ANOVA), with an effect size of
f=0.333(consideredmiddle-sized)andanalphaof0.05.The statisticalpowergiventothissample,regardlessofgender, was80%(G*Power®,version3.1.9.2;Institutefor
Experimen-talPsychologyinDusseldorf,Germany).
Twentyadolescents(Table1)ofbothgendersand
physi-callyactive,between10and16yearsofage,wererandomly
enrolledfrompublicelementaryschoolsinthemunicipality
of Icapuí. Posters about the study were fixed in the
bul-letinboardsofschools torecruitvolunteers andalecture
wasgivenat apredeterminedtimeandlocationfor those
interested.Inclusioncriteriawere:(i)availabilitytoattend
theinitialassessmentandthecontrolandexercisesessions
inthe morning;(ii)to bephysically active,that is,tobe
enrolledandregularlypracticephysicalexercises(≥1year
and≥2×/week,respectively)inextracurricularsports
pro-gramstaking placein theschools in the shift opposite to
theschoolshift;(iii)meetthecriteriaofthephysical
activ-ityreadinessquestionnaire(PAR-Q);(iv)tobeclassifiedas
‘‘pubertal’’(Tanner stages 2---4)23;and (v) have no
physi-calor intellectual disabilitiesand/orclinical, neuromotor,
psychologicaland/orcognitivecontraindications.
The researchproject wasapprovedbytheInstitutional
Review Board of Universidade Federal do Rio Grande do
Norte (Protocol No. 876286/2014 CEP/UFRN), consistent
withtheDeclarationofHelsinkiandResolutionN.466/2012
oftheNationalHealthCouncil.Allselectedadolescentshad
theinformedconsentandassentform,PAR-Qand
medical-historyquestionnairefilledoutanddulysigned.
Bodyweightandheightweremeasuredbyamechanical
scale(G-Tech®)andastadiometerfixedtothewall(Sanny®),
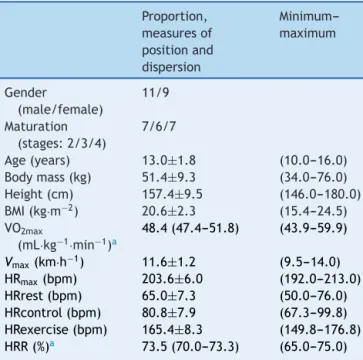
Table 1 Characterization of the sample of adoles-cents (n=20). Data expressed as mean and standard deviationfor parametricvariables,medianand95% confi-dence intervals for nonparametric variables and range (minimum---maximum).
Proportion, measuresof positionand dispersion
Minimum---maximum
Gender
(male/female)
11/9
Maturation (stages:2/3/4)
7/6/7
Age(years) 13.0±1.8 (10.0---16.0) Bodymass(kg) 51.4±9.3 (34.0---76.0) Height(cm) 157.4±9.5 (146.0---180.0) BMI(kg·m−2) 20.6±2.3 (15.4---24.5)
VO2max
(mL·kg−1·min−1)a
48.4(47.4---51.8) (43.9---59.9)
Vmax(km·h−1) 11.6±1.2 (9.5---14.0)
HRmax(bpm) 203.6±6.0 (192.0---213.0)
HRrest(bpm) 65.0±7.3 (50.0---76.0)
HRcontrol(bpm) 80.8±7.9 (67.3---99.8) HRexercise(bpm) 165.4±8.3 (149.8---176.8) HRR(%)a 73.5(70.0---73.3) (65.0---75.0)
HRcontrol,meanheartrateofthecontrolsession;HRexercise, meanheartrateoftheexercisesession;HRmax,maximumheart
rate; HRR, percentage of heartrate reserve at theexercise session;HRrest,restingheartrate;BMI,bodymassindex;Vmax,
maximumvelocity;VO2max,maximaloxygenuptake. a Nonparametricvariable.
respectively. The body mass index (BMI) was calculated throughtheformula:[BMI=bodymass(kg)×height(m)2].24
Sexual maturation wasevaluated by Tanner stage23 to
differentiatepre-pubertalindividualsfrompubertalones,as
inclusioncriteriainthestudy.25TheTablesconsistofimages
withcaptionsthatcharacterize thegenitalsandbodyhair
forboysandbreastsandbodyhairforgirls.Themethodand
itspurposewere individually explainedand,through
self-assessment,theadolescentspointedoutinaprivateform
whichimagestheirconditionmostresembled.Thepubertal
stagewasratedonascalefrom1to5,namely:
1=prepuber-tal;2---4=pubertal;and5=post-pubertal.
The maximal multistage 20-m shuttle run test26 was
appliedtoobtaintheHRmax.Thistestwasperformedinan
indoorsportscourt(between7and10h)andconsistsof
rac-ingbackandforthatadistanceof20meters.Thevelocity
wascontrolledbyametronomeaudio.The initialvelocity
was8.5km/h,followedbyincrementsof0.5km/hatevery
one-minutestage.Heartrate(HR)wasmonitored
through-outthe test bya heartrate monitor(Beurer®,Germany).
HRmaxwasconsideredasthehighestHRattainedduringthe
test,validonlywhensignsofintenseeffortwereobserved.27
Subjectswereverballymotivatedtoendureaslongas
possi-ble.Thetestwascontinueduntilvoluntaryexhaustion.The
maximumvelocity(Vmax)wasusedtoestimatethemaximum
The resting heartrate(HRrest)wasmeasured withthe
subjectatrestinthesupinepositionfor5min.Itwas
consid-eredthelowestHRobtained.TheHRreserve(HRR)wasthe
differencebetweenHRmaxandHRrest(HRR=HRmax−HRrest).
To determine the HR-targetfor exercise prescription, we
usedthe percentage of HRR. The percentage of HRR was
added to HRrest to determine the HR-targetin exercise:
HR-target=(%intensity(indecimal)×HRR)+HRrest.24
Onadifferentdayoftheexercisesession,subjectswere
familiarizedwithexerciseintensityandhowtostayonthe
HR-target.Attheexercisesession,awarm-upof5minwas
performed throughwalking,followedby 20min ofrunning
atvigorousintensity(65---75% ofHRR)and5minof cooling
down,totaling30min.TheHRwasmonitoredthroughoutthe
sessionbyaheartratemonitor(Beurer®,Germany),sothat
theparticipantremainedintheHR-target.Inadditiontothe
individualcontroloftheparticipantsthemselves,according
tothe guidelines givenduring the familiarization session,
theHRwasmonitoredandrecordedevery3min. If
neces-sary,verbaldirectionswereprovidedtoadjusttheintensity.
Theexercisewasperformedonthebeachsand(wetsand,
lowtideandflatterrain),withthesubjectsbarefoot.The
sessionswerecarriedoutbetween7and10AMwitha
tem-peratureandairhumiditybetween26---30◦Cand52%---74%,
respectively.
Duringthecontrolsession,thesubjectsremainedseated
for 30min in the school computer lab watching an
age-appropriate cartoon (Kung Fu Panda). HR was monitored
andrecordedevery3minbyaheartratemonitor(Beurer®,
Germany).
TheICperformancewasevaluatedbythecomputerized
Strooptest (Testinpacs®),withtheaid ofadesktopand a
14-inchmonitor.29Recallingwasdonebeforeeverysession.
Thetestwasappliedbeforeandafter10minofeachsession.
The instrument hasthree Phases, the first twocongruent
and the last one incongruent. The index and middle
fin-gersoftheright handremainedontheleft(←)andright
(→)arrowkeys,respectively,whichwereactivated
accord-ingtoeachstimulus.Instage1,rectanglesingreen,blue,
blackandredwereindividuallyshowninthecenterofthe
monitor.Inthelowercornersofthemonitor,answers
corre-spondingornottotherectanglecolorwereexhibiteduntil
the participantresponded tothe attemptby pressing the
keys←or →.Instage2,bothstimuliandresponseswere
shownaswords,alwaysinwhitecolor.Theresponsewas
con-sideredcorrectwhenthestimulusandresponsecoincided.
Finally,instage3, thewordofoneofthefourcolorswas
exhibitedinincompatiblecolor.Thesubjectwasinstructed
to press the key corresponding to the color of the word
andinhibittheresponsetothewordidentity.Atallstages
thestimuliwerepresentedautomaticallyandrandomly,12
attemptsperstage.Thereactiontime(RT)inmilliseconds
(ms)andthenumberoferrors(n)madeateachstagewere
recorded.
TheStatisticalprocedureswereperformedwithSPSSfor
Win/v.19.0(StatisticalPackageforSocialSciences,Chicago,
IL,USA)andG*Powerversion3.1.9.2(Institutefor
Experi-mentalPsychologyinDusseldorf,Germany).Thenormality
and homogeneity of data variance were tested by the
Shapiro---WilkstestandLevenetest,respectively.
Paramet-ricvariableswereexpressedasmeanandstandarddeviation
orstandarderror,andthenonparametricasmeanormedian
andtheirrespective95%confidenceintervals.Thelevelof
significancewassetatp<0.05.
ThereliabilityoftheStrooptest(RT)betweenbaseline
values (pre×pre) was assessed by Cronbach’s alpha (˛).
The ttest for independent sampleswasused tocompare
thebaselineRT betweenthe conditions(pre×pre).
Split-PlotAnova,adjustedforchronologicalage,wasappliedto
the comparison of intra- and inter-conditions of RT. The
hypothesisof sphericitywasverifiedby Mauchlytestand,
whenviolated,thedegreesoffreedomarecorrectedbythe
Greenhouse---Geisserestimates.Theeffectsizeofthe
vari-ance was calculated by the eta squared (2). The paired
t-test was applied to each group separately, in order to
locatethedifferences observedintheSplit-Plot. Thesize
effectwascalculatedbytheequation:
r=
t2
t2+gl
Quade’sNon-parametricAncova,adjustedby
chronolog-icalage,wasappliedtocomparethedeltas(=pre---post)
of the amount of errors made between the conditions
(control×exercise).Aftertheassessmentofnormalityand
homogeneity of the variance residues, the simple linear
regression(r2)andPearson’scorrelationcoefficient(r)were
appliedtoassociatetheRTdelta(=post-pre)ofthe
incon-gruentPhase3withchronologicalage.
Results
Among the adolescents selected for the study, it was observedthat65%had faileda schoolyear.Table1 shows
the variables of the sample characteristics, sexual
matu-ration,anthropometricmeasurements,estimatedvariables
andthoseobtainedbythemaximalmultistage20-mshuttle
runtest(VO2max,VmaxandHRmax)andmeanHRofthe
con-trolandexercisesessions.Furthermore,theHRR%meanat
whichtheadolescentsremainedduringtheexercisesession
wasdepicted.
TheRTofthestages1,2and3oftheStrooptestbetween
baseline values (pre×pre) had a reliability of ˛=0.502;
˛=0.493; ˛=0.752; respectively. There was no difference
between the baseline values of RT (pre×pre) in stage 1
(t[38]=−0.567;p=0.574),instage2(t[38]=−0.740;p=0.464)
andstage3(t[38]=−0.665;p=0.510).
Inthe congruentphase1ofthe Strooptest, the
inter-action time×conditions, adjusted for age did not differ
statistically,F(1,37)=1.98;p=0.168;2=0.051.Therewasno
differencebetweentheconditions,F(1,37)=0.00;p=0.982;
2=0.000. There was also no significant effect on time,
F(1,37)=2.79;p=0.103;2=0.070(Fig.1A).
The second congruent phase, stage 2, did not show
statistical difference in the time×conditions interaction,
adjusted for age, F(1,37)=2.28; p=0.139; 2=0.058. There
was no difference between the conditions, F(1,37)=0.00;
p=0.955; 2=0.000. There was a significant effect on
time,F(1,37)=4.66; p=0.037; 2=0.112. The paired t test
(pre×post) showedthat thecontrol conditiondid not
dif-fer,t(19)=2.05;p=0.055;r=0.43;buttheexercisecondition
In theincongruent phase,stage3, therewasan
inter-action time×conditions that was statistically significant,
adjustedbyage,F(1,37)=12.49;p=0.001; 2=0.252.There
wasnodifferencebetweentheconditions,F(1,37)=0.134;
p=0.716; 2=0.004. There was a significant effect on
time,F(1,37)=5.64; p=0.023; 2=0.132. The paired t test
(pre×post) showed that the control condition did not
differ,t(19)=0.64;p=0.532;r=0.15;buttheexercise
condi-tion significantly differed, t(19)=4.94; p<0.001; r=0.75
(Fig. 1C). The errors that were made, adjusted by age,
didnotdifferbetweenthecontrol×exerciseconditionsin
phase 1 [F(1,38)=0.105; p=0.748 (Fig. 2A)] and phase 2
[F(1,38)=0.045; p=0.834 (Fig.2B)], but phase3 showeda
significantdifference[F(1,38)=7.162;p=0.011(Fig.2C)].
Assecondaryanalysis,weverifiedapositiveassociation
(p=0.003) of RT of incongruent phase 3 of the exercise
–50
Control
Exercise
A
B
–75–100
–125
Dif. [post-pre] R
T
(ms)
Dif. [post-pre] R
T
(ms)
Dif. [post-pre] R
T
(ms)
C
*
*
–150–175
–200
–75
–100
–100 –50 0 –125
–150
–150 –175
–200
–200
–250 –225
Figure1 Acuteeffect ofvigorous aerobic exercise onthe reaction time (RT) in Phases 1 (A), 2 (B) and 3 (C) of thecomputerized Stroop Test (Testinpacs®). Split-Plot Anova
adjustedfor chronological agewas applied inthe intra- and inter-comparisons between conditions (2×2). The delta data (=post---pre)areshownasmeanandstandarderror.*p<0.001; pre×post.
conditionwithchronologicalage(Fig.3).Ontheotherhand,
there was no association of RT of phase 3 of the
con-trol conditionwith chronological age (r2=0.021;p=0.545).
Samplesizecalculation,afterwards,oftheanalysisof
asso-ciationwascarriedoutwithanalphaof0.05,sample size
of20subjectsandacoefficientofcorrelationof0.635.The
statisticalpowergivenintheanalysiswas88%.
Control
Exercise 0.6
0.4
0.2
–0.2
–0.4
–0.6 0.0
Dif. [post-pre] error (n)
Dif. [post-pre] error (n)
Dif. [post-pre] error (n)
A
0.6
0.4
0.2
–0.2
–0.4
–0.6
1.2
0.8
0.4
–0.4
–0.8
–1.2 0.0 0.0
B
C
*
Figure 2 Acute effectof vigorous aerobic exercise on the errorsmadeinPhases1(A),2(B)and3(C)ofthecomputerized StroopTest(Testinpacs®).Non-parametricAncovaadjustedfor
9 –800 –700 –600 –500 –400 –300 400
r=0.635, p=0.001 r2=0.404, p=0.003
300
–200 200
–100 100 0
10 11 12 13
Chronological age (years)
Dif. [post-pre] R
T
(ms)
14 15 16 17
Figure3 Linearregression(r2)andPearson’scoefficient
cor-relation(r)betweenthedelta(=post---pre)oftheincongruent reactiontime(RT)ofStrooptestPhase3oftheexercisesession withchronologicalage.
Discussion
Thepresentstudyinvestigatedtheacuteeffectofvigorous aerobicexerciseontheICinadolescents.Themainfindings suggestthatvigorousaerobicexerciseacutelyenhancesthe performanceoftheIC,astherewasanimprovementonthe Stroopinterference(inhibitionofresponse)observedbythe decreaseintheRToftheincongruentphase(Fig.1C),with
afocaleffectconsideredlarge(r=0.75),accompaniedbya
smallernumberoferrorsmade(Fig.2C).Theseresultscan
beveryimportanttoelucidatetheinfluenceofexerciseon
theICefficiencyand,therefore, contributetotheprocess
oflearningintheschoolenvironment.2
TheinfluencethatexercisepromotedonICperformance
isinaccordancewiththeevidencefromstudiescarriedout
withchildren16 and young healthy adults.17---19 As exercise
intensity is an important factor to enhance post-exercise
cognitiveperformance,21thisstudyadoptedavigorous
exer-ciseprotocol,whichinadditiontobeingrecommendedby
theWorldHealthOrganizationforadolescents,22 wouldbe
adequatetoinducethephysiologicalmechanisms
responsi-bleforfavoringthecognitiveperformance.21Forinstance,
inameta-analysis,theintensityoftheexercisehada
sig-nificant influence sothat the prescribedexercise <50%of
HRmax resulted ina significant negativeeffect (d=−0.113)
oncognitiveperformance,butwhenprescribedat64---76%
or77---93%oftheHRmax,theresultswerepositive,withan
effectof0.202and0.268,respectively.21
Inadditiontoexerciseintensity,othercontrolledfactors
mayhavebeenequallyimportantfortheexercisetofavor
thecognitivebenefits,includingthevolumeandtimeofthe
exercise,aswell asthetimeintervalfor the
implementa-tionof thepost-exercise cognitivetest. Inthissense, the
exercise volume was also adjusted according to the
rec-ommendations of the World Health Organization22 on the
volumeof vigorousexercisefor adolescentsand,asother
studies have shown, resulting in cognitive benefits after
20minofaerobicexerciseinchildren16andyoungadults.18
Thetimeintervaltoapplythecognitivetestwasdefined
between 10 and 20min post-exercise, according to the
best effect size obtained by the meta-analysis of Chang
et al.,21 in which this control factor was considered a
primary moderator of cognitive benefits. For instance,
Changetal.21 categorizedthetimetoapplythecognitive
testsin0---10min; 11---20min or>20minaftertheexercise
and obtained an effect size of −0.060, 0.262 and 0.171
respectively.Thatis,thepost-exerciseapplicationtime
sig-nificantly influenced the effect size. The positive effects
wereobservedonlyafter11minpost-exercise.
The statistical findings of the meta-analysis by Chang
et al.21 on the time interval for the application of the
post-exercise cognitive tests is consistent with
reticular-activating hypofrontalitymodel proposed by Dietrich and
Audiffren,30 whichsuggests adecrease inbrain activation
in regions not directly associated with physical exercise
(i.e.,PFC)tosupplydirectlyassociatedregions.
Corroborat-ingthisunderstanding,thestudy byWangetal.31 showed
adeclinein ICperformance duringvigorousaerobic
exer-cise,whichcouldinfluenceICefficiencyimmediatelyafter
theexercise.Thus,thesestudiesandthistheoreticalmodel
agreewiththeneed for atimeinterval betweenthe end
of the exercise and the beginning of a cognitive task for
homeostasistooccuratthebrainlevel,morespecificallyin
thePFC.
Our data show that aerobic exercise seems to have
been a tool capable of acutely influencing the
physiolog-icalmechanisms responsible for favoring IC performance.
The mechanisms arestill somewhat controversial and
lit-tle explored, but the main physiological hypothesis that
couldexplainthese effects refer tothe increase in
cere-bralbloodflow,whichcaninfluencecognitiveperformance
afterexercise.17---19Forinstance,Yanagisawaetal.,19using
near-infrared spectroscopy (NIRS), evaluated the cortical
activation during Stroop test performed before and after
10minofaerobicexerciseat50%of peakVO2 in20young
adults.Infact,therewasincreasedbloodflowfromthe
lat-eralPFCinbothhemispheres(↑oxygenatedhemoglobin)due
toStroopinterference(incongruence).Incontrast,this
acti-vationwassignificantlyincreasedintheleftdorsolateralPFC
andcoincidedwithimprovedperformanceonthecognitive
task(↓RT).
As secondary analysis,we demonstratedan association
ofRToftheincongruentphase(stage3)oftheexercise
condition withchronological age (r2=0.404) (Fig. 3). That
is,post-exerciseICimprovementwasreducedinolderage
groups. The regression analysis shows that 40.4% of the
variability found in post-exercise IC performance can be
explainedby chronological age.In this sense,due to
evi-dencethatoneofthephysiologicalmechanismsresponsible
forthe improvementof post-exercise ICcanbeincreased
cerebralbloodflow,17---19thevariabilityincanberelated
tothematurationofthecerebralperfusionmechanism
dur-ingadolescence.13Notably,Satterthwaiteetal.13 observed
asignificantimpactofpubertyoncerebralperfusion
devel-opment.The authorsevaluatedthecerebralblood flowof
922youngindividualsagedeightto22yearsandfoundthat,
duringadolescence, the brainflow throughout the cortex
decreasesconsiderably,includinginthePFC.The cerebral
blood flow of the middle gray matter undergoes a sharp
declineinlatechildhoodandearlyadolescenceuntilabout
16---18years,followedbyaslightincreaseduringearly
adult-hood.
Althoughoriginalthisstudyhaslimitations,oneofwhich
with a neuroimaging technique, which could promote
greatersupport inconfirming the evidence.Nevertheless,
severalstudieshaveconfirmedthatacuteaerobicexercise
canimproveICatthemacroneurallevel,asitpromotes
sig-nificantimpactonPFCbrainactivityduringICtaskmaking
after10---20minofaerobicexerciseinchildren16andyoung
adults.17,18
Theuseofsexualmaturationself-assessmentprocedure
todistinguish thepre-pubertyandpubertyasaninclusion
criterion is also considered a limiting factor. The
evalu-ation by visual inspection by a trained evaluator is the
most reliable indirect method.25 However, it can be
jus-tifiedthat the sample consisted mostly of adolescents at
the pubertal stage.25 In the study by Rasmussen et al.,25
carried out with 898 children and adolescents, the
self-assessmentofsexualmaturationshowedtobeasufficiently
accuratetechniquetosimplydifferentiatesimplesbetween
prepubertalandpubertalstages.Breaststagewascorrectly
assessed by 44.9% of the girls (=0.28; r=0.74; p<0.001)
and the genital stage by 54.7% of boys (=0.33; r=0.61;
p<0.001).Pubichairwascorrectlyassessedby66.8%ofgirls
(=0.55;r=0.80;p<0.001)and66.1%ofboys(=0.46;r=0.70;
p<0.001).
It is noteworthy that this study is a pioneer in
inves-tigating the acute effects of vigorous aerobic exercise
on IC in adolescents and contributes to elucidate the
effectsof aerobicexercisein this population,which have
an executive control performance11 and brain functions,
structures12 and perfusion13 still undergoing maturation.
Additionally, thestudy hadthe premiseof controllingthe
possiblevariables,suchastheequalnumberofadolescents
of both genders, obtaining the HRmax through a maximal
effort progressive running test, the intensity and volume
ofthe experimentalcondition exercise,the timeof
phys-ical exercise, the use of a computerized cognitive test
that records the performance in milliseconds, and the
time for application of the post-exercise cognitive test.
Therefore,the study mayprovide subsidies for the
appli-cability of this aerobic exercise protocol in the school
context.32
Inconclusion, avigorousaerobicsessionperformed for
20min seems to promote improvement in IC capacity in
adolescents.Additionally, the effectof exerciseontheIC
performance was associated with chronological age. This
demonstratesthat the benefits of exercise were reduced
in older age groups. As practical implications, the
find-ings of this study contribute to justify the inclusion of
physical education classes during school hours, that is,
among the other school subjects (i.e., Portuguese and
mathematics), as well as the inclusion of a physically
activeinterval/recess,as20minof vigorousaerobic
exer-cise can favor the IC efficiency and therefore contribute
to learning improvement after 10min of post-exercise
recovery.
Funding
Master’s Degree Scholarship granted by Programa de DemandaSocial (DS)of Coordenac¸ão de Aperfeic¸oamento dePessoaldeNívelSuperior(Capes)tothefirstauthor(RAV Browne).
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
Acknowledgements
ToProf.AntônioJosédaCosta,whoprovidedapprovaland welcomefor theresearchtobe carriedoutlocally at the educationalinstitutionofwhichheistheprincipal,Professor JoanaMarquesBezerraElementarySchool,inthe municipal-ityofIcapuí(CE).
References
1.Diamond A. Executive functions. Annu Rev Psychol. 2013;64:135---68.
2.CypelS. O papel dasfunc¸õesexecutivas nostranstornos da aprendizagem. In: Rotta N, Ohlweiler L, Riesgo R, editors. Transtornosdaaprendizagem:abordagemneurobiológica emul-tidisciplinar.PortoAlegre:Artmed;2006.p.375---87.
3.Benson JE, Sabbagh MA, Carlson SM, Zelazo PD. Individ-ualdifferencesinexecutivefunctioningpredictpreschoolers’ improvement from theory-of-mind training. Dev Psychol. 2013;49:1615---27.
4.BeckSR,RiggsKJ,GorniakSL.Relatingdevelopmentsin chil-dren’scounterfactualthinkingandexecutivefunctions.Think Reason.2009;15:337---54.
5.ApperlyIA,CarrollDJ.Howdosymbolsaffect3-to4-year-olds’ executivefunction?Evidencefromareverse-contingencytask. DevSci.2009;12:1070---82.
6.Lee HW, Lo YH, Li KH, SungWS, JuanCH. Therelationship between thedevelopmentof responseinhibition and intelli-genceinpreschoolchildren.FrontPsychol.2015;6:802.
7.VanderNietAG,HartmanE,SmithJ,VisscherC.Modeling rela-tionshipsbetweenphysicalfitness,executivefunctioning,and academicachievementinprimaryschoolchildren.PsycholSport Exerc.2014;15:319---25.
8.Badre D. Cognitive control, hierarchy, and the rostro-caudal organization of the frontal lobes. Trends Cogn Sci. 2008;12:193---200.
9.Zhang L, Sun J, Sun B, Luo Q, Gong H. Studying hemi-sphericlateralizationduringaStrooptaskthroughnear-infrared spectroscopy-basedconnectivity.JBiomedOpt.2014;19:57012.
10.Olson EA, LucianaM. The development ofprefrontal cortex functions inadolescence: Theoretical models and a possible dissociationofdorsalversusventralsubregions.In:NelsonCA, LucianaM,editors.Handbookofdevelopmentalcognitive neu-roscience.2nded.Cambridge:MITPress;2008.p.575---90.
11.RomineCB,ReynoldsCR.Amodelofthedevelopmentoffrontal lobefunctioning:findingsfromameta-analysis.Appl Neuropsy-chol.2005;12:190---201.
12.Blakemore SJ. Imaging brain development: the adolescent brain.Neuroimage.2012;61:397---406.
13.Satterthwaite TD, Shinohara RT, Wolf DH, et al. Impact of pubertyontheevolutionofcerebralperfusionduring adoles-cence.ProcNatlAcadSciUSA.2014;111:8643---8.
14.HertingMM,ColbyJB,SowellER,NagelBJ.Whitematter con-nectivity and aerobic fitness in male adolescents. Dev Cogn Neurosci.2014;7:65---75.
15.KhanNA,HillmanCH.Therelationofchildhoodphysicalactivity andaerobicfitnesstobrainfunctionand cognition:areview. PediatrExercSci.2014;26:138---46.
performance in children with ADHD. J Pediatr (Rio J). 2013;162:543---51.
17.EndoK,MatsukawaK,LiangN,etal.Dynamicexerciseimproves cognitivefunctioninassociationwithincreasedprefrontal oxy-genation.JPhysiolSci.2013;63:287---98.
18.LiL, MenWW,ChangYK,Fan MX,JiL, WeiGX. Acute aero-bicexerciseincreasescorticalactivityduringworkingmemory: afunctionalMRIstudyinfemalecollegestudents.PLoSOne. 2014;9:e99222.
19.YanagisawaH,DanI,TsuzukiD,etal.Acutemoderate exer-cise elicits increased dorsolateral prefrontal activation and improvescognitiveperformancewithStrooptest.Neuroimage. 2010;50:1702---10.
20.StrothS,KubeschS,DieterleK,RuchsowM,HeimR,KieferM. Physicalfitness,butnotacuteexercisemodulatesevent-related potentialindicesforexecutivecontrolinhealthyadolescents. BrainRes.2009;1269:114---24.
21.ChangYK,LabbanJD,GapinJI,EtnierJL.Theeffectsofacute exerciseoncognitiveperformance:ameta-analysis.BrainRes. 2012;1453:87---101.
22.WorldHealthOrganization.Globalrecommendationson physi-calactivityforhealth.Geneva:WHO;2010.
23.TannerJ.Growthatadolescencewithageneralconsideration oftheeffects ofhereditary and environmental factorsupon growthandmaturationfrombirthtomaturity.2nded.Oxford: BlackwellScientificPublications;1962.
24.HeywardVH.Avaliac¸ãofísicaeprescric¸ãodeexercício:técnicas avanc¸adas.6thed.PortoAlegre:Artmed;2013.
25.Rasmussen AR, Wohlfahrt-Veje C, Renzy-Martin KT, et al. Validityofself-assessmentofpubertalmaturation.Pediatrics. 2015;135:86---93.
26.LégerLA,LambertJ.Amaximalmultistage20-mshuttlerun test to predict VO2 max. EurJ Appl Physiol Occup Physiol. 1982;49:1---12.
27.ArmstrongN, Welsman JR. Assessmentand interpretationof aerobicfitnessinchildrenandadolescents.ExercSportSciRev. 1994;22:435---76.
28.LégerLA, MercierD, GadouryC,LambertJ. The multistage 20 metre shuttle run test for aerobic fitness. J Sports Sci. 1988;6:93---101.
29.Córdova C, Karnikowski M, Pandossio J, Nóbrega O. Caracterizac¸ão de respostas comportamentais para o teste de Stroop computadorizado-Testinpacs. Neurociencias. 2008;4:75---9.
30.Dietrich A,AudiffrenM.Thereticular-activating hypofrontal-ity (RAH) model of acute exercise. Neurosci Biobehav Rev. 2011;35:1305---25.
31.WangCC,ChuCH,ChuIH,ChanKH,ChangYK.Executive func-tionduringacuteexercise:theroleofexerciseintensity.JSport ExercPsychol.2013;35:358---67.