Woody community dynamics in two fragments of "cerrado"
stricto sensu
over a seven-year period (1995-2002), MA, Brazil
FABIANA DE GOIS AQUINO
1,3, BRUNO MACHADO TELES WALTER
2and
JOSÉ FELIPE RIBEIRO
1(received: July 5, 2006; accepted: January 4, 2007)
ABSTRACT – (Woody community dynamics in two fragments of “cerrado” stricto sensu over a seven-year period (1995-2002),
MA, Brazil). This study was conducted in two fragments of “cerrado” stricto sensu in the Gerais de Balsas Colonization
Project, located in southern Maranhão, Brazil. The objective was to evaluate the dynamics of the woody plant community, over seven years (1995-2002). Four transects of 160 × 20 m were monitored. All woody plants with a stem diameter ≥ 3 cm, at 0.30 m
above ground level, were recorded. In 1995, 983 and 1,177 stems were sampled in fragments 1 and 2, respectively; in 2002, 1057 and 1406 stems were sampled in the same fragments. In 1995, the Shannon diversity indices (H’) were 3.07 and 3.33, in
fragments 1 and 2, respectively, reaching their maximum value in 2002 of 3.11 and 3.35. The community of fragment 1 showed an increase of 7.5% in density and 4.4% in basal area between 1995 and 2002, while in fragment 2 there was an increment of 19.4% in density and 23.5% in basal area, over the same period. The annual increment in diameter was 0.13 cm year-1 and
0.17 cm year-1 in fragments 1 and 2, respectively. The mortality rate was 2.73% per year in fragment 1 and 4.88% per year in
fragment 2, while the recruitment rate was 3.25% per year and 5.86% per year, respectively. The community presented high recruitment and mortality rates compared to the studies conducted in other sites, indicating a community that was highly dynamic in the period studied.
Key words - fragmentation, permanent plots, plant mortality, recruitment, savanna
RESUMO – (Dinâmica de uma comunidade lenhosa em dois fragmentos de cerrado stricto sensu no período de sete anos
(1995-2002), MA, Brasil). O presente estudo foi conduzido em dois fragmentos de cerrado stricto sensu, no Projeto de Colonização
Gerais de Balsas, sul do Maranhão, Brasil. O objetivo do estudo foi avaliar a dinâmica da comunidade lenhosa no período de sete anos (1995-2002). Foram estabelecidos quatro transectos de 160 × 20 m nos fragmentos, onde foram marcadas todas as plantas lenhosas com diâmetro do caule ≥ 3 cm, medidos a 0,30 m acima do solo. Em 1995, 983 e 1.177 plantas foram registradas
nos fragmentos 1 e 2, respectivamente, e em 2002, 1.057 e 1.406 plantas foram amostradas nos mesmos locais. Em 1995, o índice de diversidade de Shannon (H’) foi de 3,07 e 3,33, nos fragmentos 1 e 2, respectivamente, alcançando o valor máximo em 2002
(3,11 e 3,35). A comunidade do fragmento 1 aumentou 7,5% em densidade e 4,4% em área basal entre 1995 e 2002, enquanto que o fragmento 2 apresentou aumento de 19,4% na densidade e 23,5% em área basal, no mesmo período. O incremento anual em diâmetro foi 0,13 cm ano-1 e 0,17 cm ano-1 nos fragmentos 1 e 2, respectivamente. A taxa de mortalidade foi 2,73% ano-1 no
fragmento 1 e 4,88% ano-1 no fragmento 2, enquanto a taxa de recrutamento foi 3,25% ano-1 e 5,86% ano-1, respectivamente. A
comunidade vegetal estudada apresentou altas taxas de recrutamento e mortalidade comparadas à outros trabalhos conduzidos em diferentes localidades, indicando que se trata de uma comunidade altamente dinâmica no período estudado.
Palavras-chave - cerrado, fragmentação, parcelas permanentes, mortalidade, recrutamento
Introduction
“Cerrado”
stricto sensu is a savanna vegetation
that presents a continuous grass layer overlaid by a
discontinuous tree and shrub layer. This vegetation is
one of the main physiognomies located in the Brazilian
"cerrado" biome (Eiten 1972, Ribeiro & Walter 1998,
Oliveira-Filho & Ratter 2002). Originally, the “cerrado”
biome covered about two million sq. kilometers in the
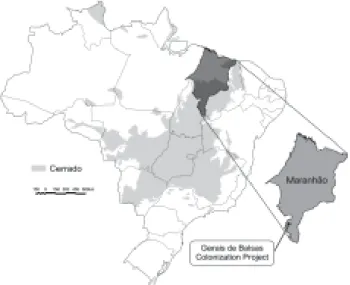
Brazilian heartland (figure 1) and it is considered one of
the world’s 25 hotspots (Mittermeier et al. 1999, Myers
et al. 2000) partly because of the number of endemic
plants, estimated at about 4400 vascular species, and
partly because it is endangered. This region is
threatened mainly by the high rate of agricultural
expansion throughout native areas, and due to the small
percentage of protected areas (2.5%) (Klink et al. 1993,
Ratter
et al. 1997).
Many studies have provided important information
about the “cerrado” stricto sensu vegetation, detailing
phytogeographical, physiognomic, floristic and
phytosociological patterns (e.g. Ratter & Dargie 1992,
1. Embrapa Cerrados, BR 020, Km 18, Caixa Postal 08223,
73301-970 Planaltina, DF, Brazil.
2. Embrapa Recursos Genéticos e Biotecnologia, Parque Estação
Biológica, 70770-900 Brasília, DF, Brazil.
Castro
et al. 1999, Felfili et al. 1994, Ratter et al. 1996,
Mendonça
et al. 2000, Ratter et al. 2003). However, it
is also very important to evaluate dynamic changes in
natural vegetation over time to understand the processes
and mechanisms that maintain the community.
Furthermore, this knowledge is important in the support
of conservation programs.
Unfortunately, as pointed out by Felfili et al. (2000),
there is still little information about structural changes
in the “cerrado” stricto sensu vegetation. There is an
absence of information concerning the influence of the
intense agricultural activities on sites of “cerrado”
stricto sensu maintained as natural reserves (fragments
of natural vegetation). Studies of community dynamics
in “cerrado” stricto sensu are essentially restricted to
Silberbauer-Gottsberger & Eiten (1987) in the state of
São Paulo, and Sato & Miranda (1996), Felfili et al.
(2000) and Henriques & Hay (2002) in Brasília, Federal
District, Brazil.
The objective of this study was to evaluate changes
in floristic composition, diversity and dynamic processes
in the woody communities in two fragments of “cerrado”
stricto sensu, in southern Maranhão State, Brazil, over
a seven-year period (1995-2002).
Material and methods
Study area – The study was carried out in two fragments of “cerrado”stricto sensu, located in the Gerais de Balsas
Colonization Project (8º29’ - 8º41’ S and 46º52’ - 46º38’ W), southern Maranhão State, Brazil (figure 1). This Project is part of the Nippon-Brazilian Cooperation Program for Development of the “cerrado” (Prodecer).
The climate of this region is Aw by the Köppen classification, with an average annual precipitation of 1049 mm and average temperature of 26 ºC (from Embrapa “cerrados” dataset). The dry season is from April to October, and the rainy season from November to March.
The Gerais de Balsas Colonization Project was established in 1995, with approximately 35,000 ha. In 1994, before its implementation, 99.7% of the area was composed of native vegetation and the remaining areas were used for subsistence cultivation. In 1995, with the Project in place, half of the area was deforested and converted for commercial agriculture and the remaining area was designated as private reserves - fragments of natural vegetation. This study was developed in two fragments denominated: fragment 1 (3,500 ha) and fragment 2 (1,500 ha) (figure 2). These fragments were monitored from 1995, when the adjacent areas to the fragments were deforested, until 2002.
Fires were registered in both fragments in 1996, 1998, 2000 and 2002. Fragment 2 was intensively burned in 1998.
Continuous inventory – Four transects of 160 m, subdivided into 16 contiguous plots of 10 × 20 m, were established in 1995, with two transects in each fragment. This allowed an area of 1.28 hectares to be monitored. The transects were allocated perpendicular to the edge. They are part of a larger research project that seeks to evaluate the edge effect along 160 meters inside the remaining fragments; for that, contiguous plots that run perpendicular to the edge were used. The first assessment was conducted at the beginning of the wet season of 1995 and the other five inventories occurred in: 1996, 1997, 1998, 2000 and 2002, always in the same season. All woody plants with a stem diameter ≥ 3 cm, at 0.30 m above ground level, were recorded, tagged and taxonomically identified (sensuAPG II 2003). Plants were
identified and vouchers were collected and deposited in the Figure 1. Map of Brazil showing the “cerrado” biome distribution and, in detail, Maranhão State with Gerais de Balsas Colonization Project.
Figure 2. Gerais de Balsas Colonization Project with two areas designated as private reserves - fragment 1 (F1) and fragment 2 (F2). ( = “Cerrado” stricto sensu; = Gallery Forest;
Embrapa Recursos Genéticos e Biotecnologia herbaria (CEN), in Brasília, Brazil. A nail in the stem allowed the position for diameter measurement to be standardized in successive assessments. Total height and stem diameter were measured during each survey.
A species list was produced for each fragment studied for each survey. Shannon’s diversity and Czekanowski’s similarity indices were obtained to compare the fragments.
Annual increment in diameter was calculated in each fragment from the average of the differences in increment between the first and last assessments, divided by the study period (seven years).
Mean annual mortality and recruitment rates were calculated using the log-model (Lieberman et al. 1985, Korning
& Balslev 1994, Sheil et al. 1995):
r = (Ct / C0)1/t– 1
where:r is the mean annual mortality (r < 0) or recruitment
(r > 0) rate, t is the elapse of time in years, and C0 and Ct are
the stand sizes at time 0 and after t years, respectively. According to Oliveira Filho et al. (1997) this calculation
allows one to evaluate the respective half-lives and doubling times calculated from the above rate. Half-life [t1/2 = ln(0,5) / ln (1 + r)] is the time required by the community
to decrease its size to half considering the present mortality rate, while doubling time [t2 = ln (2) / ln (1 + r)] is the time taken
to double its size considering the present recruitment rate. Turnover and stability values were estimated from the average of doubling time and half-life, and the numerical difference between them, respectively (Korning & Balslev 1994).
Results
In 1995, 983 and 1177 stems
≥
3 cm were sampled
in the permanent plots of fragments 1 and 2, respectively,
and in the last survey, in 2002, 1,057 and 1,406 stems
were sampled in each fragment. In fragment 1, 48
species were recorded in the first two assessments
(1995, 1996). In 2000, the single individual representative
of
Dimorphandra mollis Benth. died. In 2002, six new
species were registered, Dimorphandra gardneriana
Tul.,
Emmotum nitens (Benth.) Miers, Machaerium
acutifolium
Vogel,
Miconia albicans (Sw.) Triana,
Pterodon emarginatus
Vogel and, Salacia elliptica
(Mart. ex Schult.) G. Don, resulting in a total of 53
species. In fragment 2, 55 species were registered in
the first assessment (1995), and four new species were
included in the next two assessments (1996, 1997):
Casearia sylvestris Sw., Miconia ferruginata
DC.,
Neea theifera Oerst. and
Stryphnodendron
adstringens
(Mart.) Coville. In the 1998 survey,
Casearia sylvestris,
Guapira graciliflora (Mart. ex
J. A. Schmidt) Lundell, Emmotum nitens and Rourea
induta Planch. were not registered in the plots, and in
2000,
Neea theifera,
Stryphnodendron adstringens
and
Couepia grandiflora (Mart. & Zucc.) Benth. ex
Hook. f. were not registered. Finally, in 2002, five species
(Copaifera langsdorffii Desf., Dimorphandra mollis,
Emmotum nitens,
Rourea induta and,
Stryphnodendron adstringens) were included,
resulting in a total of 57 species.
In 1995, the diversity indices were 3.07 and 3.33, in
fragments 1 and 2, respectively, reaching a maximum
value in 2002 in both sites of 3.11 and 3.35. The
Czekanowski index varied from 0.46 to 0.45, indicating
that the sites studied showed qualitative similarity,
although quantitative dissimilarity was shown by the
varying number of individuals of each species between
fragments.
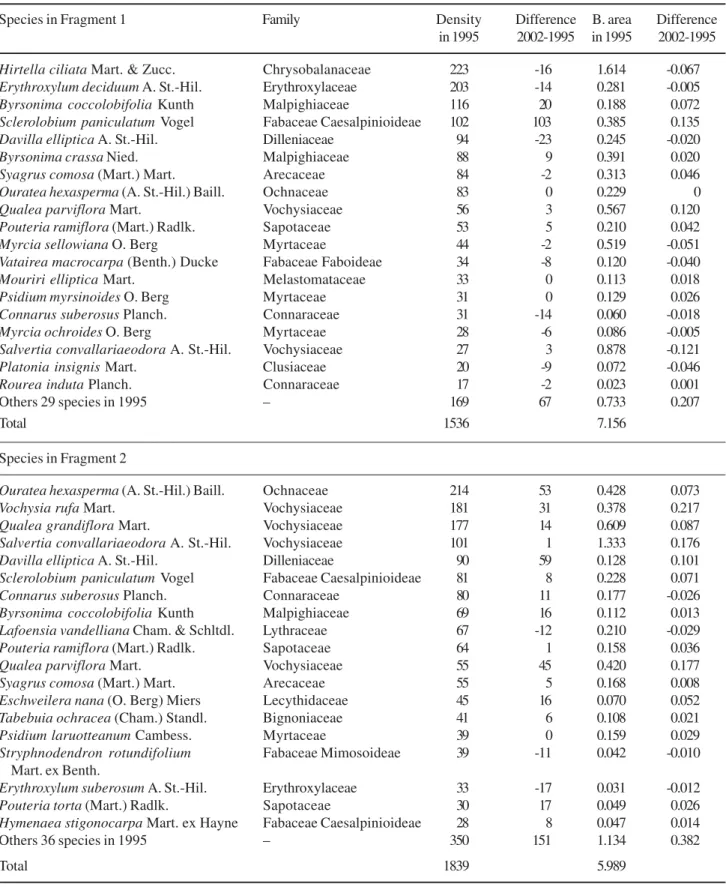
Density and basal area for 19 species with highest
number of individuals in 1995 are given in table 1. In
fragment 1, ten species (Hirtella ciliata,
Erythroxylum
deciduum,
Byrsonima coccolobifolia, Sclerolobium
paniculatum, Davilla elliptica, Byrsonima crassa,
Syagrus comosa, Ouratea hexasperma, Qualea
parviflora
and Pouteria ramiflora) represented 72%
of the total density and 62% of total basal area in 1995.
In 2002, the same ten species contained 72% and 64%
of the total density and basal area, respectively.
Although these ten species have mostly shown similar
percentages over the seven years in relation to density
and basal area, certain species showed considerable
changes in these parameters. For instance,
Sclerolobium paniculatum more than doubled its
density.
Connarus suberosus and Platonia insignis
halved their densities. Salvertia convallariaeodora
increased in density and presented a negative
difference in basal area. This was because one
individual with an 18 cm diameter died, while the
recruitment included individuals with smaller diameter,
close to the lower limit established in the method
adopted.
Ouratea hexaspema,
Mouriri elliptica and
Psidium myrsinoides maintained the same densities
over the seven years.
In fragment 2, ten species (Ouratea hexasperma,
Vochysia rufa,
Qualea grandiflora,
Salvertia
convallariaeodora,
Davilla elliptica,
Sclerolobium
paniculatum,
Connarus suberosus,
Byrsonima
coccolobifolia,
Lafoensia vandelliana and Pouteria
Table 1. Density (number of stems ha-1) and basal (B.) area (m2ha-1) for 19 species with highest number of individuals in 1995,
in the “cerrado” sensu stricto, Gerais de Balsas Colonization Project, Maranhão, Brazil.
Species in Fragment 1 Family Density Difference B. area Difference
in 1995 2002-1995 in 1995 2002-1995
Hirtella ciliata Mart. & Zucc. Chrysobalanaceae 223 -16 1.614 -0.067
Erythroxylum deciduum A. St.-Hil. Erythroxylaceae 203 -14 0.281 -0.005
Byrsonima coccolobifolia Kunth Malpighiaceae 116 20 0.188 0.072
Sclerolobium paniculatum Vogel Fabaceae Caesalpinioideae 102 103 0.385 0.135
Davilla elliptica A. St.-Hil. Dilleniaceae 94 -23 0.245 -0.020
Byrsonima crassa Nied. Malpighiaceae 88 9 0.391 0.020
Syagrus comosa (Mart.) Mart. Arecaceae 84 -2 0.313 0.046
Ouratea hexasperma (A. St.-Hil.) Baill. Ochnaceae 83 0 0.229 0
Qualea parviflora Mart. Vochysiaceae 56 3 0.567 0.120
Pouteria ramiflora (Mart.) Radlk. Sapotaceae 53 5 0.210 0.042
Myrcia sellowiana O. Berg Myrtaceae 44 -2 0.519 -0.051
Vatairea macrocarpa(Benth.) Ducke Fabaceae Faboideae 34 -8 0.120 -0.040
Mouriri elliptica Mart. Melastomataceae 33 0 0.113 0.018
Psidium myrsinoides O. Berg Myrtaceae 31 0 0.129 0.026
Connarus suberosus Planch. Connaraceae 31 -14 0.060 -0.018
Myrcia ochroides O. Berg Myrtaceae 28 -6 0.086 -0.005
Salvertia convallariaeodora A. St.-Hil. Vochysiaceae 27 3 0.878 -0.121
Platonia insignis Mart. Clusiaceae 20 -9 0.072 -0.046
Rourea induta Planch. Connaraceae 17 -2 0.023 0.001
Others 29 species in 1995 – 169 67 0.733 0.207
Total 1536 7.156
Species in Fragment 2
Ouratea hexasperma (A. St.-Hil.) Baill. Ochnaceae 214 53 0.428 0.073
Vochysia rufaMart. Vochysiaceae 181 31 0.378 0.217
Qualea grandiflora Mart. Vochysiaceae 177 14 0.609 0.087
Salvertia convallariaeodora A. St.-Hil. Vochysiaceae 101 1 1.333 0.176
Davilla elliptica A. St.-Hil. Dilleniaceae 90 59 0.128 0.101
Sclerolobium paniculatum Vogel Fabaceae Caesalpinioideae 81 8 0.228 0.071
Connarus suberosus Planch. Connaraceae 80 11 0.177 -0.026
Byrsonima coccolobifolia Kunth Malpighiaceae 69 16 0.112 0.013
Lafoensia vandelliana Cham. & Schltdl. Lythraceae 67 -12 0.210 -0.029
Pouteria ramiflora (Mart.) Radlk. Sapotaceae 64 1 0.158 0.036
Qualea parviflora Mart. Vochysiaceae 55 45 0.420 0.177
Syagrus comosa (Mart.) Mart. Arecaceae 55 5 0.168 0.008
Eschweilera nana (O. Berg) Miers Lecythidaceae 45 16 0.070 0.052
Tabebuia ochracea (Cham.) Standl. Bignoniaceae 41 6 0.108 0.021
Psidium laruotteanum Cambess. Myrtaceae 39 0 0.159 0.029
Stryphnodendron rotundifolium Fabaceae Mimosoideae 39 -11 0.042 -0.010
Mart. ex Benth.
Erythroxylum suberosumA. St.-Hil. Erythroxylaceae 33 -17 0.031 -0.012
Pouteria torta (Mart.) Radlk. Sapotaceae 30 17 0.049 0.026
Hymenaea stigonocarpa Mart. ex Hayne Fabaceae Caesalpinioideae 28 8 0.047 0.014
Others 36 species in 1995 – 350 151 1.134 0.382
density and basal area over the seven years. Lafoensia
vandelliana decreased in density and basal area, while
Psidium laruotteanum Cambess. (=Psidium
warmingianum Kiaersk.) maintained the same density
over the seven years. Connarus suberosus increased
in density and presented negative difference in basal
area due to mortality of extremely ramified individuals,
with high basal area values, while the recruitment
included individuals with smaller diameter, close to the
lower limit established in the methods.
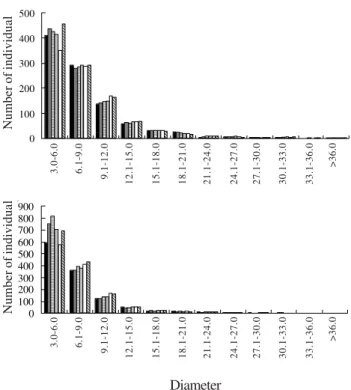
In both fragments, the diameter distribution showed
a reversed J-shape, as could be expected in a
continuously regenerating population (figure 3).
Table 2 shows features of vegetation dynamics in
both fragments. The community of fragment 1 showed
an increase of 7.5% in density and 4.4% in basal area
between 1995 and 2002, while in fragment 2 there was
an increment of 19.4% in density and 23.5% in basal
area, over the same period. In terms of density, annual
recruitment exceeded mortality in both fragments.
Consequently, stand half-lives exceeded doubling times.
In terms of basal area, the mean annual growth rate
exceeded the mean annual mortality rate in fragment 2
and the values of those rates were similar in fragment 1
(table 2). Therefore, stand half-lives exceed doubling
times, in fragment 2. Annual recruitment and mortality
varied widely depending on the period analyzed,
particularly in fragment 2 (table 3).
In fragment 1, of the 174 stems that died between
1995 and 2002, 14% belonged to Sclerolobium
paniculatum; 11% to Erythroxylum deciduum; and
10% to Davilla elliptica, while, in fragment 2, 10% of
the 352 dead plants in the same period belonged to
Ouratea hexasperma; 9% to Connarus suberosus;
and 9% to Davilla elliptica. In the 1996 and 1997
assessments, more than 70% of the stems that died
measured from 0.5 to 4 meters in height. Of the 248
recruited plants in fragment 1, 36% belonged to
Sclerolobium paniculatum; 11% to Byrsonima
coccolobifolia and 5% to Byrsonima crassa. Of the
581 recruited plants of fragment 2, 12% belonged to
Davilla elliptica; 12% to Ouratea hexasperma and
7% to Connarus suberosus.
Number of individual
Number of individual
Table 2. Features of dynamics in “cerrado” sensu stricto in
Maranhão, Brazil, during the seven-year study period (1995-2002).
Fragment 1 Fragment 2
Density
Number of plants, 1995 983 1177
Number of dead plants, 1995-2002 174 352 Number of recruits, 1995-2002 248 581
Number of plants, 2002 1057 1406
Mean annual mortality rate 2.73 4.88 (% year-1)
Mean annual recruitment rate 3.25 5.86 (% year-1)
Stand half-life (year) 25 14
Doubling time (year) 22 12
Turnover (year) 23.5 13
Stability (year) 3 2
Basal area
Total basal area, 1995 7.156 5.988
Basal area of dead plants, 1995-2002 1.566 1.030 Basal area increase of surviving 1.704 2.162
plants, 1995-2002
Basal area of recruits, 1995-2002 0.175 0.278
Total basal area, 2002 7.469 7.398
Mean annual mortality rate (% year-1) 3.47 2.66
Mean annual growth rate (% year-1) 3.39 5.00
Stand half-life (year) 20 26
Doubling time (year) 21 14
Turnover (year) 20.5 20
Stability (year) 1 12
Annual increment in diameter 0.13 0.17 (cm year-1)
Figure 3. Diameter distribution in the “cerrado” sensu stricto
of Gerais de Balsas Colonization Project, Maranhão, Brazil. Above: fragment 1; Below: fragment 2. 1995; 1996; 1997;
1998; 2000 and 2002.
Diameter
0
100 200 300 400 500
0
100 200 300 400 500 600 700 800 900
3.0-6.0 6.1-9.0 9.1-12.0
12.1-15.0 15.1-18.0 18.1-21.0 21.1-24.0 24.1-27.0 27.1-30.0 30.1-33.0 33.1-36.0
>36.0
3.0-6.0 6.1-9.0 9.1-12.0
12.1-15.0 15.1-18.0 18.1-21.0 21.1-24.0 24.1-27.0 27.1-30.0 30.1-33.0 33.1-36.0
Discussion
Species such as Hirtella ciliata,
Dimorphandra
gardneriana,
Exellodendron cordatum
(Hook.f.)
Prance,
Myrcia ochroides,
Platonia insignis and
Parkia platycephala Benth. are considered typical
of the north-northeast area of “cerrado” distribution
(Ratter
et al. 1996, 2003), especially in Maranhão State
(Eiten 1994), showing the importance of preserving
areas chosen as private reserves. Although the number
of species has increased in both fragments over seven
years, these species have been represented by only
one or two young individuals, meaning that they
occurred at low density in the plots. Therefore, the
increment in the diversity can be considered transitory,
because any deaths would eliminate these species from
the plots.
The community’s annual increment for fragments
1 and 2, at 0.13 and 0.17 cm per year, respectively, was
similar to that found by Henriques & Hay (2002)
(0.16 cm year
-1) in the Federal District, Brazil, and lower
than that registered by Silberbauer-Gottsberger & Eiten
(1987) (0.27 cm year
-1) for “cerrado” stricto sensu in
São Paulo, Brazil. Rezende (2002) found a higher
increment in diameter, ranging from 0.39 to
0.49 cm year
-1, over a period of eleven years in a
“cerrado”
stricto sensu area in the Federal District,
Brazil, as a consequence of slash and burn. These
studies indicate a low rate of increment in the aerial
parts of plants, which could be associated with poor
soils and long droughts (Henriques & Hay 2002).
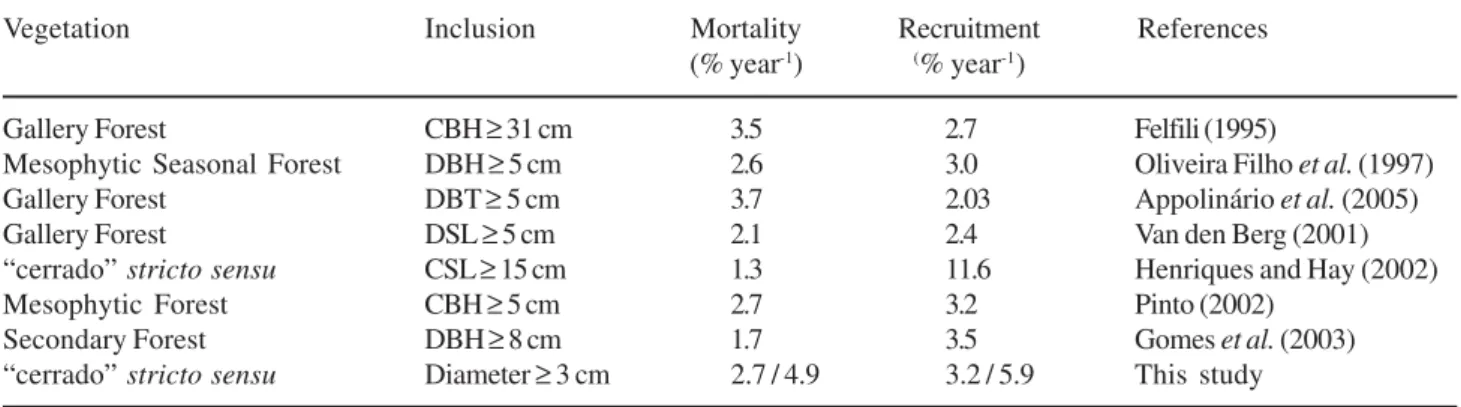
Mortality and recruitment rates registered between
1995 and 2002 were high compared to other studies in
different sites in Brazil (table 4). These high rates may
be associated to human disturbances like fire and
fragmentation of native areas, practiced by farmers in
these areas.
Mortality registered in this study was higher than
the rate obtained by Henriques & Hay (2002), in the
Federal District, Brazil. The high mortality rate recorded
may be related to the frequent burning in the study area.
Fragment 2 was intensively burned in 1998, which
possibly caused the high mortality between 1997 and
1998 (11% year
-1) and high instability. The exclusion of
four species (Casearia sylvestris,
Emmotum nitens,
Guapira graciliflora and
Rourea induta) was
registered in this period. Usually, farmers set fire to
private reserves in an attempt to reduce their size and
use them as supplementary areas for agriculture in the
future; therefore, the natural dynamics of the vegetation
is strongly affected.
Some “cerrado” species may present up to 100%
of mortality after burning (Sato & Miranda 1996,
Hoffmann 1998). Miranda et al. (2003) observed that
frequent burning can modify “cerrado” lato sensu
physiognomies to more open forms, where a larger
number of grassy species appear and favor more intense
and frequent burning. Fire protection favors
fire-sensitive species and increases the abundance of woody
plants in “cerrado” (Moreira 2000).
Additionally, fire may contribute to the high mortality
of smaller individuals, mainly young and juvenile
Table 3. Mortality and recruitment rates in “cerrado” stricto sensu in Maranhão, Brazil, during the seven-year study period(1995-2002), based on density.
Fragment 1
Period Dead Mortality % year-1 Recruits Recruitment % year-1
1995-1996 19 1.93 39 3.97
1996-1997 26 2.49 24 2.29
1997-1998 18 1.80 22 2.20
1998-2000 81 4.17 32 1.63
2000-2002 30 1.53 131 6.58
Fragment 2
1995-1996 31 2.55 187 15.80
1996-1997 20 1.42 139 10.35
1997-1998 144 11.00 30 0.54
1998-2000 135 5.18 69 2.55
individuals of the woody flora. Sato & Miranda (1996)
also observed that individuals with stem diameter
between 5 and 6 cm, measured at 0.30 m above ground
level, and total height under 2 m, presented the highest
mortality rates. At the population level, Hoffmann (1998)
showed that fire can cause high mortality in seedlings of
some woody “cerrado” species, such as Miconia
albicans. Therefore, natural mortality on top of fire
impact can affect the structure and composition of
“cerrado” vegetation.
Recruitment rates obtained here were lower than
those registered by Henriques & Hay (2002) for a
“cerrado”
stricto sensu in Central Brazil (table 4).
However, the recruitment rates found by those authors
and in the present study exceed most of those registered
for tropical forests (Korning & Balslev 1994, Phillips &
Gentry 1994) and other “cerrado” studies shown in table
4, mainly in fragment 2, where there was high
recruitment in the 1995-1997 period. One possible
explanation for the high recruitment in the “cerrado”
stricto sensu compared with other physiognomies may
be the high capacity of vegetative reproduction inherent
to many species.
The highest recruitment rate in relation to mortality
resulted from the increase in density from 1995 to 2002.
However, variations were observed in recruitment and
mortality rates when the intervals of one or two years
were analyzed. In fragment 2, this is evident, as in the
1995-1997 periods the high recruitment rates were
followed by high mortality rates. Some studies suggested
that the dynamic equilibrium is characterized by a period
of high mortality followed by another with high
recruitment and so on, maintaining a constant
physiognomy (e.g.
Felfili 1995).
Felfili
et al. (2000) observed that the changes in
density and basal area were larger than those found in
several tropical forests, and this was attributed to the
frequency of disturbances, such as fire, at intervals from
three to five years. However, the maintenance of the
species’ composition, spatial distribution and community
structure in these areas indicated that the studied
vegetation is still resilient. The same can also be said
for the “cerrado” stricto sensu in the study area, where
there were changes in density and basal area, associated
with disturbances. The communities studied presented
high stability, maintaining the original physiognomy during
this period, at least in terms of density and general woody
species composition. The maintenance of floristic
composition and community structure suggests that the
"cerrado"
stricto sensu was resilient to the disturbances
caused by fire and agriculture at least for the
seven-year period studied.
Acknowledgements – The authors are very grateful to Embrapa Cerrados, Campo (Cia. de Promoção Agrícola), Jica (Japan International Cooperation Agency) and Universidade de Brasília (Pós-graduação em Ecologia). In particular we thank José Roberto Rodrigues Pinto and Ernestino Guarino for valuable comments on the manuscript and Nelson de Oliveira Pais, José Ferreira Paixão, Valdeci de Matos Lima and João Benedito Pereira for assistance in the field. This paper is part of a project funded by DIFD and Embrapa Cerrados.
References
APG II. 2003. An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG II. Botanical Journal of the Linnean Society 141:399-436.
Table 4. Comparison of the mortality and recruitment rates among some communities of Central Brazil. (CBH = Circumference at breast height, CSL = Circumference at soil level, DBH = Diameter at breast height, DBT = Diameter at the base of the trunk and DSL = Diameter at soil level).
Vegetation Inclusion Mortality Recruitment References
(% year-1) (% year-1)
Gallery Forest CBH≥ 31 cm 3.5 2.7 Felfili (1995)
Mesophytic Seasonal Forest DBH≥ 5 cm 2.6 3.0 Oliveira Filho et al. (1997)
Gallery Forest DBT≥ 5 cm 3.7 2.03 Appolinárioet al. (2005)
Gallery Forest DSL≥ 5 cm 2.1 2.4 Van den Berg (2001)
“cerrado”stricto sensu CSL≥ 15 cm 1.3 11.6 Henriques and Hay (2002)
Mesophytic Forest CBH≥ 5 cm 2.7 3.2 Pinto (2002)
Secondary Forest DBH≥ 8 cm 1.7 3.5 Gomeset al. (2003)
APPOLINÁRIO,V., OLIVEIRA FILHO A.T. & GUILHERME, F.A.G. 2005. Tree population and community dynamics in a Brazilian tropical semideciduous forest. Revista Brasileira de Botânica 28:347-360.
CASTRO, A.A.J.F., MARTINS, F.R., TAMASHIRO, J.Y. & SHEPHERD, G.J. 1999. How rich is the flora of Brazilian Cerrados? Annals of the Missouri Botanical Garden 86:192-224.
EITEN, G. 1972. The cerrado vegetation of Brazil. Botanical Review 38:201-341.
EITEN, G. 1994. Duas travessias na vegetação do Maranhão. Universidade de Brasília. Brasília.
FELFILI, J.M. 1995. Growth, recruitment and mortality in the Gama gallery forest in central Brazil over a six-year period (1985-1991). Journal of Tropical Ecology 11:67-83. FELFILI, J.M., HARIDASSAN, M., MENDONÇA, R.C.,
FILGUEIRAS,T.S., SILVA JÚNIOR, M.C. & REZENDE, A.V. 1994. Projeto biogeografia do bioma cerrado: vegetação e solos. Caderno de Geociências 12:75-166. FELFILI, J.M., REZENDE, A.V., SILVA JÚNIOR, M.C. & SILVA,
M.A. 2000. Changes in the floristic composition of cerradosensu stricto in Brazil over a nine-year period.
Journal of Tropical Ecology 16:579-590.
GOMES, E.P.C., MANTOVANI,W. & KAGEYAMA, P.Y. 2003. Mortality and recruitment of trees in a secondary montane rain forest in southeastern Brazil. Brazilian Journal Biology 63:47-60.
HENRIQUES, R.P.B. & HAY, J.D.V. 2002. Patterns and dynamics of plant populations. In The cerrados of Brazil:
ecology and natural history of a neotropical savanna (P.S. Oliveira & R. J. Marquis, eds.). Columbia University Press. New York, p.140-158.
HOFFMANN, W. 1998. Post-burn reproduction of woody plants in a neotropical savanna: the relative importance of sexual and vegetative reproduction. Journal Applied Ecology 35:422-433.
KLINK, C.A., MOREIRA, A.G. & SOLBRIG, O.T. 1993. Ecological impact of agricultural development in the cerrados.InThe world’s savannas: economic driving
forces, ecological constraints and policy options for sustainable land use (M.D. Young & O.T. Solbrig, eds.). Paris and Carnforth: UNESCO & Parthenon Press. p.259-282.
KORNING, J. & BALSLEV, H. 1994. Growth and mortality of trees in Amazonian tropical rain forest in Ecuador. Journal of Vegetation Science 4:77-86.
LIEBERMAN, D., LIEBERMAN, M., PERALTA, P. & HARTSHORN, G.S. 1985. Mortality patterns and stand turnover rates in a wet tropical forest in Costa Rica. Journal of Ecology 73:915-924.
MENDONÇA, R.C., FELFILI, J.M., SILVA JÚNIOR, M.C., FAGG, C.W., SILVA, M.A., FILGUEIRAS, T.S. & WALTER, B.M.T. 2000. Florística da região do Espigão Mestre do São Francisco, Bahia e Minas Gerais. Boletim do Herbário Ezechias Paulo Heringer 6:38-94.
MIRANDA, H.S., SATO, M.N., ANDRADE, S.M.A., HARIDASAN, M. & MORAIS, H.C. 2003. Queimadas de cerrado: caracterização e impactos. In Cerrados:
ecologia e caracterização (L.M.S. Aguiar & A.J.A. Camargo, eds.). Embrapa Cerrados. Planaltina, DF. p.69-123.
MITTERMEIER, R.A., MYERS, N., GIL, P.R. & MITTERMEIER, C.G. 1999. Hotspots: Earth’s biologically richest and most endangered terrestrial ecoregions. CEMEX, S. A.Agrupación Sierra Madre, S. C. Cidade do México.
MOREIRA, A.G. 2000. Effects of fire protection on savanna structure in Central Brazil. Journal of Biogeography 27:1021-1029.
MYERS, N., MITTERMEIER, R.A., MITTERMEIER, C.G., FONSECA, G.A.B. & KENT, J. 2000. Biodiversity hotspots for conservation priorities. Nature 403:853-858. OLIVEIRA FILHO, A.T. & RATTER, J.A. 2002. Vegetation physiognomies and woody flora of the cerrado biome.In The cerrados of Brazil: ecology and natural
history of a neotropical savanna (P.S. Oliveira & R.J. Marquis, eds.). Columbia University Press. New York. p.91-120.
OLIVEIRA FILHO, A.T., MELLO, J.M. & SCOLFORO, J.R.S. 1997. Effects of past disturbance and edges on tree community structure and dynamics within a fragment of tropical semideciduous forest in south-eastern Brazil over a five-year period (1987-1992). Plant Ecology 131:45-66.
PHILLIPS, O.L. & GENTRY,A.H. 1994. Increasing turnover through time in tropical forests. Science 263:954-958. PINTO, J.R.R. 2002. Dinâmica da comunidade
arbóreo-arbustiva em uma floresta de vale no Parque Nacional da Chapada dos Guimarães, Mato Grosso. Tese de doutorado, Universidade de Brasília, Distrito Federal.
RATTER, J.A. & DARGIE, T.C.D. 1992. An analysis of the floristic composition of 26 cerrado areas in Brazil. Edinburgh Journal of Botany 53:153-180.
RATTER, J.A., BRIDGEWATER, S., ATKINSON, R. & RIBEIRO, J.F. 1996. Analysis of the floristic composition of the Brazilian cerrado vegetation of 98 areas. Edinburgh Journal of Botany 53:153-180.
RATTER, J.A., RIBEIRO, J.F. & BRIDGEWATER, S. 1997. The Brazilian cerrado vegetation and threats to its biodiversity.Annals of Botany 80:223-230.
RATTER, J.A., BRIDGEWATER, S. & RIBEIRO, J.F. 2003. Analysis of floristic composition of the Brazilian cerrado vegetation III: comparison of the woody vegetation of 376 areas. Edinburgh Journal of Botany 60:57-109. REZENDE, A.V. 2002. Diversidade, estrutura, dinâmica e
RIBEIRO, J.F. & WALTER, B.M.T. 1998. Fitofisionomias do bioma cerrado. In Cerrado: ambiente e flora (S.M. Sano
& S.P.Almeida, eds.). Embrapa Cerrados, Planaltina, DF. p.89-166.
SATO, M.N. & MIRANDA, H.S. 1996. Mortalidade de plantas lenhosas do cerrado após duas queimadas prescritas. In
Simpósio sobre o cerrado: Biodiversidade e produção sustentável de alimentos e fibras nos cerrados (R.C. Pereira & L.C.B. Nasser, eds.). Embrapa Cerrados. Planaltina, DF. p.204-207
SHEIL, D., BURSLEM, D.F.R.P. & ALDER, D. 1995. The interpretation of mortality rates measures. Journal of Ecology 83:331-333.
SILBERBAUER-GOTTSBERGER, I. & EITEN, G. 1987. A hectare of cerrado: I. General aspects of the trees and thick-stemmed shrubs. Phyton 27:55-91.