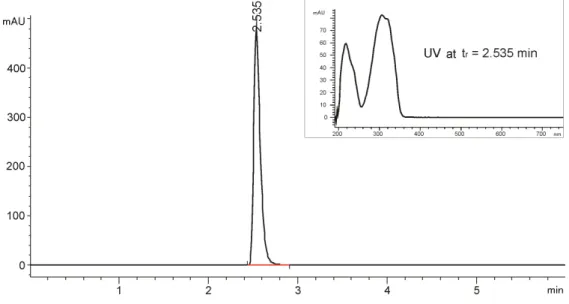
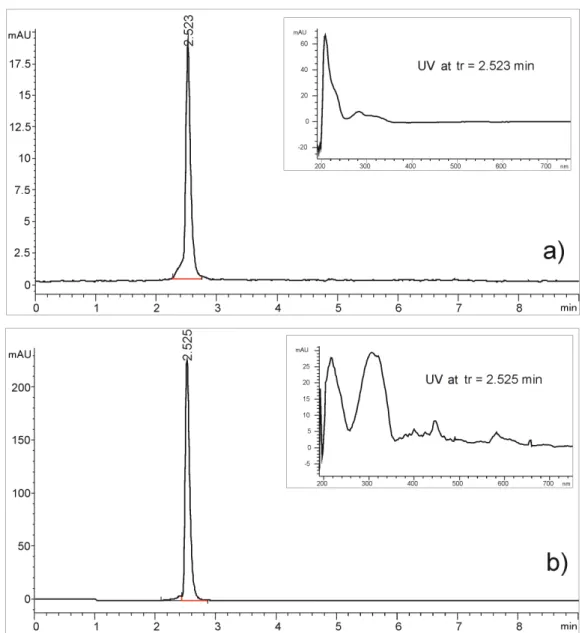
Development and validation of an RP-HPLC method for quantification of trans-resveratrol in the plant extracts
Texto
Imagem
Documentos relacionados
An isocratic reversed phase high-performance liquid chromatographic (RP-HPLC) method has been developed for the simultaneous determination of gemiloxacin and
This study presents the development and validation of a specific analytical method using high-performance liquid chromatography (HPLC) for quantification of the xanthyletin content
The present study describes the development and subsequent validation of simple and accurate stability indicating RP-HPLC method for the determination of sparloxacin and
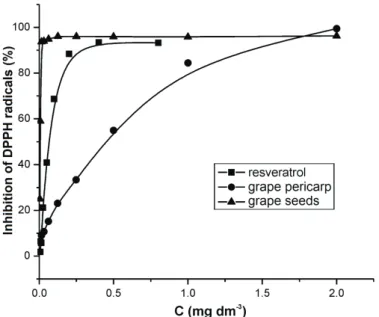
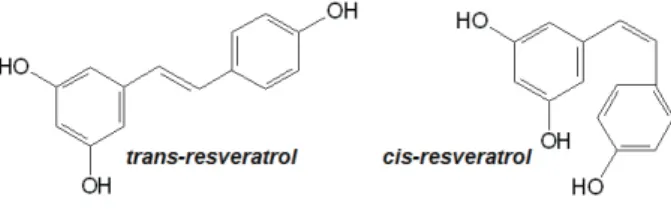
The results achieved with Salvia officinalis extract motivated us to develop and validate an analytical RP-HPLC method to detect and determine manool in this extract.. The
Commercial capsule formulation was successfully analyzed using the developed method and the proposed method is applicable to routine analysis of determination of
Preparation of stock solution: The Stock Solution has been prepared by weighing accurately and transferred 10 mg of Prasugral Working standard into a 10 mL volumetric
The following research was done in order to develop a simple, precise and accurate method for analysis of Fluconazole and Ivermectin in bulk and tablet
The analysis of the content in albendazole in suspensions of Rombendazole has been carried out under the developed method recommended for validation, using the