Eclet. Quím. vol.35 número1
Texto
Imagem
Documentos relacionados
The methods under study differed mainly in the mass of the sample, solvent, time, and temperature used for the extraction of γ -oryzanol and the separation of impurities
The main aim of the study was to prepare GSE by solvent – solvent extraction method, identifying various phytochemical compounds present in the extract, examining
Conditions: sonication time, 15 min; type of extraction solvent in UAE stage and its volume, acetonitrile, 5 mL, respectively; sample amount, 1.00 g; extraction solvent in
The sewage treatment station achieved a significant efficiency index in the elimination of fecal coliforms, and in the rainy period there was a reduction of 99.8% and
Here, we consider a general regenerative process that includes Smith’s model and show that it is important to consider finite time quantities instead of asymptotic ones and
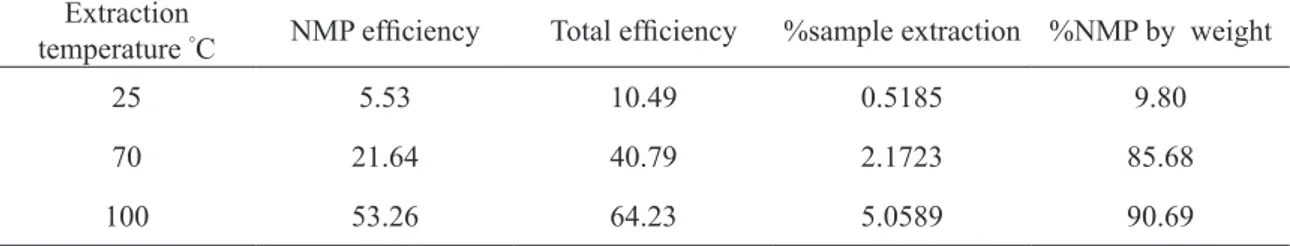
SEPARATION OF ZINC FROM MANGANESE BY SOLVENT EXTRACTION FROM ACIDIC LEACHATES OF SPENT ZINC-MnO 2 DRY CELLS USING NEUTRAL ORGANOPHOSPHORUS EXTRACTANTS.. Vinício Francisco Ibiapina a
Network simulation module 157 The first steps consist in determine the efficiency value computed by EPANET for each variable- speed pump in all periods of time (considering