REPOSITORIO INSTITUCIONAL DA UFOP: Kinetics of chalcopyrite leaching in either ferric sulphate or cupric sulphate media in the presence of NaCl.
Texto
Imagem

Documentos relacionados
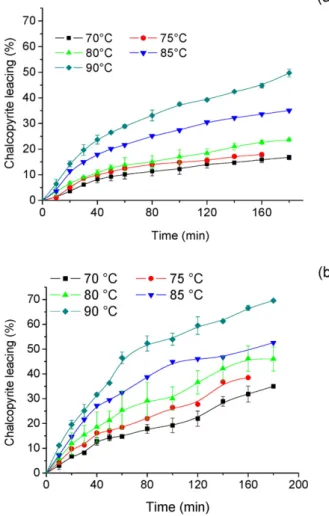
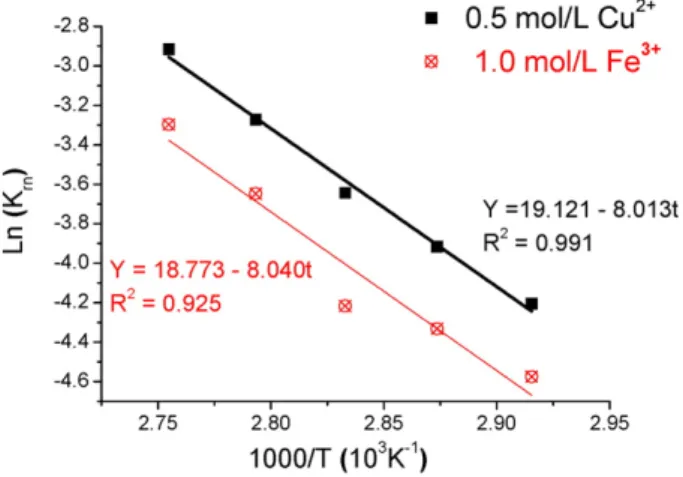
The Shrinking Core Model (SCM) with chemical reaction control and product layer diffusion control as well as the initial rate (IR) method were used to describe the dissolution
The effects of chloride ions on chalcopyrite leaching can be summarized as follows: chloride ions affect the chemistry of leaching, enabling chalcopyrite dissolution by the
Therefore, it is proposed that the zinc dissolution process be controlled by diffusion of the reagent in the porous structure of the calcine particles, as observed by Georgiou
Embora a extração de cobre tenha elevado-se de 16,4% na ausência para 29,4% na presença de 1mol/L de NaCl, a alteração na concentração de cloreto de sódio teve um
Firstly, the effects of temperature, ferric ion and sulphuric acid concentrations, agitation speed and particle size on the leaching kinetics of a zinc sulphide
he teeth were immersed in solutions of 0.1% thymol, 0.02% sodium azide and distilled water (control) (solutions previously prepared by the biochemical laboratory of BSD-USP)
The effect of drinking water acidification by lactic acid or citric acid or a combination of those with cupric sulfate and d- limonene in the reduction of Salmonella Enteritidis
As preparation for the procedure, patient was advised to intake 2 bottles of Coloprep TM composed of sodium sulphate 17.5 g, potassium sulphate 1.6 g and magnesium sulphate 3.13