Article
ISSN 0102-695X doi: 10.1590/S0102-695X2011005000069 Received 15 Dec 2010 Accepted 5 Jan 2011 Available online 22 Apr 2011
Laurencia complex seaweed species
(Ceramiales, Rhodophyta) harvested from
the Atlantic Ocean with emphasis on the
Brazilian State of Espírito Santo
Erika M. Stein,
1,2Daniel X. Andreguetti,
1Cleidiane S. Rocha,
1Mutue T. Fujii,
2,3Mauricio S. Baptista,
1Pio Colepicolo,
1Guilherme L. Indig
*,41Departamento de Bioquímica, Instituto de Química, Universidade de São Paulo, Brazil,
2Departamento de Botânica, Instituto de Biociências, Departamento de Botânica, Universidade de São Paulo, Brazil,
3Instituto de Botânica, Secretaria do Meio Ambiente, Brazil,
4Department of Chemistry and Biochemistry, University of Wisconsin, USA.
Abstract:The development of new anti-cancer drugs of algal origin represents one of the least explored frontiers in medicinal chemistry. In this regard, the diversity of micro- and macroalgae found in Brazilian coastal waters can be viewed as a largely untapped natural resource. In this report, we describe a comparative study on the cytotoxic properties of extracts obtained from the Laurencia complex: Laurencia aldingensis, L. catarinensis, L. dendroidea, L. intricata, L. translucida, L. sp, and Palisada flagellifera. All of these species were collected in the coastal waters of the State of Espírito Santo, Brazil. Four out of the twelve samples initially investigated were found to show significant levels of toxicity towards a model tumor cell line (human uterine sarcoma, MES-SA). The highest levels of cytotoxicity were typically associated with non-polar (hexane) algal extracts, while the lowest levels of cytotoxicity were found with the corresponding polar (methanol) extracts. In this report, we also describe a biological model currently in development that will not only facilitate the search for new anti-cancer drug candidates of algal origin, but also permit the identification of compounds capable of inducing the destruction of multi-drug resistant tumors with greater efficiency than the pharmaceuticals currently in clinical use.
Keywords:
macroalgae Laurencia complex anti-cancer activity MES-SA MES-SA/Dx5 Hela
Introduction
Out of the more than 25,000 known species
of algae, only about i fteen are currently grown on a
large-scale for biotechnological applications (Raja et al., 2008). Algae are considered to be a largely untapped natural resource and investigations involving them are one of the least explored frontiers of biotechnology and natural products chemistry (Pinto et al. 2000; Cardozo
et al., 2007; Chakabourty et al., 2009). Signii cant
advances in the discovery and development of new anti-cancer drugs of algal origin have been recently reported
in the literature. Newly identii ed algal drug candidates
include representatives of the terpene, steroid, and polyketide families (Mayer & Gustafson, 2008).
Drug-induced over-expression of the multi-drug efl ux pump
P-glycoprotein (P-gp) is thought to be one of the leading causes of chemotherapy failure in clinical oncology (Higgins, 2007; Dawson & Locker, 2006; Lage, 2008; Perez-Thomas, 2006). Because P-gp can remove many unrelated chemotherapeutic agents from the target cells, including agents to which the tumor had not been previously exposed (Higgins, 2007; Dawson & Locker, 2006), these cells are referred to as multi-drug resistant (MDR) mutants.
One of the most gratifying i ndings that can arise from any drug screening program is the identii cation of
new drug candidates that are capable of eliminating MDR
tumor cells more efi ciently than the drugs currently in
clinical use. The discovery of new, more benign inhibitors
of such drug efl ux pumps can also be facilitated by
laboratories.
In this report, we describe our initial results on the search for novel anti-cancer agents in marine algae found on the coast of the Brazilian State of Espírito Santo. We also outline a biological model currently in development to guide our screening efforts. This model
aids in the identiication of potential new anti-cancer
candidates and, at the same time, of those that should be less susceptible to the action of promiscuous
therapy-induced drug-eflux pumps such as P-gp.
Materials and Methods
Reagents and cell culture media
McCoy’s 5A modiied medium, Dulbecco’s modiied Eagle’s medium (DMEM), doxorubicin
hydrochloride, fungizone (amphotericin B), penicillin, streptomycin, thiazolyl blue tetrazolium bromide (MTT), ethylenediamine tetraacetic acid (EDTA; disodium salt), trypsin, and Trypan Blue were obtained from Sigma-Aldrich (St. Louis, USA). Fetal bovine serum (FBS) from Vitrocell Embriolife (Campinas, Brazil). NaCl from Synth (Diadema, Brazil), sodium bicarbonate from Merck (Darmstadt, Germany), D-glucose from Becto Chemical (São Paulo, Brazil), KCl from Cetus (São Paulo, Brazil), Dulbecco’s phosphate buffered saline (DPBS) from Gibco BRL (Grand Island, USA), and dimethylsulfoxide (DMSO) from Vetec Química Fina Ltda. (Duque de Caxias, Brazil) were all of high-purity grade and used
as received. Water was distilled, deionized and iltered prior to use (Millipore Milli-Q system; resistivity, 18 MΩ
cm).
Instruments and methods
Stock solutions of doxorubicin were prepared in water and their respective concentrations determined by absorption spectroscopy on a Shimadzu (Kyoto, Japan) Model UV-1650 PC spectrophotometer employing a
molar extinction coeficient of 11,500 M-1 cm-1 at 480 nm (Zeman et al., 1998).
Algal extracts were prepared via initial sonication (30 min) of approximately 30 g of the respective powdered and dried biomass with 300 mL of hexane. After standing overnight, the mixture was
iltered and the solid mass subjected to two more cycles
of extraction with hexane. The hexane-extracted biomass was then further processed by performing three extraction cycles with chloroform followed by three cycles of methanol extraction. The solvent was evaporated from
the iltrates on a rotary evaporator to obtain three distinct
classes of algal extracts, namely the hexane extract, the chloroform extract, and the methanol extract. In selected experiments, we extracted the original algal dry powder
sample only with methanol; these extracts are referred to here as “crude” methanol extracts. Stock solutions of
the inal algal extracts were prepared in either DMSO
or ethanol and subsequently diluted in the cell growth
media in order to maintain the inal concentration of
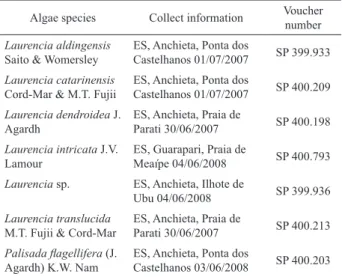
organic solvent in the medium below 0.5% (by volume). The species of algae investigated are listed in Table 1.
Table 1. Species of Laurencia complex seaweed investigated in this study. Voucher specimens were deposited in the Herbarium Maria Eneida P. Kauffman Fidalgo at the Instituto de Botânica, São Paulo, SP, Brazil.
Algae species Collect information Voucher number
Laurencia aldingensis
Saito & Womersley
ES, Anchieta, Ponta dos
Castelhanos 01/07/2007 SP 399.933
Laurencia catarinensis
Cord-Mar & M.T. Fujii
ES, Anchieta, Ponta dos
Castelhanos 01/07/2007 SP 400.209
Laurencia dendroidea J. Agardh
ES, Anchieta, Praia de
Parati 30/06/2007 SP 400.198
Laurencia intricata J.V. Lamour
ES, Guarapari, Praia de
Meaípe 04/06/2008 SP 400.793
Laurencia sp. ES, Anchieta, Ilhote de
Ubu 04/06/2008 SP 399.936
Laurencia translucida
M.T. Fujii & Cord-Mar
ES, Anchieta, Praia de
Parati 30/06/2007 SP 400.213 Palisada lagellifera (J.
Agardh) K.W. Nam
ES, Anchieta, Ponta dos
Castelhanos 03/06/2008 SP 400.203
The trypan blue dye exclusion assay (Perry et al., 1997) was routinely employed to assess the quality of our original cell cultures; in all experiments performed with cell cultures, cell viability was at least 95%. All experiments were carried out with cells in the exponential growth phase. Cell viability assays were carried out using the MTT assay (Mosmann, 1983; Garcia-Peres et al.,
2010). For this purpose, 96-well lat-bottomed microtiter
plates were seeded at a cell density of 5,000 cells per well (100 µL of growth medium per well) and the cells allowed to attach and grow for 24 h. The sub-culturing protocols used in this study were always in accord with the respective ATCC recommendations. The cells were subsequently exposed to growth medium containing either the standard drug (doxorubicin) or the algal extracts for a period of time that typically varied from 24 to 72 h prior to MTT analysis. The MTT assays were performed on a
Model Ininite M200 multi-well plate reader from Tecan
(Männedorf, Switzerland). Cells exposed to the growth medium alone or to the growth medium plus the drug/ extract vehicle (DMSO or ethanol) served as the controls.
Under our experimental conditions, no signiicant
Cell Lines
The MES-SA human uterine sarcoma cell line (ATCC CRL-1976™) and the corresponding doxorubicin-resistant mutant (MES-SA/Dx5, ATCC CRL-1977™) were obtained from the American Type Culture Collection (ATCC; Manassas, VA). These cells were
grown in modiied McCoy’s 5A medium supplemented
with 10% FBS, amphotericin B (0.63 µg/mL), penicillin (100 units/mL) and streptomycin (100 µg/mL). The medium used to grow the MES-SA/Dx5 mutants also contained doxorubicin (0.5 µM), as required for the maintenance of the drug-resistance phenotype (Harker, 1983; Harker & Sikic, 1985; Larroque-Lombard et al., 2010). HeLa (CCL-2™) human cervix adenocarcinoma cells were kindly provided by Dr. Mari Cleide Sogayar (Departamento de Bioquímica, Universidade de São Paulo) and grown in DMEM medium, supplemented as described above for the McCoy’s medium used to grow the MES-SA cells. Cells were grown at 37 oC in a
humidiied atmosphere of 5% CO2 in air.
Results and Discussion
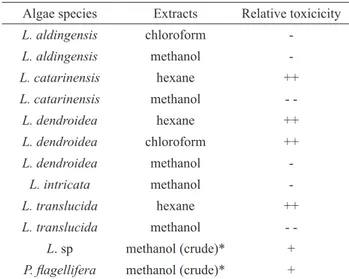
In this initial screening, we explored the extent to which the chemical agents extracted from a variety of Laurencia complex seaweed species exhibited cytotoxicity effects toward our primary model tumor cell line (MES-SA). For this purpose, we initially exposed cultures of MES-SA cells to mixtures containing substantial amounts of the algal extracts (i.e., 500 µg of algal extract/mL of cell culture media), subsequently measuring the cell survival rates after 24 h of continuous exposure to the extract. Table 2 shows that substantial cytotoxicity was associated with some, but not all, of these algal extracts. The methanol extracts obtained by our sequential extraction protocol (i.e., hexane/chloroform/methanol) showed the lowest levels of toxicity, even lower than that of the crude methanol extracts obtained via direct methanol extraction of the original algae dry powder samples. The somewhat higher toxicity of the crude methanol extracts relative to the sequential methanol extracts is presumably the result of the presence of less polar chemical entities in the former that are absent in the latter extract because of removal during the prior extractions with the less polar solvents. Indeed, the hexane extracts of Laurencia catarinensis Cord.-Mar. & M.T. Fujii, L. dendroidea J. Agardh, and L. translucida M.T. Fujii & Cord.-Mar all show high levels of cytotoxicity. Likewise, the chloroform extract of L. dendroidea also exhibits a high degree of cytotoxicity, but the corresponding extract of L. aldingensis does not (Table 2).
Table 2. Relative cytotoxic effects of algal extracts toward MES-SA cells.
Algae species Extracts Relative toxicicity
L. aldingensis chloroform
-L. aldingensis methanol
-L. catarinensis hexane ++
L. catarinensis methanol
-L. dendroidea hexane ++
L. dendroidea chloroform ++
L. dendroidea methanol
-L. intricata methanol
-L. translucida hexane ++
L. translucida methanol
-L. sp methanol (crude)* +
P. lagellifera methanol (crude)* +
Key: (++) ≥ 80% cell mortality; (+) 50-79% cell mortality; (-) 30-50% cell mortality; (--) <30% cell mortality. Cells were incubated for 24 h in the presence of algal extracts (500 µg/mL of growth medium).* See Materials and Methods.
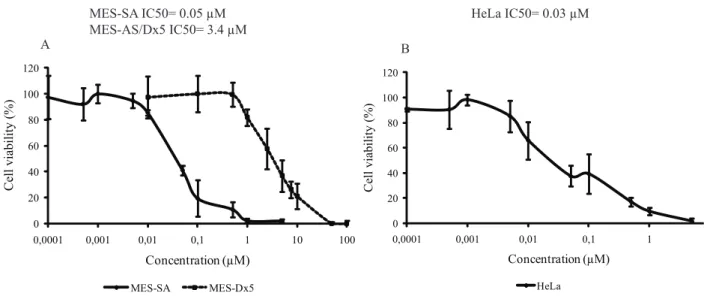
The overall trends observed in Table 2 were further confirmed by a subsequent experiment employing the highly cytotoxic hexane extracts of L. dendroidea and L. translucida and the methanol extract of L. translucida. In this second study, MES-SA cells were exposed for 72 h to variable concentrations of the extracts and cell viability again measured by the MTT assay. Figure 1 confirms that the methanol extract of L. translucida is virtually non-toxic, while the corresponding hexane extract of the same alga is highly toxic, with an apparent IC50 of about 16 µg/ mL. The IC50 associated with the hexane extract of L. dendroidea was ca. 5-fold higher (i.e., 91 µg/mL) than that of the hexane extract of L. translucida. For comparative purposes, an analogous experiment carried out with doxorubicin (a classical and potent anti-cancer drug) gave an ID50 of 0.05 µM (i.e., 0.03 µg/mL) for MES-SA cells (Figure 2).
potency associated with putative new drug candidates with that of previous candidates studied with HeLa cells. On the other hand, the MES-SA line has a feature that is quite attractive in terms of the development of novel screening protocols: the existence of a commercially-available MES-SA mutant (namely the MES-MES-SA/Dx5 line) that is also easy to work with and maintain and shows the classical multi-drug resistance phenotype (i.e., over-expression of
the drug eflux pump, P-gp). Therefore, the inclusion of
this mutant cell line in any screening protocol permits the
identiication not only of novel anti-cancer agents capable
of inducing the mortality of MDR tumor cells, but also of novel inhibitors of P-gp. For comparative purposes, the ID50 for doxorubicin was 0.05 µM for the wild type cell
line (MES-SA), but almost two orders of magnitude higher (i.e., 3.4 µM; see Figure 2, panel A) for the MDR mutant (MES-SA/Dx5). Analogous differences in the response of these cell lines to doxorubicin have been reported by others authors (Larroque-Lombard et al., 2010).
Conclusions
Our initial studies indicate that four of the twelve extracts of the Laurencia complex seaweeds currently under investigation exhibit significant cytotoxicity towards MES-SA cells. We are cautiously
optimistic with respect to these preliminary indings
and further investigations designed to isolate and identify
Figure 1. Cytotoxic effects of algal extracts toward human uterine sarcoma (MES-SA) cells. From the top, at 100 µg extract/ml of growth medium: Lt-ME, methanol extract of Laurencia translucida; Ld-HE, hexane extract of Laurencia dendroidea; Lt-HE, hexane extract of Laurencia translucida. The cell cultures were incubated for 72 h in the presence of the algal extracts.
-20 0 20 40 60 80 100 120
0 50 100 150 200
Cell viability (%)
Extracts concentration (µg/mL)
Ld-HE Lt-HE Lt-ME
Figure 2. Cytotoxic effects of doxorubicin on the three tumor cell lines. Panel A: human uterine sarcoma (MES-SA) and its corresponding multi-drug resistant mutant (MES-SA/Dx5). Panel B: human cervix adenocarcinoma (HeLa) cells.
0 20 40 60 80 100 120
0,0001 0,001 0,01 0,1 1 10 100
Cell viability (%)
Concentration (µM)
MES-SA MES-Dx5
0 20 40 60 80 100 120
0,0001 0,001 0,01 0,1 1 10
Cell viability (%)
Concentration (µM)
HeLa MES-SA IC50= 0.05 µM
MES-AS/Dx5 IC50= 3.4 µM
HeLa IC50= 0.03 µM
A B
---- Ld-HE: IC50= 91±13 µg/mL
____
novel and potent anti-cancer drug candidates from these extracts are already in progress. The biological model under development here is currently being used for a wider systematic screening of algal species present in waters along the Brazilian seashore. This should expand our knowledge of the presence of cytotoxic compounds in Brazilian algal species and may identify novel candidate compounds for the more effective treatment of multi-drug resistant tumors (Higgins, 2007; Dawson & Locker, 2006; Lage, 2008; Perez-Tomas, 2006). In addition, we envision that our biological model should also facilitate the search for novel chemical inhibitors of P-gp that show positive synergism with classical anti-cancer drugs. Thus, any enhancement in MES-SA/Dx5 cell mortality (e.g., at ID20) in the presence of doxorubicin plus a non-toxic extract relative to that of doxorubicin alone would suggest the possible presence of P-gp inhibitors or other agents (e.g., pro-apoptotic factors) in the extract. Hence, in our future screening studies, even algal extracts that do not show cytotoxic properties will be tested for synergy with the standard drug doxorubicin.
Acknowledgment
This work was supported by the Brazilian research funding agencies Conselho Nacional de
Desenvolvimento Cientíico e Tecnológico, Fundação
de Amparo a Pesquisa do Estado de São Paulo and
Coordenação de Aperfeiçoamento de Pessoal de Nível
Superior, Ministério da Saúde, Ministério de Ciência e Tecnologia and CNPq-INCT-Redoxoma.
References
Belostotsky I, da Silva SM, Paez MG, Indig GL 2011. Mitochondrial targeting for photochemotherapy. can selective tumor cell kill be predicted based on n-octanol/
water distribution coeficients? Biotech Histochem, doi :10.3109/10520295.2010.483656.
Cardozo KHM, Guaratini T, Barros MP, Falcao VR, Tonon AP, Lopes NP, Campos S, Torres MA, Souza AO, Colepicolo P, Pinto E 2007. Metabolites from algae with economical impact. Comp Biochem Physiol C-Toxicol Pharmacol 146: 60-78.
Chakabourty C, Hsu CH, Wen ZH, Lin CS 2009. Anticancer drug discovery and development from marine organisms. Curr Top Med Chem 9: 1536-1545.
Dawson RJP, Locker KP 2006. Structure of a multidrug ABC transporter. Nature 443: 180-185.
García-Pérez M, Royer M, Duque-Fernandez A, Diouf PN, Stevanovic T, Pouliot R 2010. Antioxidant, toxicological and antiproliferative properties of Canadian polyphenolic extracts on normal and psoriatic keratinocytes. J Ethnopharmacol 132: 251-258. Harker WG 1983. Development and characterization of a human
sarcoma cell line, MES-SA, sensitive to multiple drugs. Cancer Res 43: 4943-4950.
Harker WG, Sikic BI 1985. Multidrug (Pleiotropic) resistance in doxorubicin-select variants of the human sarcoma cell line MES-SA. Cancer Res 45: 4091-4096
Higgins CF 2007. Multiple molecular mechanisms for multidrug resistance transporters. Nature 446: 749-757.
Lacerda SHD, Abraham B, Stringfellow TC, Indig GL 2005. Photophysical, photochemical, and tumor-selectivity properties of bromine derivatives of triarylmethanes and rhodamine-123. Photochem Photobiol 81: 1430-1438.
Lage H 2008. An overview of cancer multidrug resistance: a still unsolved problem. Cell Mol Life Sci 65: 3145-3167. Larroque-Lombard AL, Todorova M, Golabi N, Willians C,
Jean-Claude BJ 2010. Synthesis and uptake of luorescence-labeled combi-molecules by P-glycoprotein-proicient and -deicient uterine sarcoma cells SA and
MES-SA/Dx5. J Med Chem 53: 2104-2113.
Mayer AMS, Gustafson KR 2008. Marine Pharmacology: Antitumor and cytotoxic compounds. Eur J Can 44: 2357-2387.
Mosmann T 1983. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. Immunol Meth 65: 55-64.
Perez-Tomas R 2006. Multidrug resistance. Retrospect and prospects in anti-cancer treatment. Cur Med Chem 13: 1859-1876.
Perry SW, Epstein LG, Gelbart HA 1997. Simultaneous in situ detection of apoptosis and necrosis in monolayer cultures by TUNEL and trypan blue staining. Biotechniques 22: 1102-1106.
Pinto E, Catalani LH, Lopes NP, Di Mascio, P, Colepicolo P 2000. Isolation of peridinin from chloroplasts of Gonyaulax polyedra. Biochem Biophys Res Comm 268: 496-500.
Raja R, Hemeaiswarya S, Kumar NA, Shidhar S, Rengasamy R 2008. A perspective on the biotechnological potential of algae. Crit Rew Microbiol 34: 77-88.
Zeman MS, Phillips DR, Crothers DM 1998. Characterization of covalent Adiamycin-DNA adducts. Proc Nat Acad Sci USA 95: 11561-11565.
*Correspondence
Erika M. Stein
Departamento de Bioquímica, Instituto de Química, Universidade de São Paulo
Av. Prof. Lineu Prestes, 748, Bloco 03 superior, Cidade Universitária, 05508-900 São Paulo-SP, Brazil