w ww.e l s e v i e r . c o m / l o c a t e / b j p
Original
Article
Growth
inhibition
of
human
breast
cancer
cells
and
down-regulation
of
ODC1
and
ADA
genes
by
Nepeta
binaloudensis
Akbar
Safipour
Afshar
∗,
Fatemeh
Saeid
Nematpour,
Mahshid
Meshkani,
Arezosadat
Khafi
DepartmentofBiology,NeyshaburBranch,IslamicAzadUniversity,Neyshabur,Iran
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received10February2016 Accepted8July2016
Availableonline15September2016
Keywords:
Adenosinedeaminase Anticancer Cytotoxicity
Ornithinedecarboxylase
a
b
s
t
r
a
c
t
NepetabinaloudensisJamzad,Lamiaceae,isararemedicinalplantendemictoIran.Inspiteofmanystudies
aboutthechemicalconstituentsandantibacterialeffectsofthisspecies,noreporthasbeenprovidedabout
itscytotoxicandanticanceractivities.InthisstudywehaveevaluatedtheeffectsofEtOH70%,hexane
andaqueousextractsofN.binaloudensisonthecellproliferationandn-hexaneextractontheexpression
ofadenosinedeaminaseandornithinedecarboxylase1genesinbreastcancercelllines(MCF-7,
MDA-MB-231)comparedtonon-cancerline(MCF-10A).Thecelllinesweresubjectedtoincreasingdoses
oftheextractsrangingfrom10to320g/ml.CellviabilitywasquantifiedbyMTSassay.Expressionof
adenosinedeaminaseandornithinedecarboxylase1geneswasanalyzedbyrealtimePCR.N.binaloudensis
inhibitedthegrowthofmalignantcellsinatimeanddose-dependentmanner.AmongextractsofN.
binaloudensis,thehexaneextractwasfoundtobemoretoxiccomparedtootherextracts.Resultsshowed
amarkeddecreaseintheexpressionofornithinedecarboxylase1andadenosinedeaminasegenesin
cancercelllines.At60g/mlconcentrationofN.binaloudensishexaneextractornithinedecarboxylase
1andadenosinedeaminasemRNAexpressionwerereduced4.9foldand3.5foldinMCF-7celllineand
3.6foldand2.6foldinMDA-MB-231celllinecomparedtocontrol,respectively.Theresultofourstudy
highlightsthepotentialinfluencesofN.binaloudensishexaneextractonornithinedecarboxylase1and
adenosinedeaminasegenesexpressioninbreastcancercellsanditsrelationtoinhibitionofcancercell
growth.
©2016SociedadeBrasileiradeFarmacognosia.PublishedbyElsevierEditoraLtda.Thisisanopen
accessarticleundertheCCBY-NC-NDlicense(http://creativecommons.org/licenses/by-nc-nd/4.0/).
Introduction
Canceristhemostcommoncauseofdeathintheworld.The majorityofdeathsfromcanceroccurinwomenwithbreastcancer. Statisticsindicatethatin2014about29%ofcancerswerebreast cancersamongAmericanwomen(Siegeletal.,2014).InIranalso breastcancerrateshaveincreasedinfemales(Mousavietal.,2009). Manyinvestigationsareperformedtodetectthegeneswhichare involved in breast cancer. One of themost important genes is ornithinedecarboxylase(ODC)which encodesa keyenzyme in polyaminessynthesis(Zhuetal.,2012;Xuetal.,2015).Several studiesindicatethatthereareclosecorrelationbetweenODC activ-ityinthecellsandregulationofcellproliferation(Dengetal.,2008;
Elmetsand Athar,2010; Hayesetal., 2011).Consequently,any
changesinODCactivityandpolyamineslevelhaveeffectsoncell differentiationandproliferation.Inbreastcancercells,estrogens induceODCactivity.So,thelevelofODCandestrogenisincreased
∗ Correspondingauthor.
E-mail:Asafshar@iau-neyshabur.ac.ir(A.S.Afshar).
inthesecells(Zhuetal.,2012).Inaddition,AdenosineDeaminase gene(ADA)encodestheenzymethatcatalyzeshydrolyzationof adenosinetoinosinandparticipatesinpurinesmetabolism,which hasacrucialroleindevelopmentofimmunesystemand matura-tionofmammaliancells(Rietal.,2010;RobertsandRoberts,2012). RecentstudiessuggestthatthelevelofADAenzymeisincreasedin breastcancercellsincontrastwithnormalcells(Aghaeietal.,2010;
Mahajanetal.,2013).Therefore,decreasingofODCandADAgene
expressionandrespectiveenzymeactivitiescanbeagoodstrategy forbreastcancertherapy.
Usingmedicinalplantextractsasanticancerdrugsis amore effectivewayforcancertherapy. About50%ofprescribeddrugs intheworldoriginatefromplants(VanSlambroucketal.,2007). NepetabinaloudensisJamzad,Lamiaceae,isanendemicandrare medicinalplantwhichgrowsexclusivelyintheBinaludMountains inKhorasanRazavi provincein NortheastofIran(Tundisetal., 2013).Thisplanthasbeenappliedwidelyintraditionalmedicine forthetreatmentofrespiratorydisease,digestiveproblemsand osteoarthritis(KoochekiandRezvaniMoghaddam,2012).Previous chemicalinvestigationofN.binaloudensishasshownthepresence ofnepetalactoneand1,8cineole(RustaiyanandNadji,1999).Some
http://dx.doi.org/10.1016/j.bjp.2016.07.005
studiesweredoneaboutantibacterialactivitiesofN.binaloudensis butitscytotoxicityandanticancereffectshavenotbeenstudied yet.Therefore,thepurposeofthestudywastodeterminethe anti-cancereffectsofN.binaloudensisextractontwobreastcancercell lines(MCF-7,MDA-MB-231)andnormalbreastcellline(MCF-10A). Also,thepresentstudyisfocusedonevaluatingthelevelofODC1 andADAgeneexpressioninthenormalandcancerousbreastcells.
Materialandmethods
Plantcollectionandextraction
Shoots ofNepetabinaloudensis Jamzad,Lamiaceae,were col-lected from BinaludMountains in Neyshabur, Khorasan Razavi province,northeastofIran.TheplantwasidentifiedbyMr.M.R. Joharchi,fromFerdowsiUniversityofMashhadHerbarium(FUMH). Voucherspecimen(No.1025) wasdeposited withherbariumof IslamicAzadUniversity, NeyshaburBranch.The extraction pro-cesswasperformedfromdriedshoots.Thedriedshoots (100g) werepercolatedwith100mlofeachsolvent(EtOH70%,hexaneand H2O)for4hseparately.Theextractswerefilteredandthesolvents wereevaporatedundervacuumat45◦Ctoaffordcrudeextracts. Dryextractsweredissolvedindimethylsulfoxide(DMSO)andthen subjectedtocytotoxicandgeneexpressionassays.
Celllinesandculturemedium
Twohumanbreastcancercelllines(MCF-7,MDA-MB-231)and onenormal breast cellline(MCF-10A) wereobtainedfromcell culturelaboratoryoftheFacultyofSciences,FerdowsiUniversity (Mashhad,Iran)andPasteurInstitute(Tehran,Iran).Thecellswere culturedin25cm2flaskswithRPMI-1640(RPMISigma)and sup-plementedwith10%fetalbovineserum(FBS,Gibco)and100U/ml penicillinand100g/mlstreptomycinsolutions,at37◦Cwith5% CO2and95%humidity.Themediumwasexchangedeverydayand thecellswerepassagedevery3–4days.
Cytotoxicityassay
ThecytotoxicactivityoftheextractswasdeterminedusingMTT assay(2,3,5dipheniltetrazoliumbromide).Cellswereplatedwith densityof3×104cells/dishin96wellplatesandwereincubated for24hat37◦C.Then,thecellsweretreatedwithdifferent concen-trationsofextracts(0,10,20,40,80,160and320g/ml)andwere incubatedfor24,48and72h.A2mMMTTsolutionwasaddedto eachwellandplatewasincubatedfor4h.Themediumwasthen discardedand100mlDMSO(dimethylsulfoxide)wasaddedand theplatewasshakenfor10min.Finally,opticaldensitywas deter-minedin540nmusingELISAmicroplatereader(Awareness,Palm City,FL,USA).Eachexperimentwasperformedintriplicate.Results areexpressedasthepercentagegrowthinhibitionwithrespectto theuntreatedcells.
Colonyformationassay
TheeffectofhexaneextractonMCF7andMDA-MB-231cells wasinvestigatedbycolonyformationassay(Frankenetal.,2006). Briefly,thecells(500cells/ml)wereallowedtogrowin60mmPetri dishesfor12h.Subsequently,thecellsweretreatedfor48hwith hexaneextract(10,30and60g/ml),or0.1%DMSO.Thecolonies werefixedandstainedwith0.2%crystalvioletandcountedunder stereomicroscope.
Real-timePCR
Theexpression levelsofODC1and ADAgenes,key genesin breastcancercells,wereanalyzedusingRealtimePCRassay.The cells wereculturedinthree groupsand treatedwithincreasing concentrations (0–60g/ml) ofthe hexane extract dissolvedin DMSOfor48h.After48htotalcellularRNAwasextractedfrom thetreated and untreated (control) cells using Easy BLUEtotal RNA extractionkit (iNtRON). Thetreatments wererepeatedfor threetimes.ThequalityofRNAwasdeterminedbyabsorptionat 260nmusingT80UV-VisibleSpectrophotometer(PGInstruments). Foreachspecimen2gofRNAwasreversetranscribedintocDNA withreversetranscriptionreactionmixtureofPowercDNA Syn-thesisKit(iNtRON).PrimersofODC1,ADAandACTweredesigned usingGeneRunnersoftware(version4).
Primer sequences for ODC1 were: ODC1-FW: 5′ -GTGGGTGATTGGATGCTCTTTG-3′ ODC1-RV: 5′-ACCAGGCTAACTA CTCGCTCAA-3′.
Primer sequences for ADA were: ADA-FW: 5′-ACCAG GCTAACTACTCGCTCAA-3′ ADA-RV: 5′-TCAGTAAAGCCCATG TCCCGTT-3′.
Primer sequences for ACT were: ACT-FW: 5′ -TCCATCATGAAGTGTGACGT-3′ ACT-RV: 5′-GAGCAATGATCTTGAT CTTCAT-3′.
Real-timePCRwascarriedoutusingSYBRGreenmethodonan ABIStep-Oneinstrument(AppliedBiosystems,USA).ThecDNAwas takenasatemplateforPCRamplificationofADAandODC1genes andACTgene(controlgene).Eachreactionwasrepeatedforthree times.APCRreactionmixtureof20lcontained10lSYBRGreen mastermix,0.4lReverseprimerand0.4lForwardprimer,6.2l dH2O(RNasefree)and3lcDNA(4ng).Threepairsofprimerswere usedseparately.Thethermalcyclingconditionswereasfollow:1 denaturationcycleof95◦C for10minand40cyclesof95◦Cfor 15s,60◦Cfor60s(annealingandextensiontemperature).A neg-ativecontrolwasusedineachruntoassessspecificityofprimers andpossiblecontamination.TherealtimePCRdatawereanalyzed usingtherelativegeneexpression(Ct)method,asdescribed inAppliedBiosystemsUserBulletinNo.2(LivakandSchmittgen, 2001).Briefly,thedataarepresentedasthefoldchangeingene expressionnormalizedtothereferencegene(-actin)andwere determinedusingtheequationfoldchange=2−Ct.
Statisticalanalysis
One-wayanalysisofvarianceandFisher’sLSDwereusedfordata analysis.Allresultswereexpressedasmean±SEM,andpvalues lowerthan0.05werejudgedassignificant.
Results
CytotoxicityofvariousextractsofNepetabinaloudensis
120
MCF-10A (H2O)
24 h 48 h 72 h
10 20 40 80 160 320 0
100 80 60 40
Cell viability
, %
20 0
120 MCF-10A (EtOH)
10 20 40 80 160 320 0
100 80 60 40
Cell viability
, %
Cell viability
, %
20 0
120
MDA-MB-231 (H2O)
MDA-MB-231 (EtOH)
MDA-MB-231 (n-hexane)
MCF7 (EtOH)
MCF7 (n-hexane) 10 20 40 80 160 320
0 100
80 60 40 20 0
120 100 80 60 40 20 0
120 100 80 60 40 20 0
120 100 80 60 40 20 0 120 100 80 60 40 20 0 120
MCF7 (H2O)
10 20 40 80 160 320 0
10 20 40 80 160 320
0 0 10 20 40 80 160 320
10 20 40 80 160 320
0 0 10 20 40 80 160 320
100 80 60 40 20 0
120 MCF 10A (n-hexane)
Extract concentration (µg/ml) Extract concentration (µg/ml) Extract concentration (µg/ml) 10 20 40 80 160 320
0 100
80 60 40 20 0
24 h 48 h 72 h 24 h 48 h 72 h
Fig.1.CytotoxiceffectsofNepetabinaloudensisvariousextractsonMCF-10A(Normal),MBA-MD-231andMCF-7(cancer)breastcelllines.Cellsweretreatedfor24,48and 72hwithdifferentconcentrationsofextracts.CytotoxicitywasdeterminedbyMTTassay.Resultsarethemean±SEMofthreeindependentexperiments.
Table1
IC50values(g/ml)fordifferentextractsofNepetabinaloudensisincelllines.
Plantextract Cellline
MBA-MD-231 MCF-7 MCF-10A
24h 48h 72h 24h 48h 72h 24h 48h 72h
EtOH70% >300 138.4 97.42 249.5 101.9 65.96 >300 >300 >300
Hexane 234.17 92.18 61.29 132 60.89 39.24 >300 >300 147.8
H2O >300 >300 >300 >300 281.7 229.5 >300 >300 >300
sensitivetothecytotoxiceffectsoftheextractscomparedto MBA-MD-231cells.
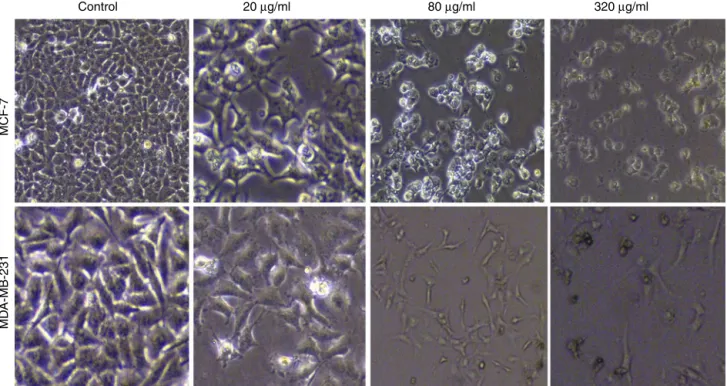
Fig.2showsthephotomicrographicimagesofMCF-7and MBA-MD-231celllines treated by hexane extract of N.binaloudensis. Cytotoxicity effects were coupled with morphological changes includingdecreaseincellvolumeandshrinkingofthecells.
Paclitaxel(700nM)wasusedasapositivecontrol.Paclitaxelat thisconcentrationdecreasedtheviabilityofMCF-7and MBA-MD-231cellsto6.3%±0.6and12.2±0.3incomparisonwithuntreated controlreceivedanequalvolumeofthesolventrespectively(data notshown).
Invitroclonogenicsurvivalassay
AsgiveninFig.3thereisasignificantdosedependent inhibi-tionincolonyformationinMCF-7andMBA-MD-231afterhexane extracttreatments.Theresultswereshowedadecreaseincolony formationby30–50%inMBA-MD-231whilea45–70%decrease
wasobservedinMCF-7whentreatedwith30g/mland60g/ml n-hexaneextractrespectively.
ChangesintheexpressionofODC1andADA
Ourresultsshowedthatexposureofcells toincreasing con-centrations of N. binaloudensis hexane extract for 48h caused significantdecreaseinthemRNAlevelsofODC1andADAincancer celllinesbut notnon-cancercells.TheeffectofN.binaloudensis on selected genes expression was shown tobe concentration-dependent.N.binaloudensishexaneextractatconcentrationsof10, 30and60g/mlsignificantlydecreasedtheODC1expressionby 1.4,2.3and4.97fold,respectivelyinMCF-7cellline. Moreover, thisextractsignificantlyreducedmRNAlevelsofODC1in MDA-MB-231celllineby1.2,1.7and3.6foldwith10,30and60g/ml, respectively(Fig.4).
Control
MCF-7
MD
A-MB-231
20 µg/ml 80 µg/ml 320 µg/ml
Fig.2.MorphologicalchangesofMCF-7andMDA-MB-231cellstreatedwithdifferentconcentrationsofthehexaneextractofNepetabinaloudensisfor48hviewedunder invertedphase-contrastmicroscope(200×magnification).Reduceincellpopulationwasnotedwiththeincreaseintheconcentrationofthetreatmentascomparedtothe control(untreatedcells).
MCF7 MDA-MB-231
Number of colonies
, %
120
100
80
60
40
20
0
0 10
Extract concentration (µg/ml)
∗∗
∗∗
30 60
120
100
80
60
40
20
0
0 10
Extract concentration (µg/ml)
∗∗
∗∗
30 60
Fig.3.ColonyformationassayshowsasignificantreductionincolonyformationinMCF-7andMDA-MB-231celllinestreatedwithdifferentconcentrationofhexaneextract ofNepetabinaloudensis.Theresultswerepresentedasmean±SEM,n=3(**p<0.01).
dose-dependentmannerby1.32,1.83and3.49foldwith10,30and 60g/ml,respectively.AsshowninFig.5,at10,30and60g/ml concentrationofN.binaloudensisextractADA mRNAexpression wasreduced1.21, 1.56and 2.56fold in MDA-MB-231cellline, respectively(Fig.5).Incancercelllines,mRNAexpressionofMCF-7 wasreducedmorethanthatofMDA-MB-231.
Discussion
Several studies suggest an increased polyamine concentra-tion in breast cancertissue compared to normal breast tissue and polyamines biosynthesis inhibitors decrease thegrowth of canceroustumors (Brownet al., 2009; Arisanet al.,2012; Zhu et al.,2012).Ornithine decarboxylase (ODC1),is a key enzyme in polyamine biosynthesis which decarboxylates Ornithine to putrescine(WangandJiang,2014).Furthermore,adenosine deami-nase(ADA)enzyme,whichcatalyzesthehydrolyticdeaminationof adenosinetoinosine,playsanimportantroleinpurinemetabolism andAdenosinehomeostasis(Garcia-Giletal.,2015).Ithasbeen proved to be a direct correlation between breast cancer and
increasedADAenzymeactivityincancertissueofpatients(Gocmen
etal.,2009;Rietal.,2010;Ogataetal.,2011).Therefore,ODC1and
ADAgenesaretheappropriatetargetinanticancerresearch. Accordingtopreviousstudiesandtheimportanceof recogniz-inganti-cancermechanismsofplantextracts,inpresentstudythe effectsofvariousextractsofN.binaloudensisonthecell prolifer-ationandtheexpressionofADAandODCgenesinbreastcancer celllines(MCF-7,MDA-MB-231)withrespecttonon-cancerlines (MCF-10A)wereevaluated.
Theresultsshowedthataftertreatmentofcells(at24,48and 72h)withextractsofhexaneandEtOH70%,excludingH2Oextract, alinearrelationshipbetweenthedoseswiththepercentageofcell viabilitywasobserved.Accordingtotheresultsofthisstudyhexane andEtOHextractofthisplanthasanti-cancereffects.
1.2
0
0 10 30 60 0 10 30 60
0 10 30 60
0.2 0.4 0.6
ODC1 mRNA (f
old)
MCF-10A
MCF-7
Extract concentration (µg/ml)
∗∗
∗∗
∗∗
∗∗ ∗∗
MDA-MB-231
0.8 1
1.2
0 0.2 0.4 0.6
ODC1 mRNA (f
old) 0.8
1
1.2
0 0.2 0.4 0.6 0.8 1
Fig.4.Quantitativereal-timePCRanalysisofODC1mRNAexpressioninonenormalbreastcellline(MCF-10A)andtwocancerousbreastcelllines(MDA-MB-231and MCF-7).ThecellswereincubatedwiththeindicatedconcentrationsofthehexaneextractofNepetabinaloudensisfor48h.ODC1mRNAlevelsdecreaseincancercelllinesin dose-dependentmanner.RelativeabundanceofmRNAisobtainedbynormalizationto-actinexpression.Resultsarethemean±SEMofthreeindependentexperiments. **p<0.01comparedtocontrol.
1.2
0 0.2 0.4 0.6
ODC1 mRNA (f
old) 0.8
1.2
0 0.2 0.4 0.6
AD
A mRNA (f
old) 0.8 1
1.2
0 0.2 0.4 0.6 0.8 1
MCF-10A
0 10 30 60
0 10 30 60
0 10 30 60
MDA-MB-231
∗
∗∗
∗
∗∗
∗∗
∗∗
MCF-7
Extract concentration (µg/ml)
components(Mohammadpouretal.,2013).Thecytotoxiceffects ofthesemonoterpeneshavebeenpreviouslyshownagainst dif-ferentcelllines.1,8-cineole,themaincompoundpresentinthe N.binaloudensisoil, hasbeen reportedtoinhibit thegrowthof liver-derived(HepG2)andextrahepatic(A549)celllines(Rodenak
Kladniew et al., 2014). The results of another study indicated
1,8-cineolesuppressedhumancolorectalcancerproliferationby inducingapoptosis(Murataetal.,2013).Alphaterpineolanother keycomponentoftheoil,hasalsobeenreportedtoinhibitsthe growthoftumorcellsthroughamechanismthatinvolves inhibi-tionoftheNF-Bpathway(Hassanetal.,2010).␦-Terpineolinhibits cellgrowthandinducesapoptosisinhumanlivercancerBEL-7402 cells(Wuetal.,2014).Anti-tumoreffectof␣-pineneonhuman hep-atomacelllinesthroughinducingG2/Mcellcyclearresthasbeen reported(Chenetal.,2015).It wasdemonstratedthat␣-pinene inhibitedBEL-7402cellsbyarrestingcellgrowthintheG2/Mphase ofthecellcycle,downregulatingCdc25CmRNAandprotein expres-sion,and reducingcycledependenceonkinase1(CDK1)activity (Chenetal.,2014).
Amongtheextracts,hexaneextractexhibitedthelargest cyto-toxic effects on cancercells. This can bedescribed by the low polarityofhexane,whichextractlow-polarcompoundsthatare eitherfavorablyabsorbedthroughthecellorhavecytotoxic activ-ity.Therefore,itisassumedthatmostofthecytotoxiccompounds wereconcentratedinthehexaneextract(Tayarani-Najaranetal.,
2013).
The results of our study revealed that the cytotoxicity of thehexane extractofN.binaloudensisontheestrogen receptor-positivecellline(MCF7)(ER+/PR+/HER2−)wasstrongerthanthat ofER/PR/HER2triplenegativebreastcancercellline(MDA-MB231). SuchfindingsaresimilartothosefoundintheworkofSrisawatetal.
(2015)whofoundahigherapoptosisandgrowthinhibitionratein
MCF-7cellsthaninMDA-MB-231cells.Thehighercytotoxiceffect ofN.binaloudensisextractonMCF-7wasprobablyduetolackof estrogenstimulationinthisexperiment.
Thedecreaseofthecellsviabilitycouldbebecauseofanincrease ofapoptosisand/ordecreasedcellreproduction(Islametal.,2013). Inourstudy,acolonyformationassaywasperformedtoevaluate thehexaneextractcapabilityofarrestingproliferationof cancer-ouscellsbyloweringreproductiveability.Ourresultsrevealedthat cytotoxiceffectsofhexaneextractcouldbeduetoadecreaseofcell proliferation.
Accordingtothecytotoxicityresults,hexaneextractexhibited thelargest cytotoxiceffects oncancercells. Therefore, in gene expressionstudieshexaneextractwasusedinsafeconcentrations (0–60g/ml).SignificantreductioninexpressionofODC1genewas observedinbreastcancercelllines(MCF-7andMDA-MB-231)after 48htreatmentwithN.binaloudensishexaneextract.Other stud-ieshave showntheinhibitoryeffectsofplantmaterialsonODC activityand expressionin cancerouscelllines andtissues. Liao
etal.(2008)foundthatenzymeactivityandproteinexpression
ofODCwerereducedincurcumintreatedleukemiaHL-60cells. Theysuggestedthatcurcumin-inducedapoptosisoccursthrougha down-regulatingmechanismofODC.Inanotherstudy,Hakimuddin
etal.(2008)reportedmorethan3-foldreductioninODC1mRNA
levelintumorsofwinepolyphenol-treatedmice.
However, the mechanism by which N. binaloudensis extract reducesODC1geneexpressionisunknown.IntheODC1promoter, therearemultiplebindingsitesforAp-1,Ap-2andc-Myc(Liaoetal., 2008).ThemechanismsoftheinhibitionofODC1expressionby N.binaloudensisextractlikelyinvolvetranscriptionalinhibitionof thesefactors.
ODCenzymeactivityisrelatedtotheproductionofpolyamines such as spermine that function as a free radical scavenger
(Smirnovaetal.,2012).InhibitionofODCgeneexpressionbyN.
binaloudensisextractdecreasestheconcentrationofpolyamines.
Thus,reductionofpolyaminescouldincreaseintracellularROSand causeapoptosisincancercells(Wangetal.,2011).
ADA gene expression decreased in the cancerous human breastcelllinestreatedbyN.binaloudensishexaneextract.Many researchersreportedthatplantextractshaveinhibitoryeffecton ADA expression and activity. For example, Durak et al. (2005)
showedthatblackgrapeextractssignificantlyinhibitedtheADA enzymeactivityincancerouscolontissues.Inanotherstudy,ithas beenfoundthatexpressionoftheadenosinedeaminasegenewas consistentlydecreasedbyalltheberriesinintestinaladenoma
tis-sueofrats(Mutanenetal.,2008).Tomatojuicehasbeenreportedto
causessignificantinhibitiononADAactivityintheprostatecancer tissues(Duraketal.,2003).
Our datademonstratethatdecreaseofadenosine deaminase expressionabolishesadvantageofcancercellsinobtainingmore nucleotides inDNA synthesis and proliferation(Namuslu etal., 2014).Also,inhibitionofADAexpressioncausesincreased avail-ability of adenosine which is a protective molecule in case of tumorouscondition(Mahajanetal.,2013).
Inconclusion,thestudydescribedheredemonstrates thatN. binaloudensishexaneextracthasinhibitoryeffectsonODC1and ADAgeneexpressioninbreastcancercellline.Therefore,a possi-blemechanismforobservedcytotoxicityeffectofthisplantextract couldbeexpressioninhibitionofthesegenes.Thisisthefirstreport aboutthecytotoxicityofN.binaloudensishexaneextractby inhibi-tionofODC1andADAinmRNAlevel,hencefurtherstudieswillbe necessarytosupplementourfindings.
Ethicaldisclosures
Protectionofhumanandanimalsubjects. Theauthorsdeclare thatnoexperimentswereperformedonhumansoranimalsfor thisstudy.
Confidentialityofdata. Theauthorsdeclarethatnopatientdata appearinthisarticle.
Righttoprivacyandinformedconsent.Theauthorsdeclarethat nopatientdataappearinthisarticle.
Authorcontributions
ASAdesigned and supervised theexperiments,analyzed the dataanddraftedthepaper;FSNperformedexperiments,analyzed dataanddraftedthepaper;MMperformedexperimentsand ana-lyzeddata;AKdesignedandanalyzedthedata.Alltheauthorshave readthefinalmanuscriptandapprovedthesubmission.
Conflictsofinterest
Theauthorsdeclarenoconflictsofinterest.
Acknowledgment
This work was financially supported by Neyshabur Branch, IslamicAzadUniversityNeyshabur,Iran.
References
Aghaei,M.,Karami-Tehrani,F.,Salami,S.,Atri,M.,2010.Diagnosticvalueof adeno-sinedeaminaseactivityinbenignandmalignantbreasttumors.Arch.Med.Res. 41,14–18.
Arisan,E.D.,Obakan,P.,Coker,A.,Palavan-Unsal,N.,2012.Inhibitionofornithine decarboxylasealterstheroscovitine-inducedmitochondrial-mediated apopto-sisinMCF-7breastcancercells.Mol.Med.Rep.5,1323–1329.
Chen,W.,Liu,Y.,Li,M.,Mao,J.,Zhang,L.,Huang,R.,Ye,L.,2015.Anti-tumoreffectof alpha-pineneonhumanhepatomacelllinesthroughinducingG2/Mcellcycle arrest.J.Pharmacol.Sci.127,332–338.
Chen,W.Q.,Xu,B.,Mao,J.W.,Wei,F.X.,Li,M.,Liu,T.,Zhang,L.R.,2014.Inhibitory effectsofalpha-pineneonhepatomacarcinomacellproliferation.AsianPac.J. Cancer.Prev.15,3293–3297.
Deng,W.,Jiang,X.,Mei,Y.,Sun,J.,Ma,R.,Liu,X.,Sun,X.,2008.Roleofornithine decarboxylaseinbreastcancer.ActaBiochim.Biophys.Sin.40,235–243. Durak, ˙I.,Biri,H.,Avcı,A.,Sözen,S.,Devrim,E.,2003.Tomatojuiceinhibitsadenosine
deaminaseactivityinhumanprostatetissuefrompatientwithprostatecancer. Nutr.Res.23,1183–1188.
Durak,I.,Cetin,R.,Devrim,E.,Erguder,I.B.,2005.Effectsofblackgrapeextracton activitiesofDNAturn-overenzymesincancerousandnoncanceroushuman colontissues.LifeSci.76,2995–3000.
Elmets,C.A.,Athar,M.,2010.Targetingornithinedecarboxylasefortheprevention ofnonmelanomaskincancerinhumans.CancerPrev.Res.3,8–11.
Formisano,C.,Rigano,D.,Senatore,F.,2011.Chemicalconstituentsandbiological activitiesofNepetaspecies.Chem.Biodivers.8,1783–1818.
Franken,N.A.,Rodermond,H.M.,Stap,J.,Haveman,J.,vanBree,C.,2006.Clonogenic assayofcellsinvitro.Nat.Protoc.1,2315–2319.
Garcia-Gil,M.,Tozzi,M.G.,Varani,S.,DellaVerde,L.,Petrotto,E.,Balestri,F.,Camici, M.,2015.Thecombinationofadenosinedeaminaseinhibitionand deoxyadeno-sineinducesapoptosisinahumanastrocytomacellline.Neurochem.Int.80, 14–22.
Gocmen,E.,Tez,M.,Ozturk,S.,Koc,M.,Demirci,S.,2009.Activitiesofadenosine deaminaseand5′-nucleotidaseincancereousandnon-cancereoushuman gas-trictissues.Bratisl.Lek.Listy110,416–418.
Hakimuddin,F.,Tiwari,K.,Paliyath,G.,Meckling,K.,2008.Grapeandwine polyphe-nolsdown-regulatetheexpressionofsignaltransductiongenesandinhibitthe growthofestrogenreceptor–negativeMDA-MB231tumorsinnu/numouse xenografts.Nutr.Res.28,702–713.
Hassan,S.B., Gali-Muhtasib,H., Goransson,H., Larsson,R., 2010. Alpha terpi-neol:apotentialanticanceragentwhichactsthroughsuppressingNF-kappaB signalling.AnticancerRes.30,1911–1919.
Hayes,C.S.,Defeo,K.,Dang,H.,Trempus,C.S.,Morris,R.J.,Gilmour,S.K.,2011.A prolongedandexaggeratedwoundresponsewithelevatedODCactivitymimics earlytumordevelopment.Carcinogenesis32,1340–1348.
Islam,M.,Sharma,S.,Kumar,B.,Teknos,T.N.,2013.AtorvastatininhibitsRhoC func-tionandlimitsheadandneckcancermetastasis.OralOncol.49,778–786. Khakdan,F.,Rassam,G.A.,2014.Chemicalcomposition,antibacterialactivity,and
cytotoxicityofessentialoilfromNepetaucrainicaL.spp.kopetdaghensis.Ind. Crop.Prod.58,315–321.
Koocheki,A.,RezvaniMoghaddam,P.,2012.Firstexperimentsoncultivationof
NepetabinaloudensisJamzad–anexampleofdomesticationofahighly
endan-geredmedicinalplantofIran.Z.Arznei-Gewurzpfla17,64–71.
Liao,Y.F.,Hung,H.C.,Hour,T.C.,Hsu,P.C.,Kao,M.C.,Tsay,G.J.,Liu,G.Y.,2008. Curcumininducesapoptosisthroughanornithinedecarboxylase-dependent pathwayinhumanpromyelocyticleukemiaHL-60cells.LifeSci.82,367–375. Mahajan,M.,Tiwari,N.,Sharma,R.,Kaur,S.,Singh,N.,2013.Oxidativestressand
itsrelationshipwithadenosinedeaminaseactivityinvariousstagesofbreast cancer.IndianJ.Clin.Biochem.28,51–54.
Mohammadpour,N.,Emami,S.A.,Asili,J.,2013.Identificationofvolatileoil com-ponentsofNepetabinaloudensisJamzadbyGC-MSand13C-NMRmethodsand
evaluationofitsantimicrobialactivity.J.Essent.OilBear.Plants16,102–107. Mousavi,S.M.,Gouya,M.M.,Ramazani,R.,Davanlou,M.,Hajsadeghi,N.,Seddighi,Z.,
2009.CancerincidenceandmortalityinIran.Ann.Oncol.20,556–563. Murata,S.,Shiragami,R.,Kosugi,C.,Tezuka,T.,Yamazaki,M.,Hirano,A.,Koda,
K.,2013.Antitumoreffectof1,8-cineoleagainstcoloncancer.Oncol.Rep.30, 2647–2652.
Mutanen,M.,Pajari,A.M.,Paivarinta,E.,Misikangas,M.,Rajakangas,J., Martti-nen,M.,Oikarinen,S.,2008.Berriesaschemopreventivedietaryconstituentsa
mechanisticapproachwiththeApcMin/+mouse.AsiaPac.J.Clin.Nutr.17(Suppl. 1),123–125.
Namuslu,M.,Kocaoglu,H.,Celik,H.T.,Avci,A.,Devrim,E.,Genc,Y.,Durak,I.,2014. Effectsofaqueoussoybean,mistletoeandredcloverextractsonactivitiesof adenosinedeaminaseandxanthineoxidaseenzyme. Bratisl.Lek.Listy115, 367–371.
Ogata,Y.,Aoe,K.,Hiraki,A.,Murakami,K.,Kishino,D.,Chikamori,K.,Tanimoto,M., 2011.Isadenosinedeaminaseinpleuralfluidausefulmarkerfor differenti-atingtuberculosisfromlungcancerormesotheliomainJapan,acountrywith intermediateincidenceoftuberculosis?ActaMed.Okayama65,259–263. Ri,G.,Ohno,S.,Furutani,M.,Furutani,Y.,Tsukahara,T.,Hagita,N.,Matsuoka,R.,2010.
AnindicationforcorrelationbetweentheserumADAlevelandgastriccancer risk.AnticancerRes.30,2347–2349.
Rigano,D.,Arnold,N.A.,Conforti,F.,Menichini,F.,Formisano,C.,Piozzi,F.,Senatore, F.,2011.CharacterisationoftheessentialoilofNepetaglomerataMontbretet AucherexBenthamfromLebanonanditsbiologicalactivities.Nat.Prod.Res. 25,614–626.
Roberts,E.L.,Roberts,O.T.,2012.Plasmaadenosinedeaminaseisoform2incancer patientsundergoingchemotherapy.Br.J.Biomed.Sci.69,11–13.
RodenakKladniew,B.,Polo,M.,MonteroVillegas,S.,Galle,M.,Crespo,R.,Garciade Bravo,M.,2014.Synergisticantiproliferativeandanticholesterogeniceffectsof linalool,1,8-cineole,andsimvastatinonhumancelllines.Chem.Biol.Interact. 214,57–68.
Rustaiyan,A.,Nadji,K.,1999.CompositionoftheessentialoilsofNepetaispahanica
Boiss,andNepetabinaloudensisJamzadfromIran.FlavourFrag.J.14,35–37. Siegel,R.,Ma,J.,Zou,Z.,Jemal,A.,2014.Cancerstatistics.CACancerJ.Clin.64,9–29. Smirnova,O.A.,Isaguliants,M.G.,Hyvonen,M.T.,Keinanen,T.A.,Tunitskaya,V.L., Vepsalainen,J.,Ivanov,A.V.,2012.Chemicallyinducedoxidativestressincreases polyaminelevelsbyactivatingthetranscriptionofornithinedecarboxylaseand spermidine/spermine-N1-acetyltransferaseinhumanhepatomaHUH7cells. Biochimie94,1876–1883.
Srisawat,T.,Sukpondma,Y.,Graidist,P.,Chimplee,S.,Kanokwiroon,K.,2015.The dosedependentinvitroresponsesofMCF-7andMDA-MB-231celllinesto extractsofVaticadiospyroidesSymingtontypeSSfruitincludeeffectsonmode ofcelldeath.Pharmacogn.Mag.11(Suppl.1),S148.
Tayarani-Najaran,Z.,Asili,J.,Aioubi,E.,Emami,S.A.,2013.Growthinhibitionand apoptosisinductionofSalviachloroleucaonMCF-7breastcancercellline.Iran. J.Pharm.Res.12,789–799.
Tundis,R.,Nadjafi,F.,Menichini,F.,2013.Angiotensin-convertingenzymeinhibitory activityandantioxidantpropertiesofNepetacrassifoliaBoiss&BuhseandNepeta
binaloudensisJamzad.Phytother.Res.27,572–580.
VanSlambrouck,S.,Daniels,A.L.,Hooten,C.J.,Brock,S.L.,Jenkins,A.R.,Ogasawara, M.A.,Steelant,W.F.,2007.Effectsofcrudeaqueousmedicinalplantextractson growthandinvasionofbreastcancercells.Oncol.Rep.17,1487–1492. Wang,M.F.,Liao,Y.F.,Hung,Y.C.,Lin,C.L.,Hour,T.C.,Lue,K.H.,Liu,G.Y.,2011.
Hydroxydibenzoylmethaneinduces apoptosis throughrepressingornithine decarboxylaseinhumanpromyelocyticleukemiaHL-60cells.Exp.Mol.Med. 43,189–196.
Wang,X.,Jiang,L.,2014.Effectsofornithinedecarboxylaseantizyme1onthe pro-liferationanddifferentiationofhumanoralcancercells.Int.J.Mol.Med.34, 1606–1612.
Wu,Z.L.,Yin,Z.Q.,Du,Y.H.,Feng,R.Z.,Ye,K.C.,Wei,Q.,Wang,Y.,2014. gamma-Terpineolinhibitscellgrowthandinducesapoptosisinhumanlivercancer BEL-7402cellsinvitro.Int.J.Clin.Exp.Pathol.7,6524–6533.
Xu,L.,Long, J.,Wang, P.,Liu, K.,Mai, L.,Guo,Y.,2015. Associationbetween theornithinedecarboxylaseG316Apolymorphismandbreastcancersurvival. Oncol.Lett.10,485–491.