w w w . r e u m a t o l o g i a . c o m . b r
REVISTA
BRASILEIRA
DE
REUMATOLOGIA
Original
article
Assessment
of
fatigue
and
dryness
in
primary
Sjögren’s
syndrome:
Brazilian
version
of
“Profile
of
Fatigue
and
Discomfort
–
Sicca
Symptoms
Inventory
(short
form)
(PROFAD-SSI-SF)”
夽
Samira
Tatiyama
Miyamoto
a,b,
Maurício
Aquino
Paganotti
c,
Érica
Vieira
Serrano
b,d,
Raquel
Altoé
Giovelli
b,d,
Valéria
Valim
a,b,∗aEscolaPaulistadeMedicina,UniversidadeFederaldeSãoPaulo,SãoPaulo,SP,Brazil bUniversidadeFederaldoEspíritoSanto,Vitória,ES,Brazil
cUniversidadedeVilaVelha,Vitória,ES,Brazil
dEscolaSuperiordeCiências,SantaCasadeMisericórdiadeVitória,Vitória,ES,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received27May2014 Accepted6October2014 Availableonline6January2015
Keywords:
Sjögren’ssyndrome Questionnaire Validationstudy Evaluation PROFAD-SSI-SF
a
b
s
t
r
a
c
t
Objective:Toperformacross-culturaladaptationandvalidationoftheProfileofFatigue andDiscomfort–SiccaSymptomsInventory(shortform)(PROFAD-SSI-SF)questionnaire assessingthesubjectiveaspectsofthesymptomsofprimarySjögrensyndrome(pSS),for theBrazilianPortugueselanguage.
Method:Conceptual,oftheitem,semanticandoperationalequivalenceswereevaluated. TheBrazilianversionofPROFAD-SSI-SFwasadministeredto62womenwithpSSaccording totheEuropean-Americanconsensus2002toassessmeasurementequivalence.␣-Cronbach
wasusedforinternalconsistency;intraclasscorrelationcoefficient(ICC)forintraobserver reproducibility;andSpearmancorrelationcoefficientforvaliditybycomparingwithPatient Global Assessment(PaGA), EULARSjögren’s SyndromePatientReportedIndex (ESSPRI), FunctionalAssessmentofChronicIllnessTherapyFatigueSubscale(FACIT-F)andEuroQOL (EQ-5D).
Results:TheinternalconsistencyofPROFAD,SSIandtotalscorewas0.80;0.78;and0.87, respectively.TheintraobserverreproducibilityoftotalPROFADwas0.89;oftotalSSIwas 0.86;andtotalscorewas0.89.Intermsofvalidity,PROFADcorrelatedsignificantlywithPaGA (r=0.50),FACIT-F(r=0.59),ESSPRI(r=0.58)andalldomainsofEQ-5D,withtheexceptionof Mobility.Ontheotherhand,SSIcorrelatedsignificantlywithPaGA(r=0.43),FACIT-F(r=0.57), ESSPRI(r=0.55)andmostdomainsofEQ-5D.ThetotalscoreofPROFAD-SSI-SFhada non-statisticallysignificantcorrelationonlywithMobilitydomainandwith1–100rangeofEQ-5D.
夽
Institution:RheumatologyOutpatientClinic,HospitalUniversitárioCassianoAntôniodeMoraes(HUCAM),UniversidadeFederaldo EspíritoSanto(UFES),Vitória,ES,Brazil.
∗ Correspondingauthor.
E-mail:val.valim@gmail.com(V.Valim).
http://dx.doi.org/10.1016/j.rbre.2014.10.002
Conclusion: ThePortugueseversionofPROFAD-SSI-SFprovedtobeanadaptable, repro-ducibleandvalidtoolfortheBrazilianPortugueselanguage.
©2014ElsevierEditoraLtda.Allrightsreserved.
Avaliac¸ão
da
fadiga
e
da
secura
na
síndrome
de
Sjögren
primária:
versão
brasileira
do
“Profile
of
Fatigue
and
Discomfort
–
Sicca
Symptoms
Inventory
(short
form)
(Profad-SSI-SF)”
Palavras-chave:
SíndromedeSjögren Questionário Estudodevalidac¸ão Avaliac¸ão
Profad-SSI-SF
r
e
s
u
m
o
Objetivo: Realizaraadaptac¸ãotransculturaleavalidac¸ãodoProfileofFatigueand Dis-comfort–SiccaSymptomsInventory(shortform)(PROFAD-SSI-SF),questionárioqueavalia osaspectossubjetivosdossintomasdasíndromedeSjögrenprimária(SSp),paraalíngua portuguesabrasileira.
Método: Foiavaliadaaequivalênciaconceitual,deitem,semânticaeoperacional.Aversão brasileiradoProfad-SSI-SFfoiaplicadaa62mulherescomSSpconformeconsenso europeu-americanode2002paraavaliaraequivalênciademensurac¸ão.Foiusadoo␣-Cronbach
paraconsistênciainterna;coeficientedecorrelac¸ãointraclasse(ICC)parareprodutibilidade
intraobservador;ecoeficientedecorrelac¸ãodeSpearmanparavalidadecorrelacionadocom oPatientGlobalAssessment(PaGA),EULARSjögren’sSyndromePatientReportedIndex(ESSPRI),
FunctionalAssessmentofChronicIllnessTherapyFatigueSubscale(FACIT-F)eEuroQOL(EQ-5D).
Resultados: AconsistênciainternadoPROFAD,doSSIedapontuac¸ãototalfoide0,80,0,78e 0,87,respectivamente.AreprodutibilidadeintraobservadordoPROFADtotalfoide0,89;do SSItotalde0,86edapontuac¸ãototalde0,89.Navalidade,oPROFADapresentoucorrelac¸ão significativacomoPaGA(r=0,50),FACIT-F(r=0,59),ESSPRI(r=0,58)etodososdomínios doEQ-5D,comexcec¸ãodamobilidade.JáoSSIapresentoucorrelac¸ãosignificativacom oPaGA(r=0,43),FACIT-F(r=0,57),ESSPRI(r=0,55)eamaioriadosdomíniosdoEQ-5D.A pontuac¸ãototaldoPROFAD-SSI-SFsónãoobtevecorrelac¸ãoestatisticamentesignificante comodomíniomobilidadeeescala1a100doEQ-D5.
Conclusão: AversãoemportuguêsdoPROFAD-SSI-SFmostrouseradaptável,reprodutívele válidaparaalínguaportuguesabrasileira.
©2014ElsevierEditoraLtda.Todososdireitosreservados.
Introduction
Primary Sjögren’s syndrome (pSS) is a systemic disorder
characterized by lymphocytic infiltration and progressive
destruction ofexocrine glands;however, the inflammatory
processcanaffectanyorgan.Theclinicalmanifestationscan besplitintotwoaspects:(1)benign,butdisabling,findings,as dryness,painandfatigue,affectingthemajorityofpatients and(2)systemicfindingsthatcanbepotentiallysevere, affect-ingabout20–40%ofpatients.1Duetothelackofuniversally
acceptedclassificationcriteria,theevaluationoftheincidence andprevalenceofthediseasevariessignificantly,depending
onthecriteriaused.Theprevalencemay rangefrom0.03%
to2.7%ofthe population.2 Inthe Brazilianpopulation,the
prevalence,accordingtotheAmerican-Europeancriteria, is 0.17%.3
Inrecentdecades,expertsestablishedaconsensusthatit iscriticaltoobtainobjectivemeasuresofsaliva4andtear5
pro-duction,aswellastoassessdrynessandfatiguesymptoms, qualityoflifeandactivityanddiseasedamage,throughvalid andreliableassessmentinstruments,6–8 bothtoimprovethe
accuracyofclinicalassessmentbeforeandaftertreatment,as toenableconductingclinicaltrials.
Therearefewself-assessmentinstrumentsfocusedonthe perceptionofthepatienttoassessthesubjectiveaspectsof his/hersymptomsinthisdisease,astheProfileofFatigueand Discomfort(PROFAD),9,10SiccaSymptomsInventory(SSI)10,11
andthemostrecentlytoolcreatedbyEULARandalready val-idatedforBrazilianPortugueselanguage,theEULARSjögren’s SyndromePatientReportedIndex(ESSPRI),1,12whichisa
ques-tionnaireconsistingofthreedomains: fatigue,drynessand pain,assessedbyascalenumberedfrom0to10,withequal weights,andwhosefinalscoreistheaverageofthescoresof thethreedomains.
TheProfileofFatigue andDiscomfort –SiccaSymptoms
Inventory(longform)(PROFAD-SSI-LF)consistsof64questions scoredfrom0to7foreightdomains:somaticfatigue, men-tal fatigue,arthralgia,vascular dysfunction,oculardryness, oraldryness,vaginaldrynessandcutaneousdryness.9,10
which consists of 19 questions distributed in the same 8 domainsofthelongversion,havingtwoscores,onefor PRO-FADandtheotherforSSI.PROFADhasnineitemssplitinto fourdomains: cutaneousfatigue, mentalfatigue,arthralgia andvascular,andSSIhas10itemssplitinto fourdomains: oculardryness,oraldryness,vaginaldryness,andcutaneous dryness.Inbothtoolsthefinalscoreisthesumofitsfour domains,andvariesfrom0to28.11Itsadvantageisthe
dis-tinctionoffatigueinphysicalandmentaltypes,aswellasin sitesofdryness,whichdonotalwaysappearsimultaneously andatthesameintensity.
Recent studies in which PROFAD, SSI and ESSPRI were
correlated showedthat all thesetoolsexhibited significant
correlation amongthemselves and with the patient global
assessmentfordiseaseactivity,thusshowingconfirmationof constructvalidity.13,14 PROFAD wasalsovalidated forother
rheumaticdiseases,suchassystemiclupuserythematosus
and rheumatoid arthritis,and it has been shownthat this
toolhavemoderatetohighsensitivity(65–85%)andspecificity (55–77%).9
Besidesthe needforvalidation ofthis tool,it iscritical thatithasitspsychometricpropertiestestedinseveral lan-guagesandindifferentpopulationsofpatientswithpSS.This wouldallowtherealizationofcross-culturalstudiesand of comparisonsbetweennationalandinternationalstudies,thus providingthescientificcommunicationbetweencountriesof differentlanguages.15–17Thisstudyaimstoconducta
cross-culturaladaptationandvalidationofPROFAD-SSI-SFforthe BrazilianPortugueselanguage.Anattemptwasalsomadeto evaluatethepsychometricpropertiesofatotalscoreforthe instrument,althoughtheoriginalversiondidnotpresentit.
Method
Thisisacross-sectionalobservationalstudyapprovedbythe
Ethics Committeein Research of the Health Science
Cen-ter, UFES (Universidade Federal do Espírito Santo), under
Opinionnumber008/10anddevelopedintheSjögren’s
Syn-dromeAmbulatory,RheumatologyOutpatientClinic,Hospital UniversitárioCassianoAntoniodeMoraes(HUCAM),Vitória, EspíritoSanto.
Theproceduresfortheconductionofacross-cultural
adap-tationofPROFAD-SSI-SFfollowedthemethodologyproposed
byHerdman,Fox-RushbyandBadia(1998),18whichcoverssix
steps:conceptual,ofitem,semantic, operational, measure-mentandfunctionalequivalence.
Conceptual, of item, semantic and operational
equiv-alences of PROFAD-SSI-SF were checked at the time of
translation and retranslation (back translation), bya
com-mittee formed by a rheumatologist and a physiotherapist
specializedinRheumatology,bothusedinthemanagement
ofpSS and fluentin English, and an English teacher. The
translationofPROFAD-SSI-SFwasperformedindependently
bytwoEnglishlanguageteachershavingPortugueseastheir
nativelanguageand awareofthepurposeofthe study.On
retranslation,thisversionwassubmittedtoatranslationinto EnglishbytwootherEnglishteachershavingtheEnglishas
their native language and who were unaware ofthe
origi-nalversionandofthepurposeofthestudy.Toevaluatethe
semanticequivalence,thePortugueseconsensusversionwas appliedto20consecutive patientsdiagnosedwithpSSbya
rheumatologistusedinmanagingpatientswithpSS.
Sixty-two patients were selected for the evaluation of
measurement equivalence across psychometric properties,
accordingtoStreinerandNorman(2008)19andKirshnerand
Guyatt (1995) methodology20; the calculationto composing
this sample wasbased on the use ofatleastfive patients perdomainoftheinstrument.21Theinclusioncriteriawere:
diagnosisofpSSaccordingtotheAmerican-European classi-ficationcriteriaforSjögren’ssyndrome,22 18yearsofageor
over,andsigningafreeandinformedconsentterm.Patients
withotherconcomitantautoimmunediseaseswereexcluded.
Diseaseactivitywassubjectivelyassessedasinactiveand active(low,moderateandhighactivity),accordingtotheonset orworseningofpotentiallyreversiblesignsandsymptomsof diseaseinthelastfourweeks,definedbyamedicalexpert,as wellasbyEULARSjögren’sSyndromeDiseaseActivityIndex (ESSDAI), whose score ranges from 0 to 123.17,23 The
psy-chometricpropertiesevaluatedwere:reliability(intraobserver reliability) and construct validity. The 62 patients under-went clinical evaluationbyaphysicianusedinthecareof patientswithpSS,withcompletionofthefollowing
instru-ments: PROFAD-SSI-SF, Patient Global Assessment (PaGA),
ESSPRI, Functional Assessment of Chronic Illness Therapy
FatigueSubscale(FACIT-F)24,25andEuroQOL(EQ-5D)26ina
sin-glemoment(visit1)andaftertwodays(visit2).
We opted to replace the self-application of the
instru-mentforaface-to-faceinterviewintheevaluationstagesof
semanticequivalenceandofmeasurement,consideringthat
thischangedoesnotinvalidatetheuseofthequestionnaire byself-application27; thiscouldbeexplainedbythe lackof
practiceofpatientsincompletingself-administered question-naires,orbytheirlowlevelofschooling,28andalsotoavoidthe
exclusionofilliteratepatientsorthosewitheyeproblems.29
␣-Cronbachwasusedinthe analysisofinternal
consis-tency;forintra-observerreproducibility,theintraclass corre-lationcoefficient(ICC)wasused.Fortheconstructvalidityof thecomparisonofPROFAD-SSI-SFwithPaGA,ESSPRI,FACIT-F andEQ-5D,theSpearmancorrelationcoefficientwasused.In allanalyzes,thelevelofsignificancewasacceptedasp≤0.05.
Results
Inthephaseofconceptual,ofitem,semanticandoperational equivalence,changesinquestions14and19wereintroduced. Inquestion14,inthestudyofsemanticequivalence,theliteral translationof“blurredvision”wouldbevisãoturva;however, the wordturva wasreplacedbyembac¸ada,asthis isaterm considered morepopular. Likewise,the term “swollen sali-varyglands”inquestion19wouldmeaninchac¸odasglândulas salivares,butglândulassalivaresisatechnicaltermdifficultto understandand locateforthe Brazilianpopulation;thus,it wasreplacedbyregiãonorostoàfrentedasorelhaseabaixodo queixoinchados.Therewasnoquestionoritemmisunderstood bymorethan15%ofthe20patientsinthepre-test.Thus,the finalBrazilianlanguageconsensusversionwasobtained.
In the evaluation of measurement equivalence across
Table1–Demographicandclinicalcharacteristicsof62 patientswithprimarySjögren’ssyndrome.
Variables n(%)orMean± SD
Age(years) 49.4 ± 11.6
Femalegender 62(100)
Timeelapsedfromthefirstsymptoms(years) 7.2 ± 5.4
Timesincediagnosis(years) 3.0 ± 3.3
Intervalbetweensymptomsanddiagnosis (years)
4.3 ± 4.9
Systemicmanifestationsinthelastvisit 29(46.8)
Inactivedisease 29(46.8)
Activedisease
Lowactivity 24(38.7)
Moderateactivity 9(14.5)
ESSDAI 4.95 ± 6.73
Clinicalfeatures:
Objectivexerophthalmia(SchirmerI and/orRoseBengal)
48(77.4)
Objectivexerostomia(Salivary flow/Parotidscintigraphy)
53(85.5)
Lymphocyticsialadenitis≥1focus-score 56(90.3)
Anti-Ro 27(43.6)
Anti-La 12(19.4)
Previousparotidglandswelling 16(25.8)
SD,standarddeviation;ESSDAI,EULARSjögren’sSyndromeDisease ActivityIndex.
comingfromthe Sjögren’sSyndromeAmbulatory, Rheuma-tologyOutpatientClinicHUCAM,wereincluded.
Themeanagewas49.4±11.6years.Thedurationofillness was7.2±5.4years;46.8%ofthepatientshadsomesystemic manifestationand97.9%hadbeenmedicatedwith immuno-suppressive agents in some time (Table 1). Most patients (56.41%) hada lowlevel ofschooling (<8years),and 8.06% wereilliterate.Mostpatientsexhibiteddiseaseactivity(53.2%),
however,therewas apredominanceoflowactivity disease
(38.7%).Thislow activitywas confirmedbythe meantotal scoreof4.95withESSDAI(0–39),inwhich11.3%ofpatientshad ascore≥12.ThemeanofPROFADwas18.68±6.23;ofSSIwas 18.19±6.40;andofthetotalscoreofPROFAD-SSI-SF(obtained byaveragingPROFADandSSIresults)was18.34±5.69(Table2). TheinternalconsistencyofPROFAD,SSIandofthetotal scorewasconsideredhigh,anda␣-Cronbachcorresponding
to0.80;0.78;and0.87,respectively,wasobtained.
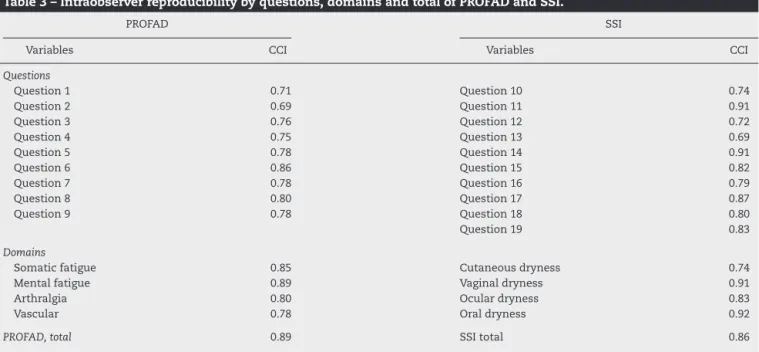
TheintraobserverreproducibilityofPROFADwas0.89;of SSI,0.86;andofthetotalscorewas0.89,showinghigh
repro-ducibility. The concordance ofthe questions and domains
betweenthetwovisitswasconsideredasgood toexcellent (Table3).
WithrespecttotheconstructvalidityofPROFAD, statis-ticallysignificant correlationsofdomains “Somaticfatigue” and“Arthralgia”withallothervariableswereobtained.The domain “Mental fatigue” had astatistically significant cor-relation with PaGA, FACIT-F, ESSPRI (Fatigue, Dryness, and total)andEQ-D5(Pain/Discomfortand Anxiety/Depression).
Ontheotherhand,thedomain“Vascular”showeda
statis-ticallysignificant correlationonly withFACIT-Fand ESSPRI (Pain,Drynessandtotal).AstoPROFAD,alackofstatistically significantcorrelationonlyoccurredwiththedomain “Mobil-ity”ofEQ-D5(Table4).Withrespecttotheconstructvalidity
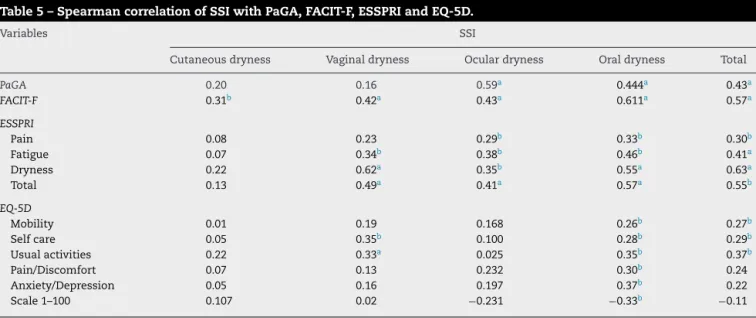
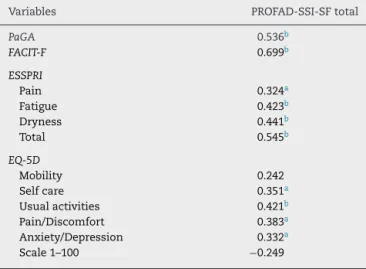
ofSSI,thedomain“Cutaneousdryness”showedastatistically significantcorrelationonlywithFACIT-F;thedomain “Vagi-naldryness”withFACIT-F,ESSPRI(Fatigue,Dryness,andtotal) andEQ-D5(SelfcareandUsualactivities);thedomainOcular drynesswithPaGA,FACIT-FandESSPRI;andthedomain“Oral dryness”,withallvariables.ThetotalscoreofSSIshowed sta-tisticallysignificant correlationswithPaGA,FACIT-F,ESSPRI andEQ-D5(Mobility,SelfcareandUsualactivities)(Table5).On theotherhand,thetotalscoreofPROFAD-SSI-SFonlylackeda statisticallysignificantcorrelationwiththedomain“Mobility” andwith1–100scaleofEQ-D5(Table6).
Discussion
Theeditorialofthisjournalin200630highlightedthe
impor-tanceofthetranslationandvalidationofassessmenttoolsin rheumatologyfortheBrazilianPortugueselanguage, remin-ding that, withagood-quality instrument availabletothat effect, the creationofa newinstrument requiresa greater commitmentoftimeandcost.However,itisnotenoughthat the instrument be simplytranslated; it is critical an accu-rate assessment ofits translation and cultural adaptation, aswellasanevaluationofitsmeasurement(psychometric) properties afterthecompletionofthis process,evenwhen
such properties have already been demonstrated with the
originalinstrument,asimportantculturaldifferencesmaybe present.30,31
PROFAD-SSI-SFwasoriginallywrittenintheEnglish lan-guage,withquestionsappropriatetoitsownculture.Hence, forapplicationofthisinstrumenttoourpopulation,we pro-ceededtoaculturalequivalence,sothatthetoolcouldactually beadequatelyinterpretedbybothpatientandevaluator.
Demographic data and clinical characteristics of 62
patientsinthestageofmeasurementequivalenceevaluation were similartothosefoundinother studiesofassessment
toolsforpSS(PROFAD,SSI,PROFAD-SSI-SFandESSPRI) and
theircohorts:prevalence>93%inwomen,meanagebetween 47and62years,longdiseasedurationaveraging7.21,9–12years,
and mean interval between symptoms and diagnosis of5
years,whichdemonstratesalongdelaytimefordiagnosisof thisdisease,anaspectalreadyreportedbyothers.32
TheBrazilianversionofPROFAD-SSI-SFhasdemonstrated
high internal consistency(0.80 and 0.78), aswell asin the validation study ofthe originalversion,whose ␣-Cronbach
was 0.99.11 The intraobserver reproducibility (r=0.69–0.85)
also was considered high, similar to the reproducibility of the original PROFAD-LF (r=0.67–0.79), tested 24h after the first application.9 Thereproducibilityofthedomains
“Ocu-lardryness”(r=0.92)and“Oraldryness”(r=92)inthestudy ofPROFAD-SSI-SFdevelopmentwasalsosimilartothatinthe presentstudy:0.83and0.92,respectively.33
In the validation study of PROFAD-LF, the questions in
thedomain“Somaticfatigue”showedcorrelationonlywith
the domains “Vitality” of SF-36 (r=−0.53–0.70) and “Physi-calhealth”ofWHOQOL-BREF(r=−0.62andr=−0.69),9unlike
Table2–DescriptivemeasuresofPROFAD-SSI-SF,ESSPRI,EQ-D5,FACIT-FandPaGA.
Variables Mean(±) Minimum Median Maximum SD
PROFAD
Somaticfatigue 5.37 0.50 6 7 1.72
Mentalfatigue 5.1 0 6 7 2.17
Arthralgia 5 0 5.5 7 1.98
Vascular 3.21 0 3.5 7 3.03
TotalPROFAD 18.68 1.5 18.63 28 6.23
SSI
Cutaneousdryness 4.79 0 6 7 2.57
Vaginaldryness 4 0 5 7 2.96
Oculardryness 5.38 0 60 7 1.85
Oraldryness 4.02 0 4.20 7 1.85
TotalSSI 18.19 3.53 18.33 28 6.40
PROFAD-SSI-SF,total 18.34 2.52 18.13 27.63 5.69
ESSPRI
Pain 7.9 0 8 10 2.53
Fatigue 6 0 7.5 10 3.8
Dryness 6.54 0 7 10 2.76
Total 6.81 1.33 7. 10 2.12
EQ-5D
Mobility 1.66 1 2 2 0.48
Selfcare 1.35 1 1 2 0.48
Usualactivities 1.68 1 2 3 0.57
Pain/Discomfort 2.18 1 2 3 0.53
Anxiety/Depression 2.05 1 2 3 0.66
Scale1–100 56.73 0 60 100 24.21
FACIT-F 30.73 1 31 52 11.35
PaGA 7.58 2 8 10 2.15
PROFAD,ProfileofFatigueandDiscomfort;SSI,SiccaSymptomsInventory;PROFAD-SSI-SF,ProfileofFatigueandDiscomfort–SiccaSymptoms Inventory(shortform);PaGA,Patient’sGlobalAssessment;FACIT-F,FunctionalAssessmentofChronicIllnessTherapyFatigueSubscale;ESSPRI, EULARSjögren’sSyndromePatientReportedIndex;EQ-5D,EuroQOL;SD,standarddeviation.
Scoresofinstruments:PROFAD,0–28;SSI,0–28;PROFAD-SSI-SFtotal,0–28;ESSPRI0–10;EQ-5D,domains–1–3,range–0–100;FACIT-F,0–52; PaGA,0–10.
Table3–Intraobserverreproducibilitybyquestions,domainsandtotalofPROFADandSSI.
PROFAD SSI
Variables CCI Variables CCI
Questions
Question1 0.71 Question10 0.74
Question2 0.69 Question11 0.91
Question3 0.76 Question12 0.72
Question4 0.75 Question13 0.69
Question5 0.78 Question14 0.91
Question6 0.86 Question15 0.82
Question7 0.78 Question16 0.79
Question8 0.80 Question17 0.87
Question9 0.78 Question18 0.80
Question19 0.83
Domains
Somaticfatigue 0.85 Cutaneousdryness 0.74
Mentalfatigue 0.89 Vaginaldryness 0.91
Arthralgia 0.80 Oculardryness 0.83
Vascular 0.78 Oraldryness 0.92
PROFAD,total 0.89 SSItotal 0.86
Table4–SpearmancorrelationofPROFADwithPaGA,FACIT-F,ESSPRIandEQ-5D.
Variables PROFAD
Somaticfatigue Mentalfatigue Arthralgia Vascular Total
PaGA 0.60a 0.26b 0.66a 0.23 0.50a
FACIT-F 0.72a 0.43a 0.51a 0.37b 0.59a
ESSPRI
Pain 0.43a 0.13 0.66a 0.33b 0.49a
Fatigue 0.45a 0.50a 0.31b 0.21 0.46a
Dryness 0.43a 0.32b 0.42a 0.25b 0.42a
Total 0.53a 0.46a 0.58a 0.32b 0.58b
EQ-5D
Mobility 0.28b −0.03 0.35b 0.06 0.18
Selfcare 0.49a 0.22 0.31b 0.01 0.27b
Usualactivities 0.45a 0.12 0.22 0.13 0.28b
Pain/Discomfort 0.45a 0.29b 0.54a 0.23 0.43a
Anxiety/Depression 0.48a 0.33b 0.31b 0.07 0.29b
Scale1–100 −0.42a −0.06 −0.36b −0.24 −0.31b
PROFAD,ProfileofFatigueandDiscomfort;PaGA,Patient’sGlobalAssessment;FACIT-F,FunctionalAssessmentofChronicIllnessTherapy FatigueSubscale;ESSPRI,EULARSjögren’sSyndromePatientReportedIndex;EQ-5D,EuroQOL.
a P<0.001.
b P<0.05.
Surprisingly, the domain “Mental fatigue” showed a
significant but low correlation with the domain
“Anx-iety/Depression” of EQ-5D (r=0.33). However, this low
correlationwasalsoobservedbetweenthequestionsofthe domain“Mentalfatigue”oftheoriginalPROFAD-LFwiththe domains“Mentalhealth”ofSF-36(r=−0.27to−0.44),and “Psy-chologicalhealth”ofWHOQOL-BREF(r=−0.32to−0.47)and ofHospitalAnxietyandDepressionScale(HADS)(r=−0.34to 0.48).9
Thedomain“Arthralgia”ofPROFAD-SSI-SFinthis study
showedasignificantcorrelationwith“Mobility”(r=0.35)and “Pain/Discomfort”domains(r=0.54)ofEQ-5D.Thiswasalso
found in the correlation of the questions of “Discomfort”
domainoftheoriginalPROFAD-LFwith“Pain”domainof SF-36(r=0.65)and“Physicalhealth”domainofWHOQOL-BREF (r=0.62).9
In the validation study of ESSPRI, a good correlation
betweentotal scoreofPROFAD (r=0.66) andofSSI(r=0.56)
with PaGA1 was obtained – values similar to those in the
presentstudy(0.50and0.43,respectively).
As important as the objective measures is the
assess-ment of pSS symptoms focused on patient’s perception,
both with the aim todetermine the clinical improvement
in clinical trials, as to observe the progressof patients in
Table5–SpearmancorrelationofSSIwithPaGA,FACIT-F,ESSPRIandEQ-5D.
Variables SSI
Cutaneousdryness Vaginaldryness Oculardryness Oraldryness Total
PaGA 0.20 0.16 0.59a 0.444a 0.43a
FACIT-F 0.31b 0.42a 0.43a 0.611a 0.57a
ESSPRI
Pain 0.08 0.23 0.29b 0.33b 0.30b
Fatigue 0.07 0.34b 0.38b 0.46b 0.41a
Dryness 0.22 0.62a 0.35b 0.55a 0.63a
Total 0.13 0.49a 0.41a 0.57a 0.55b
EQ-5D
Mobility 0.01 0.19 0.168 0.26b 0.27b
Selfcare 0.05 0.35b 0.100 0.28b 0.29b
Usualactivities 0.22 0.33a 0.025 0.35b 0.37b
Pain/Discomfort 0.07 0.13 0.232 0.30b 0.24
Anxiety/Depression 0.05 0.16 0.197 0.37b 0.22
Scale1–100 0.107 0.02 −0.231 −0.33b −0.11
SSI,SiccaSymptomsInventory;PaGA,Patient’sGlobalAssessment;FACIT-F,FunctionalAssessmentofChronicIllnessTherapyFatigueSubscale; ESSPRI,EULARSjögren’sSyndromePatientReportedIndex;EQ-5D,EuroQOL.
a P<0.001.
Table6–SpearmancorrelationofPROFAD-SSI-SFtotal withPaGA,FACIT-F,ESSPRIandEQ-5D.
Variables PROFAD-SSI-SFtotal
PaGA 0.536b
FACIT-F 0.699b
ESSPRI
Pain 0.324a
Fatigue 0.423b
Dryness 0.441b
Total 0.545b
EQ-5D
Mobility 0.242
Selfcare 0.351a
Usualactivities 0.421b Pain/Discomfort 0.383a Anxiety/Depression 0.332a Scale1–100 −0.249
PROFAD-SSI-SF,ProfileofFatigueandDiscomfort–SiccaSymptoms Inventory(shortform);PaGA,Patient’sGlobalAssessment;FACIT-F, FunctionalAssessmentofChronicIllnessTherapyFatigueSubscale; ESSPRI,EULARSjögren’sSyndromePatientReportedIndex;EQ-5D, EuroQOL.
a P<0.001.
b P<0.05.
clinicalpractice.UnlikeESSPRI,whichhasonlythree ques-tions(domains),dilutingrelevantgroupsofsymptomsthat
are not always present together, PROFAD evaluates
differ-entaspectsoffatigue(somaticandmental)and discomfort
(arthralgia and vascular), and SSI evaluates dryness
(cuta-neous, vaginal dryness, ocular dryness, and oral dryness)
separately,andthisturnsPROFAD-SSI-SFintoamore conve-nienttooltouseinclinicalpracticeformonitoringpatients’ symptoms,andindetailingsymptomsinclinicalresearch.
Conclusion
TheBrazilianPortugueseversionofPROFAD-SSI-SF(Annex1) provedtobeanadaptable,reproducibleandvalidtoolforthe PortugueselanguageandcanbeusedintheBraziliancontext. Inaddition,thetotalscoreobtainedbyaveragingPROFAD andSSI,whichisnotcalculatedintheoriginalversion,has demonstratedinternalconsistency,reproducibilityand valid-ity.
Funding
FinancialsupportofConselhoNacionaldeDesenvolvimento
CientíficoeTecnológico(CNPq),Brazil.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
Annex
1.
Profile
of
Fatigue
and
Discomfort
–
Sicca
Symptoms
Inventory
(short
form)
(PROFAD-SSI-SF)
in
Brazilian
Portuguese
language
Porfavor,avalieoquantoosseussintomastêmestadoruinsnapiorfasenasúltimasduassemanas,assinalandoumnúmerode 0a7.
1.Amaiordificuldadequeeutivenasduasúltimassemanascomnecessidadededescanso,mesentindocansado,
exaustoouprecisandodormir:
nenhumanecessidadededescansar 0 1 2 3 4 5 6 7 tãoruimquantovocêpuder
imaginar 2.Amaiordificuldadequeeutivenasduasúltimassemanasparacomec¸arumaatividade,usandomuitoesforc¸o parafazerascoisasousentindocomoseestivesse“numabatalha”:
nenhumadificuldadeparacomec¸ar
umaatividade
0 1 2 3 4 5 6 7 tãoruimquantovocêpuder
imaginar
3.Amaiordificuldadequeeutivenasduasúltimassemanasparacontinuarumaatividade,mesentindofacilmente
exaustoousemenergia:
nenhumadificuldadeemcontinuar
umaatividade
0 1 2 3 4 5 6 7 tãoruimquantovocêpuder
imaginar
4.Amaiordificuldadequeeutivenasduasúltimassemanascomfaltadeforc¸anosmeusmúsculosoumesentindo fraco:
nenhumafaltadeforc¸a 0 1 2 3 4 5 6 7 tãoruimquantovocêpuder
imaginar 5.Amaiordificuldadequeeutivenasduasúltimassemanasemnãopensarclaramenteouachandodifícilme concentrar:
nenhumproblema 0 1 2 3 4 5 6 7 tãoruimquantovocêpuder
imaginar 6.Amaiordificuldadequeeutivenasduasúltimassemanasesquecendocoisasoucometendoerros:
nenhumproblema 0 1 2 3 4 5 6 7 tãoruimquantovocêpuder
7.Amaiordificuldadequeeutivenasduasúltimassemanascomdesconfortonosmeusmembros,como,por exemplo:desconfortooudornasgrandesjuntas(quadril,joelho,ombro)ounosmeusmúsculosoudorportodoo corpo:
nenhumproblema 0 1 2 3 4 5 6 7 tãoruimquantovocêpuder
imaginar 8.Amaiordificuldadequeeutivenasduasúltimassemanascomdorouinchac¸onosdedosoupunhos:
nenhumproblema 0 1 2 3 4 5 6 7 tãoruimquantovocêpuder
imaginar 9.Amaiordificuldadequeeutivenasduasúltimassemanascomasmãosdesconfortavelmentefrias:
nenhumproblema 0 1 2 3 4 5 6 7 tãoruimquantovocêpuder
imaginar 10.Amaiordificuldadequeeutivenasduasúltimassemanascompelesecaoucoceira:
nenhumproblema 0 1 2 3 4 5 6 7 tãoruimquantovocêpuder
imaginar
11.Amaiordificuldadequeeutivenasduasúltimassemanascomsecuravaginalcomo,porexemplo:momentosde
desconfortodurantearelac¸ãosexualdevidoàsecuravaginal:
nenhumproblema 0 1 2 3 4 5 6 7 tãoruimquantovocêpuder
imaginar
12.Amaiordificuldadequeeutivenasduasúltimassemanascomolhossensíveiscomo,porexemplo:sensac¸ãode areia,dor,queimac¸ão,coceiraouirritac¸ão.
nenhumproblema 0 1 2 3 4 5 6 7 tãoruimquantovocêpuder
imaginar 13.Amaiordificuldadequeeutivenasduasúltimassemanascomirritac¸ãonosolhoscomo,porexemplo:nos
ambientescomfumac¸a,sentindodesconfortoaovento,noarcondicionadooulugarescombaixaumidade:
nenhumproblema 0 1 2 3 4 5 6 7 tãoruimquantovocêpuder
imaginar
14.Amaiordificuldadequeeutivenasduasúltimassemanasnavisão(mesmousandoóculos)como,porexemplo, visãoembac¸ada,fraca,comlimitac¸ãoparaleitura,paraassistirtelevisãooudirigirànoite,verateladocomputador ouateladocaixaeletrônicodebancos:
nenhumproblema 0 1 2 3 4 5 6 7 tãoruimquantovocêpuder
imaginar
15.Amaiordificuldadequeeutivenasduasúltimassemanasparamealimentarcomo,porexemplo:sentiraboca seca,dificuldadeparaengoliralimentos,necessidadedeingerirlíquidosparaengolirosalimentos,alimentos entalados,necessidadederetirarrestosdealimentosdabocaouapreciarmenosacomida:
nenhumproblema 0 1 2 3 4 5 6 7 tãoruimquantovocêpuder
imaginar
16.Amaiordificuldadequeeutivenasduasúltimassemanascomgargantaounarizsecoscomo,porexemplo:boca secaquandorespirando,dificuldadeemfalarcombocaseca,necessidadedeingerirlíquidoparafalarfacilmente, sentindoonarizseco,gargantaseca,ouarcondicionadoressecandoaminhaboca:
nenhumproblema 0 1 2 3 4 5 6 7 tãoruimquantovocêpuder
imaginar 17.Amaiordificuldadequeeutivenasduasúltimassemanascommauhálitocomo,porexemplo:sentindoqueo hálitotinhacheiroruim,salivagrossa:
nenhumproblema 0 1 2 3 4 5 6 7 tãoruimquantovocêpuder
imaginar 18.Amaiordificuldadequeeutivenasduasúltimassemanascomnecessidadedelíquidoparamolharaminha bocacomo,porexemplo:levaralgoparabebernacama,precisarbeberalgoduranteanoite,acordarànoitepara urinar,ternecessidadeurgenteemurinar:
nenhumproblema 0 1 2 3 4 5 6 7 tãoruimquantovocêpuder
imaginar
nenhumproblema 0 1 2 3 4 5 6 7 tãoruimquantovocêpuder imaginar
SCORECALCULATION
PROFAD:meanof“somaticfatigue”(1+2+3+4/4)+“mentalfatigue”(5+6/2)+“arthralgia”(7+8/2)+“vascular”(9) domains
SSI:meansof“cutaneousdryness”(10)+“vaginaldryness”(11)+“oculardryness”(12+13+14/3)+“oculardryness” (15+16+17+18+19/5)
PROFAD-SSI-SFtotal:meanofPROFAD+SSI
r
e
f
e
r
e
n
c
e
s
1. SerorR,RavaudP,MarietteX,BootsmaH,TheanderE,Hansen A,etal.EularSjogren’ssyndromepatientreportedindex (Esspri):developmentofaconsensuspatientindexfor primarySjogren’ssyndrome.AnnRheumDis. 2011;70(6):968–72.
2. PatelR,ShahaneA.TheepidemiologyofSjögren’ssyndrome. ClinEpidemiol.2014;6:247–55.
3. ValimV,PereiraAM,SerranoEV,BritoFilhoOH,MussoC, CiconelliRM,etal.PrevalênciadesíndromedeSjögrenem umaregiãometropolitanadoBrasil.RevBrasReumatol. 2013;53(1):24–34.
4. FalcãoDP,MotaLMH,PiresAL,BezerraACB.Sialometria: aspectosdeinteresseclínico.RevBrasReumatol. 2013;53(6):525–31.
5. FonsecaEC,ArrudaGV,RochaEM.Olhoseco:etiopatogeniae tratamento.ArqBrasOftalmol.2010;73(2):197–203.
6. PillemerSR,SmithJ,FoxPC,BowmanSJ.Outcomemeasures forSjogren’ssyndrome.JRheumatol.2005;32(143):e9.
7. BowmanSJ,PillemerS,JonssonR,AsmussenK,VitaliC, ManthorpeR,etal.RevisitingSjogren’ssyndromeinthenew millennium:perspectivesonassessmentandoutcome measures.Rheumatology(Oxford).2001;40(1180):e8.
8. SerorR,BootsmaH,BowmanSJ,DörnerT,GottenbergJE, MarietteX,etal.OutcomemeasuresforprimarySjögren’s syndrome.JAutoimmun.2012;39(1–2):97–102.
9. BowmanSJ,BoothDA,PlattsRG.Measurementoffatigueand discomfortinprimarySjogren’ssyndromeusinganew questionnairetool.Rheumatology(Oxford).2004;43(758):e64.
10.BowmanSJ,BoothDA,PlattsRG,FieldA,RostronJ.Validation oftheSiccasymptomsinventoryforclinicalstudiesof Sjogren’ssyndrome.JRheumatol.2003;30(1259):e66.
11.BowmanSJ,HamburgerJ,RichardsA,BarryRJ,RauzS. Patient-reportedoutcomesinprimarySjögren’ssyndrome: comparisonofthelongandshortversionsoftheProfileof FatigueandDiscomfort–SiccaSymptomsInventory. Rheumatology.2009;48:140–3.
12.PaganottiMA,MiyamotoST,SerranoEV,AltoeR,ValimV. Adaptac¸ãotransculturaleavaliac¸ãodaspropriedades psicométricasdoEularSjogren’sSyndromePatientReported Index(Esspri).In:XXIXCongressoBrasileirodeReumatologia, Vitória(ES).RevBrasReumatol.2012;52:23–4.
13.NgW-F,BowmanS,GriffithsB.TheUKPSSRstudygroup relationshipbetweendiseaseactivityofprimarySjogren’s syndromeandpatientreportedoutcomeedatafroman interimanalysisoftheUKprimarySjögren’ssyndrome registry.AnnRheumDis.2011;70:510.
14.SerorR,RavaudP,BowmanSJ,MarietteX.Patients’ complaintsdependonsystemicstatusinpatientwith primarySjögren’ssyndrome.AnnRheumDis.2010;69:569.
15.GiustiE,Befi-lopesDM.Traduc¸ãoeadaptac¸ãotranscultural deinstrumentosestrangeirosparaoportuguêsbrasileiro. Pró-FonoRAtualCient.2008;20(3):207–10.
16.MachadoRIL,SoutoLM,FreireEAM.Traduc¸ão,adaptac¸ão culturalevalidac¸ãoparaalínguaportuguesa(Brasil)do SystemicSclerosisQuestionnaire(SySQ).RevBrasReumatol. 2014;54(2):95–101.
17.SerranoEV,ValimV,MiyamotoST,AltoeR,PaganottiMA, CadeNV.Adaptac¸ãotransculturaldoEularSjögren’s SyndromeDiseaseActivityIndex(Essdai)paraalíngua portuguesa.RevBrasReumatol.2013;53(6):483–93.
18.HerdmanM,Fox-RushbyJ,BadiaX.Amodelofequivalencein theculturaladaptationofHRQoLinstruments:the
universalistapproach.QualLifeRes.1998;7:323–5.
19.StreinerDL,NormanGR.Healthmeasurementscales:a practicalguidetotheirdevelopmentanduse.4thed.Oxford: OxfordUniversityPress;2008.
20.KirshnerB,GuyattG.Amethodologicalframeworkfor assessinghealthindices.JChronicDis.1985;38(1):27–36.
21.HairJF,AndersonRE,TathamRL,BlackWC.Factoranalysis. In:Multivariatedataanalysis.4thed.NewJersey:Prentice Hall;1995.
22.VitaliC,Bombardieri,JonssonR,MoutsopoulosHM, AlexanderEL,CarsonsSE,etal.Classificationcriteriafor Sjögren’ssyndrome:arevisedversionoftheEuropeancriteria proposedbytheAmerican-EuropeanConsensusGroup.Ann RheumDis.2002;61:554–8.
23.SerorR,RavaudP,BowmanSJ,BaronG,TzioufasA,Theander E,etal.EularSjogren’ssyndromediseaseactivityindex: developmentofaconsensussystemicdiseaseactivityindex forprimarySjogren’ssyndrome.AnnRheumDis.
2010;69(6):1103–9.
24.WebsterK,CellaD,YostK.TheFunctionalAssessmentof ChronicIllnessTherapy(FACIT)measurementsystem: properties,applications,andinterpretation.HealthQualLife Outcomes.2003;1:79.
25.WebsterK,OdomL,PetermanA,LentL,CellaD.The FunctionalAssessmentofChronicIllnessTherapy(FACIT) measurementsystem:validationofversion4ofthecore questionnaire.QualLifeRes.1999;8(7):604.
26.TheEuroQol,Group.EuroQol–anewfacilityforthe measurementofhealth-relatedqualityoflife.HealthPolicy. 1990;16(3):199–208.
27.MoraesCL,HasselmannMH,ReichenheimME.
Portuguese-languagecross-culturaladaptationoftheRevised ConflictTacticsScales(CTS2),aninstrumentusedtoidentify violenceincouples,vol.18.CadSaúdePública/Ministérioda Saúde,Fundac¸ãoOswaldoCruz,EscolaNacionaldeSaúde Pública;2002.p.163–76.
28.OrfaleAG,AraújoPMP,FerrazMB,NatourJ.Translationinto BrazilianPortuguese,culturaladaptationandevaluationof thereliabilityofTheDisabilitiesoftheArm,Shoulderand Handquestionnaire.BrazJMedBiolRes.2005;38: 293–302.
30.LageLV,LevyRA,CiconelliRM.Instrumentosdeavaliac¸ãoem reumatologia:importânciadesuatraduc¸ãoevalidac¸ãoem nossoidioma.RevBrasReumatol.2006;46:237.
31.GuilleminF,BombardierC,BeatonD.Cross-cultural adaptationofhealth-relatedqualityoflifemeasures: literaturereviewandproposedguidelines.JClinEpidemiol. 1993;46:1417–32.
32.ManthorpeR,AsmussenK,OxholmP.PrimarySjogren’s syndrome:diagnosticcriteria,clinicalfeatures,anddisease activity.JRheumatol.1997;24Suppl.:8–11.