ww w . r e u m a t o l o g i a . c o m . b r
REVISTA
BRASILEIRA
DE
REUMATOLOGIA
Review
article
Relevant
aspects
of
imaging
in
the
diagnosis
and
management
of
gout
夽
Eloy
De
Avila
Fernandes
a,b,
Samuel
Brighenti
Bergamaschi
a,∗,
Tatiane
Cantarelli
Rodrigues
c,
Gustavo
Coelho
Dias
d,
Ralff
Malmann
d,
Germano
Martins
Ramos
d,
Soraya
Silveira
Monteiro
a,b aHospitaldoServidorPúblicoEstadual(Iamspe),SãoPaulo,SP,BrazilbUniversidadeFederaldeSãoPaulo(Unifesp),DepartamentodeDiagnósticoporImagem,SãoPaulo,SP,Brazil cHospitaldoCorac¸ão(HCor),HCor/Teleimagem,SãoPaulo,SP,Brazil
dHospitalEstadualVilaAlpina,SãoPaulo,SP,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received29October2014 Accepted31March2016 Availableonline24June2016
Keywords:
Gout Ultrasound
Magneticresonanceimaging Dual-energycomputedtomography
a
b
s
t
r
a
c
t
Goutisaninflammatoryarthritischaracterizedbythedepositionofmonosodiumurate crystalsinthesynovialmembrane,articularcartilageandperiarticulartissuesleadingto inflammation.Menaremorecommonlyaffected,mainlyafterthe5thdecadeoflife.Its incidencehasbeengrowingwiththepopulationaging.
Inthemajorityofthecases,thediagnosisismadebyclinicalcriteriaandsynovialfluid analysis,insearchformonosodiumuratecrystals.Nonetheless,goutmaysometimeshave atypicalpresentations,complicatingthediagnosis.Inthesesituations,imagingmethods haveafundamentalrole,aidinginthediagnosticconfirmationorexcludingotherpossible differentialdiagnosis.
Conventionalradiographsarestillthemostcommonlyusedmethodingoutpatients’ evaluation;nevertheless,thisisnotasensitivemethod,sinceitdetectonlylatealterations. Inthelastyears,therehavebeenseveraladvancesinimagingmethodsforgoutpatients. Ultrasoundhasshownagreataccuracyinthediagnosisofgout,identifyingmonosodium uratedepositsinthesynovialmembraneandarticularcartilage,indetectingand character-izingtophiandinidentifyingtophaceoustendinopathyandenthesopathy.Ultrasoundhas alsobeenabletoshowcrystaldepositioninpatientswitharticularpainintheabsenceofa classicalgoutcrisis.
Computedtomographyisanexcellentmethodfordetectingboneerosions,being use-fulinspineinvolvement.Dual-energy CTisanewmethodabletoprovideinformation aboutthe chemicalcompositionof tissues,with highaccuracy in theidentification of monosodiumuratedeposits,evenintheearlystagesofthediseaseandincasesofdifficult characterization.
夽
StudydevelopedatHospitaldoServidorPúblicoEstadual“FranciscoMoratodeOliveira”(HSPE-FMO),SãoPaulo,SP,Brazil. ∗ Correspondingauthor.
E-mail:[email protected](S.B.Bergamaschi).
http://dx.doi.org/10.1016/j.rbre.2016.05.001
Magneticresonanceimagingisusefulintheevaluationofdeeptissuesnotaccessibleby ultrasound.
Besidesthediagnosis,withtheemergenceofnewdrugsthataimtoreducetophaceous burden,imagingmethodshavebecomeusefultoolsinmonitoringthetreatmentofpatients withgout.
©2016PublishedbyElsevierEditoraLtda.ThisisanopenaccessarticleundertheCC BY-NC-NDlicense(http://creativecommons.org/licenses/by-nc-nd/4.0/).
Aspectos
relevantes
do
diagnóstico
e
seguimento
por
imagem
na
gota
Palavras-chave:
Gota
Ultrassonografia Ressonânciamagnética Tomografiacomputadorizadade duplaenergia
r
e
s
u
m
o
Agotaéumaartritecaracterizadapeladeposic¸ãodecristaisdemonouratosódicona mem-branasinovial,nacartilagemarticularenostecidosperiarticularesquelevaaumprocesso inflamatório.
Namaioriadoscasosodiagnósticoéestabelecidoporcritériosclínicosepelaanálise dolíquidosinovial,embuscadoscristaisdeMSU.Porém,agotapodesemanifestarde maneirasatípicasedificultarodiagnóstico.Nessassituac¸ões,osexamesdeimagemtêm papelfundamental,auxiliamnaconfirmac¸ãodiagnósticaouaindaexcluemoutros diagnós-ticosdiferenciais.
Aradiografiaconvencionalaindaéométodomaisusadonoacompanhamentodesses pacientes,poréméumexamepoucosensível,pordetectarsomentealterac¸õestardias.
Nosúltimosanos,surgiramavanc¸osnosmétodosdeimagememrelac¸ãoàgota.O ultra-ssomsemostraumexamedegrandeacurácianodiagnósticodegota,identificadepósitos deMSUnacartilagemarticularenostecidosperiarticularesedetectaecaracterizatofos, tendinopatiaseentesopatiasportofos.
Atomografiacomputadorizadaéumótimoexameparaadetecc¸ãodeerosõesósseas eavaliac¸ãodoacometimentonacoluna.Atomografiacomputadorizadadedupla-energia, ummétodonovo,forneceinformac¸õessobreacomposic¸ãoquímicadostecidos,permitea identificac¸ãodosdepósitosdeMSUcomelevadaacurácia.
Aressonânciamagnéticapodeserútilnaavalic¸ãodostecidosprofundos,nãoacessíveis aoultrassom.
Alémdodiagnóstico,comosurgimentodedrogasquevisamreduziracargatofácea, osexamesdeimagemsetornamumaferramentaútilnoacompanhamentodotratamento dospacientescomgota.
©2016PublicadoporElsevierEditoraLtda.Este ´eumartigoOpenAccesssobuma licenc¸aCCBY-NC-ND(http://creativecommons.org/licenses/by-nc-nd/4.0/).
Introduction
Goutisaninflammatoryarthritis,characterizedbyperiodsof hyperuricemiaanddepositionofmonosodiumurate(MSU) inthearticularcartilage,subchondralbone,synovium, cap-sule,periarticulartissuesandinlowertemperatureareassuch as the superficial tissues ofthe extremities, leading to an inflammatory reaction.1,2 Genetic and dietary factors have beenimplicatedinincreasingthelevelofMSU.2
Goutoccursinapproximately0.2–0.35per100inhabitants inthegeneralpopulation.Theincidenceishigherattheend ofthethird/beginningofthefourthdecadeoflife, predom-inantlyin malesand inabout 5%of women,usually after menopause.2,3
Usually,thediagnosisisestablishedbyclinicaland labo-ratoryworkup,andthereferencemethodistheanalysisof synovialfluid;however,thisisaninvasivetechnique.Thus, thetherapycanbestartedbasedonthediagnosticcriteriaof theAmericanCollegeofRheumatology(ACR).4
Theimportanceofanaccuratediagnosisandtreatmentof goutshould notbeunderestimated,becausethesepatients willdependonthe therapythroughouttheir lives,inorder todiminishthosemorbiditiesassociatedwithhyperuricemia. Duetothemultipledifferentialdiagnoses,andalso consider-ingtheatypicalpresentationsofgout,imagingstudiesmaybe usefulinvariousstagesofthisdisease.5
Methods
Asearchofmajordatabases(Medline,Lilacs,CochraneLibrary andPubMed)wasconductedwiththeuseoftheterms“gout,” “arthritis,” “tophaceous gout” and “urate”. Our search was limitedtooriginalarticlespublishedinthelast5years,but reviewarticlesandcasereportsofsignificantclinicalrelevance wereincludedinthesurvey.
More than 700 articles and abstracts published on the proposedtopicinthis reviewwere identifiedinthe search. The selection was based on clinical importance; articles that showed norelationship with diagnostic imagingwere excluded.Attheend,39articlesremained.
Discussion
Four distinct clinical phases are recognized: asymptomatic hyperuricaemia, acute monophasic gout, recurrent acute attackswithintercriticalperiodsand chronicgoutwhichis oftentophaceous1,2(asequenceofsucheventsisnot manda-toryandasymptomaticpatientsdonotreceiveadiagnosisof gout).Ineachofthesephases,itispossibletouseimaging studies.
Thetypicalclinicalpictureofcasesofacutegoutyarthritis includesapainfulacutemonoarthritisofthefirstmetatarsal orknee,withlocalinflammationandswelling,inassociation withelevatedlevelsofuricacid.
Hyperuricemiaisoneoftheclinicalcriteriaforadiagnosis ofgout,butalthoughthisisthepredominantriskfactorfor thisdisease,elevatedserumuricacidlevelsnotalwaysresult indepositionofcrystals.6Serumuratelevels>6.8mg/dlcan leadtoprecipitationand depositionofuratecrystals inthe jointsandsofttissues,butacutegoutcrisiscanoccureven inpatients withnormalserum uratelevels; inthese cases theclinicaldiagnosisismoredifficult,andthedoctorcanget helpthroughimagingmethods.Imagingstudiescanalsobe usedinatypicalpresentations,forexample,incases involv-ingunusualagegroupsorlocations,withprolongedandless intensesymptomsatthetimeofpresentation.5,6
Theintercriticalperiodisthatfollowinganacuteepisode ofapainfulgoutcrisis,inwhichthepatientremains asymp-tomatic. Advances in imaging techniques showed that, in patients in the intercritical period, it is possible to detect tophaceousdepositsinupto50%ofjointsthathavealready beenaffectedbyacuteepisodes.5Ithasalsobeenshownthat asymptomaticpatientshadtophaceousdepositsinthespinal column,detectedbycomputedtomography(CT).7
Knowingthatinmanycasespatients,evenwhen asymp-tomatic,mayhavealreadytophaceousdepositsnotdetected clinically,itisworthquestioningwhetheritisimportantto evaluateearlyjoint,boneandtendondamage–oreventhe tophaceousload–throughimagingexamsduringthe inter-criticalphase.Atthisstage,theimagingevaluation,asfaras ourknowledgegoes,isnotyetarecommendedpracticeinthe literature.
Afewyearsmustelapsesothatthetophusbecomes clin-icallyevidentafter thefirst attack; rarelythese formations areidentifiedduringthisfirstepisode.1Chronictophaceous
goutischaracterizedclinicallybythepresenceoftophi,which areformedsecondarilytotheaccumulationofuricacid, pro-teinmatrix,inflammatorycellsandforeignbodygiantcellsin thetendons,ligaments,cartilages,bursae,subcutaneous cel-lulartissueandinperiarticularregions.5Thetophiaremore commonontheextensorsurfacesofthehands,elbows,feet, knees,auricularappendagesandthetipofthenose.
Althoughgenerallychronictophaceousgouthasa straight-forward clinicaldiagnosis,insomecasesthediagnosis can bechallenging,whentheproblemisassociatedwithunusual symptomsorwithanatypicaldisease.Atypicalclinical mani-festationsareseenmorefrequentlyincertainsegmentsofthe population, includingtheelderly,patientswhohave under-goneorgantransplants,andcancerpatients.6
Thetophinodulescanalsonotbetypical,andthe physi-cian should take into account some differential diagnosis asganglion,cysts,bursitis,hematoma,amyloidosis, throm-bophlebitis, sarcoidosis, psoriatic arthritis, pyrophosphate depositarthritis,neoplasms,tenosynovitis,rheumatoid nod-ules,osteoarthritisandinfection.8,9
At this stage, the imagingstudies may be validfor the assessmentofdiseaseseverity,theextentofMSUdeposition, andthepresenceofchronicinflammation.Moreover,images mayconstituteausefultoolformonitoringtheresponsetoa reductionofuricacidtherapy.10
Monitoring
and
response
to
treatment
Severalmethodshavebeendevelopedandevaluatedbythe OutcomesMeasuresinRheumatology(OMERACT)inorderto quantifytophiinpatientswithchronictophaceousgout,from thesimplestmethods,forinstance,byphysicalexamination, tomoresophisticatedmeanswiththeuseofimagingtests. Inpatientswithachronictophaceousgout,quantificationof tophi and documentation ofregression withtreatment are importantmeasuresofmonitoring,inordertopreventjoint destruction.Patientswithelevatedlevelsofuricaciddonot necessarily exhibithigher masses oftophi, comparedwith patientswithlowlevelsofuricacid.11,12
Amongthemethodsusingphysicalexamination,themain onesare number oftophicounting, physicalmeasurement withameasuringtape,theuseofaspecificinstrumentcalled Verniercaliper,anddigitalphotography.13
Ultrasoundisagoodtoolfortheevaluationofresponseto treatment,thankstoitsavailabilityand alsobecauseithas goodsensitivity.Theuseofcomputedtomographyand mag-neticresonanceimaging,althoughlessavailabletechniques, resultsinsomeadvantages,suchasdatastoragepossibility forlaterreadingandvisualizationofintra-articulartophi,in theabsenceofsubcutaneoustophi.14–16
toclinicaltrialsofnewtherapeuticagents,withlittleusein clinicalpractice.16
Image
methods
Theimagingmethodsusedfortheevaluationofgoutareplain radiography(X-ray),ultrasound(US),DECT,CTandmagnetic resonanceimaging(MRI).
Plainradiography
Blochetal.2classifiedtheradiologicalfindingsinearly, inter-mediate and latechanges. Radiographicchanges are more frequent in the feet, especially in the first metatarsopha-langealjoint.2,10 Intheinitialpresentationofgout, thereis nospecific radiographic signs –only anincreasedsoft tis-suevolumeanddensity.WithX-rays,onecannotassessearly changesofthesofttissuessuchaseffusion,initialerosion,
synovial hypertrophy, hypervascularization,or small tophi. Ontheother hand,withMRI–whichshowsthesechanges – is not always that the physician can establish an accu-ratedifferentiationbetweengoutandsomeofitsdifferential diagnosis.
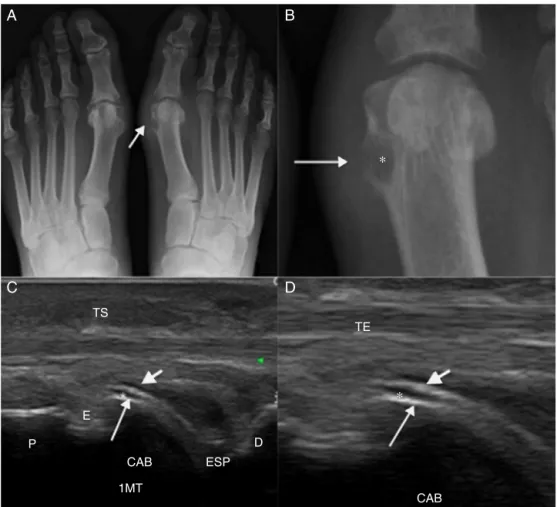
X-rayisafastmethod,andgenerallyitisusedinthefirst investigationofgout.X-rayhaslowsensitivityfordiagnosis, and theremay beagap from 6to12years forthe finding ofradiologicalevidence.2In2008,Rettenbacheretal.found asensitivityof31–55%andaspecificityof93%forX-rayin thediagnosisofgout.17Incasesofchronictophaceousgout, radiographicsignsincludevisualizingtophiassofttissueor intraosseousmasses,whetherornotcontainingcalcifications; andthepresenceofanon-demineralizingarthropathy accom-paniedbyerosionspresentingmarginswhichmaybesclerotic orprotruding.TheMartel’ssign(Fig.1)consistsinthepresence ofaprotruding, salientboneedgeseparated fromatophus and leaningonit.18,19 InX-ray,generallythe joint spaceis preserveduntilthediseaseisadvanced.10
A
C
B
D
TS
P
E
1MT CAB
TE
ESP
CAB D
∗
∗
AccordingtoBloch,16X-rayisamethodwithlittleusein theevaluationoftreatment,primarilybyitslowsensitivityin detectingthediseaseinitsearlystages,butalso,becauseit isbasedonthepresenceoflatefindings,suchasincreasesin softtissue,corticalerosions,andlyticinjuries.
Ultrasound
ThehighresolutionofUS allowsustoidentifythevarious formsofpresentationofgoutand itsrelationshipwiththe differenttissues,helpinginanearlyand noninvasive diag-nosis,therapeuticdecisionandmanagementoftreatment.In addition,USisaveryusefulmethodofassessingtheextent andmeasurementoflesionsandintheinvolvementof adja-centstructures,withlowinterobservervariability;besides,US fulfillthenecessarycharacteristicsfortheevaluationof ther-apeuticresponse.Asforhistopathologicconfirmationofgout, USisalsousefulinguidingpuncturesorbiopsies.19,20
USdetectsearlychangesinthesofttissuesincasesofgout andcanbeusedespeciallywhentheclinical,laboratoryand radiographicstudiesarenegativeorinconclusive.Thereare severalotheradvantageswiththeuseofthistechnology:itis noninvasive,easytorepeattheexam,abilitytodifferentiate betweensolidversuscysticlesions,lowcost,patientcontact, absenceofionizingradiation,thepossibilityofacquisitionof multiplanarandhigh-resolutionimages,dynamicassessment ofthejointandtendons,andeffectiveguidanceininvasive procedures.9,19,21Somestudiescomparingthesensitivityand specificityofUSversusX-rayshowedthatUSismore sensi-tiveandearlierthatX-ray,becausethesonographicchanges arepresentatearlierstages,incomparisontotypicalX-ray signals.17,19,22
Sonographicaspectswhicharecharacteristicforthe diag-nosisofgoutarethe“doublecontoursignal”(Fig.1),whichis characterizedbyanirregularlinearhyperechoiclayeronthe superficialmarginoftheanechoichyalinecartilageand par-alleltothebonecortex,withoutaposterioracousticshade. InaThiele’s2007study,19thissignalwasobservedin92%of patientswithbiopsy-confirmedgout,andnoneofthecontrols. Butpatientswithasymptomatichyperuricemiamayshow thesignofthedoublecontour,observedinthestudyofPineda etal.in25%ofthemetacarpophalangealjoints.20Moreover,
withthereductionofuricacidlevels,thissignaltendsto dis-appearinupto7months.22
In2008,Rettenbacheretal.17foundsensitivityand speci-ficityofrespectively80%and75%forthebrighthyperechoic foci in synovial tissue (microtophi) and 79% and 95% for hyperechoicareasforthediagnosisofgout.Consideringthe presenceofeitherofthetwofindings,ultrasoundhada sensi-tivityof96%andspecificityof73%.Thespecificityisnothigher because punctate hyperechoic foci, which may represent microtophi, canalsobeobservedincasesofosteoarthritis, rheumatoidarthritisandchondrocalcinosis.23
Whenthebrighthyperechoicfociareseenincombination withthedoublecontoursignal,thespecificityreaches100%, butwithaconsiderablereductionofsensitivity.24
Incaseswherethedoctordependsonanimagingstudy, theknowledgeofthesonographicfeaturesoftophusis impor-tantsothatonecandifferentiatebetweentophaceousnodules andnodulesfromotheretiologies(Fig.2).USusescriteriathat helptodifferentiatebetweennodulescausedbycancerand inflammatory and infectious processes. In 2003,Nalbant,10 comparing tophinodulesandrheumatoid nodules,showed that 80% of tophi were heterogeneous and of these, 75% werehyperechoic;furthermore,thisauthorobservedonly15% ofheterogeneityandhyperechogenicityinrheumatoid nod-ules.Therheumatoidnoduleismorehomogeneousandcan present a well-defined, hypoechoiccentral area dueto the occurrenceofnecrosis.Sincerheumatoidnodulesrarely cal-cify,thisfeaturealsohelpstodifferentiatetheseformations fromtophi,whichcancalcify.
Thepresenceofcalcificationswithposterioracoustic shad-owing and irregularities ofthe cortical bone underlyingto nodulesalsofavoradiagnosisoftophi.Thereisnocorrelation betweendiseasedurationandthepresenceofcalcifications in tophi. The tophi are hyperechoic in 96.3% and hypoe-choicin3.7%ofcases.12Thehyperechogenicitynotedintophi represents deposits of urateor calcifications. Small hyper-echoic particles,or brightfoci, measureless than 1mmin sizeandrepresentsynovialmicrotophi.17Anagglomerateof microtophi form hyperechoic the tophus; therefore, hyper-echogenicity and heterogeneity are strong indicators of a tophus.
Thehypoechoichalointophusperipheryisahypoechoic bandobservedpartiallyorcompletelyaroundthetophusand
p
cb
5
D
B
A
tr
mtt
maycorrespondtoinflammation,fibrosisoredema.Thishalo isobservedinlargepartsoftophiand canbeamarkerfor tophaceousnodule.12
Thesonographicfeaturesoftophiinrelationtothetendons mayhelptoexplaintheclinicalpicturesshowingmovement restrictionsofpatientswithchronictophaceousgout; further-more,theirknowledgecanavoidinvasiveprocedures,suchas biopsies.Afive-typeclassificationoftherelationshipbetween tophiandthetendoninpatientswithchronictophaceousgout wasproposed,25basedontheirlocation:tendonsurrounded bytophi,norelationshipbetweentophusandtendon,tophiin thetendoninsertionsite(enthesopathy),extrinsic compres-sion,andtophiwithinthetendon.
Enthesopathysecondarytotophiisarecentfindinginthe literature;andalthoughithasbeendescribedinonly7%of casesofchronictophaceousgout,thispossibilityshouldbe considered in the differential diagnosis, depending on the clinical context.Thedifferential diagnosis ofenthesopathy isbroadandincludescalciumpyrophosphatedeposition dis-ease,degenerativedisease,acromegaly,hyperparathyroidism, hypoparathyroidism,andrheumatoidarthritis,amongothers. Theintratendinoustophustendstoevolvetotheruptureof thetendon26andanearlysonographicdiagnosismayassist the physician in establishing an effective treatment while avoidingdamagethat, ifnottreated medically,may evolve intotheneedforsurgicaltreatment. Insidethetendon,the tophimayhavemicrodepositsdemonstratedbybright,ovoid andhyperechoicspots.Chronicintratendinoustophicanbe evidencedashyperechoic bands,occasionally accompanied byaposterioracousticshadow.27
USallowsthevisualizationofthechangesinthe inflamma-toryprocessofgout.TheevaluationofgoutbycolorDoppler
US demonstrates increasedflow inthe acute phaseof the podagracrisis,whichnormalizespartiallyin7days. Gener-ally,colorshowsnoflowwhenthepatientisoutofthegout crisis,10butintheauthor’spracticeithasbeenobservedthat painfulperiarticularareasinpatientswithaknowndiagnosis ofgoutcanexhibithyperechoictophiandflowwithDoppler, evenwithoutthe classicsignsofcrisis,whichexplainsthe atypicalclinicalpicturesofgoutyarthralgia.However,these findingsdependonfurtherstudiestojustifytheiruse(Fig.3). Bone erosions are defined as cortical discontinuities observedintwoperpendicularplanes.Thisisalatefinding andhaslowsensitivityindiagnosingtophaceousgout; how-ever,theseerosionsareslightlybetterdetectedbyUSversus X-ray(24%versus20%ofcases).17
Thedimensionsofthegoutytophusassumeimportance in assessingthe responseto treatment; therefore,in order toacquirepracticalutility,themethodusedforthispurpose must showagood reproducibility.Perez-Ruizet al.showed thatUSisabletodetect allperiarticulartophiidentifiedby MRI.10,28Afterthepublicationofthesestudies,OMERACThas consideredUSasapossiblyusefulmethodinmeasuringgouty tophi;butnewclinicaltrialsmustbeconductedforvalidation ofthismethod.
Computedtomography(CT)
CTallowsthevisualizationoftophiinboththesubcutaneous tissueandinintra-articularareas.Thismethodalsohelpsto identifyboneerosion,beingmoresensitivethanX-rayandMRI forthispurpose.AsystematicanalysisshowedthatCTdetects thepresenceofintraosseoustophiin81%ofjointspresenting
T
A
B
T T
Erosoes
Terosionandin100%ofcaseswhentheerosionisgreaterthan 7.5mm.29
CTcanalsorevealMSUdepositsinsidetophi,thankstoits attenuationcloseto160UH,comparedtocalciumdeposits, whichhave higherattenuation, around 450UH.30 Thus,CT canassistinthedifferentiationwithrespecttoothertypesof softtissuenodules.CTcanbeusedasacomplementary imag-ingmethodintheevaluationofdamagetodeeperstructures, whenmethodsthatdonotuseionizingradiationcouldnot confirmitspresence.Incasesofinvolvementofdeep struc-turessuchasthespine,possiblyCTcancomplementanMRI study,bydemonstratingmassesoftophiwithcompressionof nervestructures.
Whileitmaybeuseful,CTisnotrecommendedasamethod ofchoiceintheevaluationofgoutonsurfacestructuresdue totheionizingradiationexposure.
Dual-energycomputedtomography(DECT)
DECTisamethodthatprovidesinformationaboutthe chem-icalcompositionoftissuesand allowstheirdifferentiation. Throughthistechnique,itispossibletodistinguishMSU crys-tals from gout, bone or dystrophic calcification.6 In some studiesexaminingthe depositionofarticularand periartic-ularcrystalsusingDECT,highsensitivityandspecificitywere observed.Variationsonthedataobtainedcouldbeduetothe differentjointsevaluated,protocolsusedanddifferentstages ofthisdisease.30–34
IntheearlystagesofthediseasewhentheMSUdeposits aremicroscopicandwithanintra-articularlocation,notbeing macroscopictophi,DECTmaynotbeabletodetectthem,since this techniquehasa sizethreshold, usuallyof2mm –the techniqueisnotabletodistinguishdepositswithdimensions belowthislimit.35Inadditiontothelimitationsrelatedtothe sizeofdeposits,DECTissusceptibletosomeartifacts,mainly relatedtometallicdevices,calluses,nailsandthickenedskin areassuchasthatintheheel.
Most studies did not explore the diagnostic accuracy of DECT in the first presentation ofgouty arthritis. Based on limited published data, in the first episode of gouty
arthritis, thediagnostic sensitivityislow, around50%.36 In astudyof21patientscomparingthediagnosticaccuracyof US versus DECTincaseswith suspectedgout, the authors observedsimilarsensitivityinthesetwomethods,with false-negativeresultsinDECTaccuratelydetectedwithUS.16
DECT may be used toassess gout, regardless of serum uricacidlevels,andcanconfirmthediseaseinpatientswith normalserumuricacidlevels,orexcludeitinpatientswith hyperuricemia. The generalburdenor uric acid deposition volumemaybecalculatedonindividuallesions,joints,orin theentirelydigitalizedarea.ApossibledisadvantageofDECT couldbethepatient’sexposuretoionizingradiation,butthe doseislessthanthenaturalannualdosereceivedandmuch lowerthanthepotentiallymalignancy-inducingvalues.22Due tothecostandtotheradiationexposure(albeitlow),itsmain role,monitoringthetreatment,islimitedtoclinicaltrialsof newtherapeuticagents,morethaninclinicalpractice.16
Magneticresonance
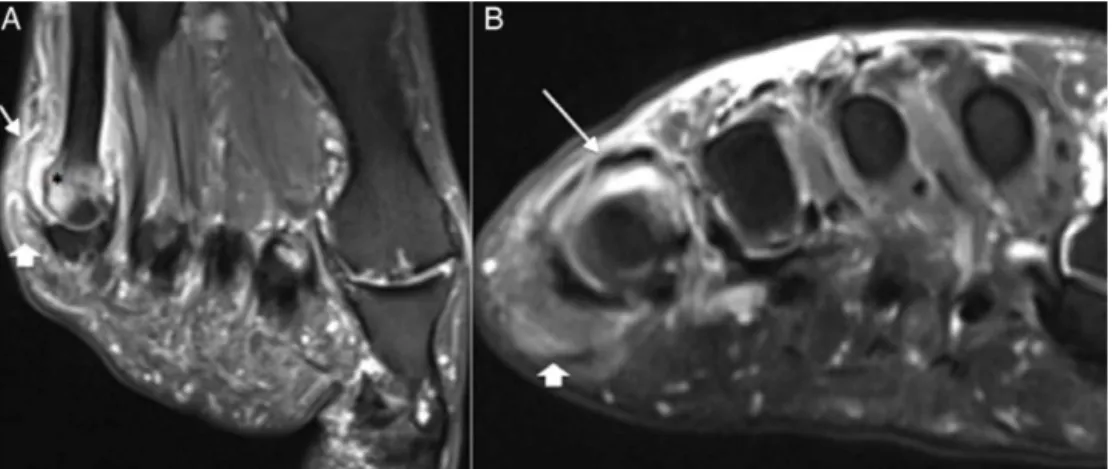
MRIisnotroutinelyusedtoevaluatetophaceousgout.This techniquecanbeusedtorecognizethecauseofmotion lim-itations,motororpainfuldisorderssecondarytochangesin deepstructuresorthatarecoatedwithbone;insuchevent, theaccessthroughUSisnotsuitable(Fig.4).
MRIisalsousefulforassessingthedifferentialdiagnosis of softtissue masses on the extremities.31 In MRIstudies, usuallythetophuspresents itselfasajuxta-articularmass of soft tissue, causing periarticular erosions and synovial thickening.Usingthistechnique,theappearanceoftophiis variable.InT2-weightedsequence,thereisavariationofa low-to-high signal, with a homogeneous or non-homogeneous pattern, depending on the degree of hydration and calci-fication. The most common appearance in a T2-weighted sequenceisalow-to-intermediate,heterogeneoussignal.In a T1-weighted sequence, its appearance is more consis-tent, generallyexhibiting alow-tointermediate signal.The enhancementpatternisalsovariable.Anotherfeatureisthe enhancementsurroundingthetophus;probablythisisrelated totheadjacentgranulationtissue(Fig.5).37
Fig.4–MRIimagesofthefootonthelong(A)andshort(B)axis,showingjointeffusion(arrow)inthefifth
Fig.5–SagittalMRIimages:T1-weighted(A),T2-weighted(B),axialT2-weighted(C)andsagittalaftercontrast(D)imagesof thefootshowingheterogeneousmaterialwithisosignalonT1,heterogeneoushyperintensesignalonT2,with
heterogeneousenhancementafterIVinjectionofparamagneticcontrastinthedorsalaspectofthefoot(arrows).
MRIalsoprovidesinformationonthemorphologyoftophi, which can range from small nodular masses deposited in poorly defined anatomical planes, evenwith a permeative aspect.38
Perez-Ruizetal.evaluatedthe measurementoftophi in acomparisonbetweenUSandMRI,includingthechangein tophussizeanditsassociationwithserumconcentrationsof urateinthecourseof12months.Inthisstudy,thediameters evaluatedinMRIwerelargerthanthoseobtainedwiththeuse ofUS,andthismayberelatedtobetterMRIimagingforthesoft tissuecomponentofthetophus,whichcancontainregionsof inflammationandhypervascularity.10
RecentstudieshaveshownthatMRIcandetectearly artic-ularerosionsthatarenotradiographicallyapparent.39Faced withanacutegoutyarthritis,it iscommonthe occurrence ofperiarticularedema,synovitis,andjointeffusion,aswellas thepresenceofahighsignalinbonemarrowandperiarticular softtissues;butthesechangescanbeseeninany inflamma-toryarthropathy,thusbeingunspecificfindings.MRIisnota relevanttechniquetohelpintheestablishmentoftheinitial diagnosisofgout.Forthesereasons,MRIshouldnotbe rou-tinelyusedforthediagnosisofgoutinitsearlystages,bothin casesoftypicalclinicalpresentation,asinatypicalcases.
Conclusion
Imagingmethodscanbeuseful toaidinthediagnosis and monitoringoftreatmentofpatientswithgoutand,in partic-ular,theuseofultrasound,whichisanavailable,noninvasive toolwhichprovidesgoodresults.Thepotentialofultrasound diagnosishasincreasedtheinterestamongphysicians.This isareadilyavailable,non-invasivetoolthatcanbeusedfor theinvestigationofthisdisease.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1.ChenCK,YehLR,PanHB,YangCF,LuYcResnickD.
2. SmithEUR,Díaz-TornéC,Perez-RuizF,MarchLM. Epidemiologyofgout:anupdate.BestPractResClin Rheumatol.2010;24:811–27.
3. MalikA,SchumacherHR,DinnellaJE,ClayburneGM.Clinical diagnosticcriteriaforgout:comparisonwiththegold standardofsynovialfluidcrystalanalysis.JClinRheumatol. 2009;15:22–4.
4. MonuJU,PopeTLJr.Gout:aclinicalandradiologicreview. RadiolClinNAm.2004;42:169–84.
5. DesaiMA,PetersonJJ,GarnerHW,KransdorfMJ.Clinical utilityofdual-energyCTforevaluationoftophaceousgout. Radiographics.2011;31:1365–75.
6. HelitoPVP,DeMelloFM,FullerR,Goldensteim-SchainbergC, HalpernARS,Bordalo-RodriguesM,etal.Tomografia computadorizadadecolunaemumapopulac¸ãocomgota. TemaLivreJPR.2013.
7. DunganDH,SeegerLL,GrantEG.Casereport707: hemorrhagicBaker’scystoftherightcalf.SkeletRadiol. 1992;21:52–5.
8. AbrahamssonSO.Goutytenosynovitissimulatingan infection.Acasereport.ActaOrthopScand.1987;58:282–3.
9. Perez-RuizF,DalbethN,UrresolaA,DeMiguelE,Schlesinger N.Gout.Imagingofgout:findingsandutility.ArthritisRes Ther.2009;11:232.
10.NalbantS,CorominasH,HsuB,ChenLX,SchumacherHR, KitumnuaypongT.Ultrasonographyforassessmentof subcutaneousnodules.JRheumatol.2003;30:1191–5.
11.deÁvilaFernandesE,KubotaES,SandimGB,MitraudSA, FerrariAJ,FernandesAR.Ultrasoundfeaturesoftophiin chronictophaceousgout.SkeletalRadiol.2011;40:309–15.
12.DalbethN,SchauerC,MacdonaldP,Perez-RuizF,Schumacher HR,HamburgerS,etal.Methodsoftophusassessmentin clinicaltrialsofchronicgout:asystematicliteraturereview andpictorialreferenceguide.AnnRheumDis.
2011;70:597–604.
13.DalbethN,MerrimanT.Mechanismsofboneerosioningout. Aquantitativeanalysisusingplainradiographyand computedtomography.AnnRheumDis.2009;68:1290–5.
14.SchumacherHRJr,BeckerMA,EdwardsNL.Magnetic resonanceimaginginthequantitativeassessmentofgouty tophi.IntJClinPract.2006;60:408–14.
15.DalbethN.Clinicalapplicationsofdual-energycomputed tomographyforrheumatology.Rheumatologist.2014.
16.BlochC,HermannG,YuTF.Aradiologicreevaluationofgout: astudyof2,000patients.AmJRoentgenol.1980;134:781–7.
17.RettenbacherT,EnnemoserS,WeirichH,UlmerH,HartigF, KlotzW,etal.Diagnosticimagingofgout:comparisonof high-resolutionUSversusconventionalX-ray.EurRadiol. 2008;18:621–30.
18.MartelW.Theoverhangingmarginofbone:aroentgenologic manifestationofgout.Radiology.1968;91:755–6.
19.ThieleRG,SchlesingerN.Diagnosisofgoutbyultrasound. Rheumatology(Oxford).2007;46:1116–21.
20.PinedaC.Jointandtendonsubclinicalinvolvement
suggestiveofgoutyarthritisinasymptomatichyperuricemia: anultrasoundcontrolledstudy.ArthritisResTher.2011;13:R4.
21.Balbir-GurmanA,NahirAM,Braun-MoscoviciY,SoudackM. Sonographicfeaturesofatophaceousnodule.IsrMedAssocJ. 2005;7:746–7.
22.ThieleRG,SchlesingerN.Ultrasounddetectsmoreerosions ingoutthanconventionalradiography.AnnRheumDis. 2010;69Suppl.3:612.
23.LaiKL,ChiuYM.Roleofultrasonographyindiagnosinggouty arthritis.JMedUltrasound.2011;19:7e13.
24.WrightSA,FilippucciE,McVeighC,GreyA,McCarronM, GrassiW,etal.High-resolutionultrasonographyofthefirst metatarsalphalangealjointingout:acontrolledstudy.Ann RheumDis.2007;66:859–64.
25.FernandesEA,SandimGB,MitraudSAV,KubotaES,Ferrari AJL,FernandesARC.Sonographicdescriptionand
classificationoftendinousinvolvementinrelationtotophiin chronictophaceousgout.InsightsImaging.2010;1:143–8.
26.LagoutarisED,AdamsHB,DiDomenicoLA,RothenbergRJ. Longitudinaltearsofbothperonealtendonsassociatedwith tophaceousgoutyinfiltration.Acasereport.JFootAnkle Surg.2005;44:222–4.
27.GrassiW,MeenaghG,PascualE,FilippucciE.Crystal clear-sonographicassessmentofgoutandcalcium pyrophosphatedepositiondisease.SeminArthritisRheum. 2006;36:197–202.
28.Perez-RuizF,MartimI,CanteliB.Ultrasonographic measurementoftophiasanoutcomemeasureforchronic gout.JRheumatol.2007;34:9.
29.DalbethN,CollisJ,GregoryK,ClarkB,RobinsonE,McQueen FM.Tophaceousjointdiseasestronglypredictshandfunction inpatientswithgout.Rheumatology.2007;46:1804–7.
30.DalbethN,ClarkB,GregoryK,GambleGD,DoyleA,McQueen FM.Computedtomographymeasurementoftophusvolume: comparisonwithphysicalmeasurement.Arthritis
Rheumatol.2007;57:461–5.
31.GlazebrookKN,GuimaraesLS,MurthyNS.Identificationof intraarticularandperiarticularuricacidcrystalswith dual-energyCT:initialevaluation.Radiology.2011;261:516–24.
32.NicolaouS,Yong-HingCJ,Galea-SolerS,HouDJ,LouisL, MunkP.Dual-energyCTasapotentialnewdiagnostictoolin themanagementofgoutintheacutesetting.AmJ
Roentgenol.2010;194:1072–8.
33.ChoiHK,BurnsLC,ShojaniaK.DualenergyCTingout:a prospectivevalidationstudy.AnnRheumDis.
2012;71:1466–71.
34.GruberM,BodnerG,RathE,SuppG,WeberM,
Schueller-WeidekammC.Dual-energycomputedtomography comparedwithultrasoundinthediagnosisofgout.
Rheumatology(Oxford).2014;53:173–9.
35.GlazebrookKN,KakarS,IdaCM,LauriniJA,ModerKG,LengS. False-negativedual-energycomputedtomographyina patientwithacutegout.JClinRheumatol.2012;18:138–41.
36.MangerB,LellM,WackerJ.Detectionofperiarticularurate depositswithdualenergyCTinpatientswithacutegouty arthritis.AnnRheumDis.2012;71:470–2.
37.KhooJN,TanSC.MRimagingoftophaceousgoutrevisited. SingapMedJ.2011;52:840.
38.McQueenFM,DoyleA,DalbethN.Imagingingout–whatcan welearnfromMRI,CT,DECT,andUS?ArthritisResTher. 2011;13:246.