* Corresponding author:Hideo Onishi, Program in Health and Welfare, Graduate School of Comprehensive Scientific Research, Prefectural University of Hiroshima, 1-1 Gakuenmachi, Mihara, Hiroshima, Japan 723-0053. Tel: +81-848-60-1268; Fax: +81-848-60-1186; E-mail: onisi@pu-hiroshima.ac.jp
© 2013 mums.ac.ir All rights reserved.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/3.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Differential
impact
of
multi
focus
fan
beam
collimation
with
L
mode
and
conventional
systems
on
the
accuracy
of
myocardial
perfusion
imaging:
Quantitative
evaluation
using
phantoms
(ideo Onishi
,, Norikazu Matsutomo , Yoshiharu Kangai ,Tatsunori Saho ,
(izuru Amijima
Program in (ealth and Welfare, Graduate on School of Comprehensive Scientific Research, Prefectural University of (iroshima
Program in Biological System Sciences, Graduate on School of Comprehensive Scientific Research, Prefectural University of (iroshima
Department of Radiology, Kurashiki Central (ospital Department of Nursing,(yogo University of (ealth Sciences
A R T I C L E I N F O
Article type:
Original article
Article history:
Received: ‐Jun‐ Revised: ‐Jul‐ Accepted: ‐Jul‐
Keywords:
Multi‐focus fan beam Collimator
)mage quality Acquisition mode
A B S T R A C T
Objective(s): A novel )Q‐SPECTTM method has become widely used in clinical studies. The present study compares the quality of myocardial perfusion images MP) acquired using the )Q‐SPECTTM )Q‐mode , conventional ° apart: C‐mode and L‐ mode ° apart: L‐mode systems. We assessed spatial resolution, image reproducibility and quantifiability using various physical phantoms.
Methods: SPECT images were acquired using a dual‐headed gamma camera with C‐ mode, L‐mode, and )Q‐mode acquisition systems from line source, pai and cardiac phantoms containing solutions of mTc. The line source phantom was placed in the center of the orbit and at ± . , ± . , ± . , ± . and ± . cm off center. We examined quantifiability using the pai phantom comprising six chambers containing . , . , . , . , . , and . MBq/mL of m‐Tc and cross‐calibrating the SPECT counts. )mage resolution and reproducibility were quantified as myocardial wall thickness MWT and %uptake using polar maps.
Results: The full width at half maximum FW(M of the )Q‐mode in the center was increased by % as compared with C‐mode, and FW(M in the periphery was increased % compared with FW(M at the center. Calibrated SPECT counts were essentially the same when quantified using )Q‐and C‐modes. )Q‐SPECT images of MWT were significantly improved P< . over L‐mode, and C‐mode SPECT
imaging with )Q‐mode became increasingly inhomogeneous, both visually and quantitatively C‐mode vs. L‐mode, ns; C‐mode vs. )Q‐mode, P< . .
Conclusion: Myocardial perfusion images acquired by )Q‐SPECT were comparable to those acquired by conventional and L‐mode SPECT, but with significantly improved resolution and quality. Our results suggest that )Q‐SPECT is the optimal technology for myocardial perfusion SPECT imaging.
Please cite this paper as:
Onishi (, Matsutomo N, Kangai Y, Saho T, Amijima (.
Differential impact of multi‐focus fan beam collimation with L‐mode and conventional systems on the accuracy of myocardial perfusion imaging: Quantitative evaluation using phantoms. Asia Oceania J Nucl Med Biol. ; : ‐ .
Introduction
Myocardial ischemia, infarcts and viability are widely assessed using myocardial perfusion imaging MP) using single photon emission computed tomography SPECT . Nuclear cardiology pioneers have developed several new technologies with which to perform myocardial perfusion SPECT. Until recently, myocardial
Multi-focus fan beam collimation Onishi H et al
Asia Oceania J Nucl Med Biol. 2013; 1(2):28-34. 29
optimized acquisition geometry, collimator design and associated reconstruction software to alleviate this problem.
Advances in hardware and software have reduced imaging or dose duration and improved patient comfort as well as image quality. With respect to software, Jinghan et al reported
that matched filtering improves SPECT resolution, and (ughes et al and Onishi et al
validated image quality using a novel resolution recovery method in phantom studies. Myers et al , DePuey et al , and Ali et al
showed that these reconstruction methods clinically reduced the amount of time needed for acquisition and the radiation dose. With respect to hardware, several cameras have increased photon sensitivity achieved via customized detector geometry and innovative collimation design , , focusing the field of view on the heart and using solid state detectors . Optimized geometry for cardiac imaging has significantly reduced or totally eliminated detector motion, with the simultaneous collection of counts from the entire heart. The )Q‐SPECTTM has a combined astigmatic collimator SMARTZOOM , optimized organ‐of‐interest centered acquisition and iterative reconstruction with CT‐based attenuation correction, and it has been applied in clinical studies. Corbette et al found in a single–center clinical trial that )Q‐ SPECTTM provided better quality images than conventional SPECT.
The current study quantifies the differential impact of )Q‐SPECTTM )Q‐mode conventional ° apart: C‐mode and L‐mode ° apart: L‐ mode systems in myocardial perfusion imaging, as well as the effect of spatial resolution, image reproducibility and quantifiability using various physical phantoms.
Materials and Methods
Study design
This study qualitatively compares the quality of myocardial perfusion images using a dual‐headed camera with C‐mode, L‐mode, and )Q‐mode systems. We evaluated image quality in terms of spatial resolution, image reproducibility and quantifiability using line source, pai, and myocardial torso phantoms, respectively.
Phantom study Line source phantom
Since the tomographic spatial resolution of gamma cameras is often analyzed using acquisition with small line sources capillary tubes placed in air, we also performed capillary tube experiments. Spatial resolution was evaluated at full width at half maximum FW(M determined from an in‐house line source
phantom. Eleven capillary tubes mm long and mm in diameter containing Tc‐ m – MBq per tube were positioned on a Styrofoam support on the camera scanning bed parallel to the axis of rotation of the camera in the center of the orbit and at ± . , ± . , ± . , ± . and ±
cm off center.
Pai phantom
A pai phantom shaped like a pie‐chart divided into six chambers was symmetrically positioned in a cylinder mm in diameter and mm in length. Each chamber contained mL of homogeneous solutions of . , . , . , . , . , and . MBq/mL mTc. Linear regression was then analyzed between the actual and measured radioactivity activity concentrations image quantitation .
Myocardial torso phantom
The conditions of capillary tube experiments do not realistically represent those of patients. Thus, we used a myocardial torso phantom to more closely model patient scans with photon attenuation and scatter in a non‐uniform medium, while maintaining control over the true activity distribution. The commercially available, model R( elliptical cylinder torso phantom mm wide and mm thick has simulated bone, lung, mediastinum, hepatic, and myocardial regions Kyoto Kagaku Co. Ltd., Kyoto, Japan . The mediastinum, left ventricle, right ventricle and hepatic areas were filled with water. Assuming that MBq of mTc tracer was administered, the myocardial area would contain . KBq/mL. The phantom also contained a ‐ mL insert modeling a healthy myocardium
without perfusion defects .
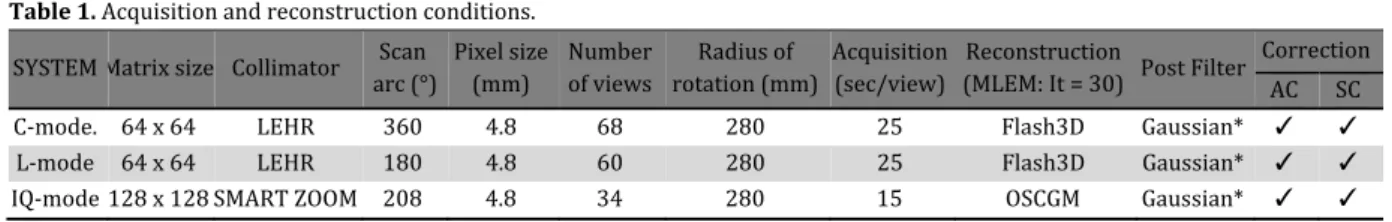
Image acquisition and processing
Datasets were acquired during SPECT studies using Symbia T SPECT/CT dual‐headed gamma cameras Siemens AG, Erlangen, Germany equipped with a low energy high resolution LE(R collimator FW(M . mm at the center and with )Q‐SPECT modification using SMARTZOOM collimators FW(M . mm at the center . The radius of the circular orbit was mm in both systems and saved to a SMARTZOOMTM system. C‐mode and L‐mode systems were equipped with LE(R collimators. C‐model and L‐modes both comprised over
°contoured orbits with and views per detector for sec in x matrices and . ‐ mm pixels. )Q‐mode images were acquired over
Onishi H et al Multi-focus fan beam collimation
30 Asia Oceania J Nucl Med Biol. 2013; 1(2):28-34.
were reconstructed on a Syngo M) Workplace Siemens . The SPECT data were reconstructed using ordered subset expectation maximization OSEM with depth‐dependent D resolution recovery Flash DTM, and with ordered subset conjugates‐gradient minimization OSCGM for SMARTZOOM collimators. Subsets and iterations were and , respectively. Both images were attenuation‐corrected based on images acquired immediately before SPECT with the integrated CT scanner. Patient‐induced scatter was corrected using an energy‐window‐based estimate. The scatter correction was incorporated into an OSEM reconstruction algorithm. We used a Gaussian pre‐filter FW(M = mm in the Pai and myocardial torso phantom studies. A pre‐ filter was not used and neither scatter nor attenuation was corrected in the study of the line source phantom. The reconstruction parameters used the setting recommended by the manufactures in clinical study. Table summarizes these conditions.
Evaluation
Spatial resolution was determined as the FW(M of images of the line sources in representative transaxial slices of the line source phantom after Gaussian fitting. The FW(M was calculated using an in‐house analytical tool based on the method specified by the NEMA standard for calculating FW(M corrected for background. )mage distortion was assessed as the aspect ratio ASR defined as the ratio of the radialFW(M
and tangentialFW(M in the radial and tangential directions of FW(M at each position. Furthermore, C‐mode acquisition datasets were reconstructed with filtered back projection used a ramp filter and non pre‐ and post‐filters C‐ mode_FBP , as compared with )Q‐SPECT.
We acquired SPECT data in air using a ‐mm diameter calibration source and measured activity in C‐, L‐, and )Q‐modes. SPECT counts were measured within a circular RO) with a radius measuring: rod radius + FWTM of SPECT image / . We then computed the cross‐ calibration factor CCF as Bq/SPECT counts. )mage quantifiability was validated with the linearly cross‐calibrated SPECT value Bq vs. the radioactivity concentration Bq of each hot chamber in the pie‐phantom for each system.
We performed myocardial image resolution to determine myocardial wall thickness MWT
when FW(M was calculated using a profile of ° per step generated from the short axis slices. Profiles were drawn through four slices of the short axis in the mid‐ventricular region of reconstructed myocardial phantom images. Myocardial wall thickness was determined by fitting each profile to a Gaussian model. To evaluate myocardial image resolution, the MWT was defined as FW(M values of myocardial wall thickness in the torso phantom. The true MWT was calculated from the myocardial areas of short‐axis images in the myocardial torso phantom using CT datasets.
Data were quantified from myocardial torso phantom SPECT images using the American (eart Association ‐segment model for the left ventricle. Polar maps were normalized to % peak activity and the relative ratio % of gamma ray count uptake %uptake was assessed for each segment. )n addition, homogeneity and mismatches were quantified in the three datasets by comparing the ‐segment values. The homogeneity, h (n) index for each segment was
calculated as:
h(n)
‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐‐
Where n represents the maximal and minimal
values measured for each segment. The value of a completely uniform segment based on this equation is zero.
The reference image was the C‐mode %uptake and the process images were L‐mode and )Q‐ mode %uptake for each segment, respectively. A mismatch m s was calculated using equation
.
m ‐‐‐‐ ,
Where s is one segment.
Statistical analysis
All data are shown as means ± standard deviation SD and were analyzed by ANOVA followed by the Bonferroni test. Probability values of < . were considered statistically significant. All data were analyzed using SPSS . )BM Corp., Chicago, )L, USA software. )mage quantifiability between SPECT values and radioactivity values for the pai phantom were assessed using linear regression analysis.
Results
Figure shows the FW(M radial and ASR of
Table 1. Acquisition and reconstruction conditions.
*FW(M = mm; AC, attenuation correction X‐CT ; SC, scatter correction.
SYSTEM Matrix size Collimator arc ° Scan Pixel size mm Number of views rotation mmRadius of Acquisition sec/view Reconstruction MLEM: )t = Post Filter Correction AC SC
C‐mode. x LE(R . Flash D Gaussian* ✓ ✓
L‐mode x LE(R . Flash D Gaussian* ✓ ✓
Multi-focus
Asia Ocean
each ac phantom locations center o
Figure 2. L A Linear and )Q B Pai pha
Figure 3. C FW(M of p
s fan beam collim
nia J Nucl Med B
cquisition m m. The valu
s is shown of rotation. T
Linear regressio regression anal Q‐modes.
antom SPECT im
Correlations amo phantom. NS, no
mation
iol.. 2013; 1(2):2
mode in th ue of FW(M
in rough sy The FW(M in
n analysis and p lysis shows relat
mages. Top, )Q‐m
ong C‐, L‐ and )Q t significant.
28-34.
he line sou M at differ ymmetry in
n the C‐ and
pai phantom SPE tionship betwee
mode; middle, C‐m
Q‐modes.Myocar
urce rent the d L‐
mo Th sou mo
ECT images. n relative SPECT
mode; bottom, L‐
rdial wall thickne
odes did not e FW(M val urce was incr ode, and FW(
Figur locat and ) A We a
FBP of FW locat
B away acqui ASR=
T counts and rel
‐mode.
ess was lower w
t significantly lue of )Q‐mo
reased by (M in the per
re 1. Estimated ion of line sourc Q‐modes.
Estimated added FW(M of
reconstructed W(M at C‐mod ion and was rea Estimated y from center
isition with
=tangential FW(
ative radioactivi
with )Q‐mode tha
On
y vary with de at the cen
% compared riphery was in
d FW(M and A ces using acquis
d FW(M in radi the filtered back data C‐mode_F de_FBP did not
sonably uniform ASR tended t r. C‐mode_FBP
FBP reco
(M / radial FW
ity concentratio
an C‐mode and L
nishi H et al
31
location. ntral line d with C‐ ncreased
ASR at each sition C‐, L‐,
ial position. k projection FBP . Value depend on m.
to increase P: C‐mode onstruction. W(M
ns for C‐, L‐
Onishi H et
32 Table 2. F
Segment C, Convent The segme anterolate : apical a by % ASR of )Q of ASR w were inc
Figure calibrate concentr in six ch m‐Tc. between concentr . X + . R R = . from L‐ function y‐interce
The tr the myo higher in
. ± .
P< .
modes Figure significa with L‐ a Figure %uptak with the torso ph mismatc The max three m segment the mism t al indings of uptak C . . . . . . . . . . . . . . . . . tional mode; L, L
ent is : basal ral, : mid anter anterior, : api
compared w Q‐mode was was . in th creased more e shows the ed SEPCT rations as a f hambers of t The results n SPECT va rations were
. R = R = . and
. Compare ‐mode, the for C‐ and ) ept value of )Q ue MWT mea ocardial torso n L‐ and C‐, th , P< . a
, respectiv did not si . The MWT ntly improve and C‐modes. shows th ke, mismatch e ‐segment hantom. Tab ch and homog ximal differe modes was
t model. L‐ a match more ke, mismatch and Uptake % L . . . . . . . . . . . . . . . . L‐mode; )Q, )Q‐m anterior, : bas rior, : mid ante
cal septal, : ap
with the cent . ‐ . . No a he center, an e in the periph
e correlation values and function of a the pai phan
of linear reg lues and th e as follows . ; L‐mo d )Q‐mode; Y ed with the r gradient of Q‐modes wa Q‐mode was asured at . o phantom w han in )Q‐mo and . ± . vely. Conven
gnificantly in the torso ed by )Q‐mod
.
hree sets o h for C‐, L‐,
t model and ble shows geneity of the ence in %upt % in the q and )Q‐mode than C‐mod d homogeneity in )Q . . . . . . . . . . . . . . . . . mode; Seg, segme sal anteroseptal roseptal, : mid pical inferior,
tral FW(M. T acquisition m
nd those of A hery. n between cro d radioactiv cquisition m ntom contain gression analy he radioactiv s: C‐mode, Y ode, Y = .
= . X – . results obtain
the regress s about . . T
negative. ± . mm fr was significan ode . ± .
vs. . ± ntional and differ P= .
phantom wa de, as compa
of polar m , and )Q‐mo the myocar s the %upta e three datas take among quantitative es both affec
e. )n particu
n segments u Misma L ‐ . ‐ . ‐ . . ‐ . ‐ . ‐ . ‐ . ‐ . . ‐ . ‐ . ‐ . ‐ . ‐ . ‐ . ‐ . ent.
l, : basal infer inferoseptal, : apical lateral, The ode ASR oss‐ vity ode ning ysis vity Y = X+ ned sion The rom ntly vs. . , L‐ . s ared maps odes dial ake, sets. the ‐ cted ular, bas ant Figu seg Valu map we mo hom seg hom wa mo res thr Dis T ima clin sys Asia using myocardial atch )Q ‐ . ‐ . ‐ . . ‐ . ‐ . ‐ . . ‐ . . ‐ . ‐ . ‐ . . ‐ . ‐ . ‐ .
roseptal, : basa : mid inferior,
: apex
sal anterose teroseptal, an
ure 4. Polar ma
ment model. ues for % uptake ps.
ere significa odel. The e mogeneity s gment mode
mogeneity in as significantl ode vs. L‐m spectively; C‐ ree regions .
scussion
The rapid ac ages can red nical studies stem reduces
Mult
a Oceania J Nucl l torso phantom (om C . . . . . . . . . . . . . . . . .
al inferior, : b : mid inferolat eptal, basal nd mid‐infero ap of myocardia e and mismatche ntly larger ffects of th significantly
el. The eff n the basal, m ly larger than ode: P= .
‐mode vs. )Q
quisition of duce the bur s have show s the amoun
ti-focus fan beam
l Med Biol. 2013; . mogeneity % L . . . . . . . . . . . . . . . . . asal inferolater teral, : mid an l inferolater olateral mism al torso phantom es are overlaid o in the he three m
differed in fect of C‐m mid, and apica n that of )Q‐m
, . an Q‐mode: P< .
myocardial p rden on pati wn that the ) nt of time n
m collimation
; 1(2):28-34. )Q . . . . . . . . . . . . . . . . .
al, : basal nterolateral, ral, mid matches m using ‐ on polar ‐segment modes on the ‐ mode on al regions mode C‐ nd . ,
for all
Multi-focus fan beam collimation Onishi H et al
Asia Oceania J Nucl Med Biol. 2013; 1(2):28-34. 33
acquire myocardial perfusion SPECT images. The present assessment of image quality in C‐, L‐, and )Q‐ acquisition modes using phantoms showed that that )Q‐mode can improve image resolution and quantifiability.
)mage resolution in the center of the field of view was similar among the three modes, but the resolution in )Q‐mode was degraded at the periphery region compared with the other modes Figure A . Paramithas et al and )mbert et al reported that FW(M between and
mm confers a slight improvement towards the collimator face. Our results roughly coincided with theirs when FW(M was centrally located. The differences between the central and periphery were larger for )Q‐mode than for C‐ and L‐modes; mainly because the Smartzoom focused collimator produced distortions in the absence of focused targets. These notions were supported by the findings of the ASR Figure B which at the periphery increased . fold compared with the center, and images were distorted at the periphery in the absence of focused targets. The center of the suggested ASR was not . , possibly due to the low iteration updates obtained using the conditions recommended by the manufacturer for the clinical study. )mbert et al indicated a difference
between radial and tangential FW(M . Our ASR findings agreed with those described by Onishi et al , who noted that S) product of
subset and iteration > updates are needed for ASR to be . . Resolution recovery clearly improved resolution more than FBP.
)mage resolution of MWT was significantly better in )Q‐mode than in C‐ and L‐modes Figure . Resolution recovery correction in C‐ and L‐ modes did not significantly improve resolution. We previously reported that resolution recovery is limited by the radii of rotation < mm , and that the resolution recovery ratio decreases as the radii of rotation increase . On the other hand, )Q‐mode slightly underestimates MWT, but was closer to the true MWT value.
We evaluated the accuracy of the three acquisition modes for quantitative imaging using a cross‐calibration technique Figure . The gradients of the regression function for C‐, L‐, and )Q‐modes were . , . , and . , respectively. The accuracy of the y‐intercept values was within % of the maximum activity of the chambers in the pai phantom. )mage degradation was obvious in L‐mode Figure b . The accuracy of acquisition modes for quantitative imaging was the same for the C‐ and )Q‐modes. )n contrast, L‐ mode underestimated the SPECT count by % according to the gradient of the regression function.
This phantom study showed that L‐mode had a considerable and negative impact on image homogeneity and the accuracy of image quantitation. )maging the myocardial torso phantom using L‐mode significantly increased %uptake and basal anteroseptal, basal inferolateral, mid anteroseptal, and mid inferolateral mismatches compared with C‐mode because the myocardial region was positioned off center in the phantom Figure . Liu et al
revealed significant erroneous homogeneity and artifacts when the target object is off the center of a ° orbit. )n contrast, the %uptake and homogeneity did not change with C‐mode in the segments Table , because the acquisition orbit was ° and SPECT images were reconstructed from ° projection datasets. The )Q‐mode sets the rotational center at the cardiac region and thus improved %uptake and mismatch compared with L‐mode. A slight mid‐inferolateral mismatch . % that occurred in )Q‐mode might have been due to the acquisition angle being limited to a
° arc.
The )Q‐mode equipped with Smartzoom system increase sensitivity and resolution and decreased homogeneity more significantly p
< . than C‐mode.
The performance accuracy and homogeneity of )Q‐mode is the same as C‐mode and is thus useful for acquiring MP). )n addition, performance is maintained during a short acquisition period . (owever, although the myocardial torso phantom is representative of a normal human chest, upward creep of the heart provoked reconstruction errors and created artifacts. Other artifacts such as those arising from motion generated by patients or the heart during acquisition for CT attenuation correction
are unpredictable.
We consider that )Q‐mode is useful for myocardial perfusion imaging despite decreasing projection datasets and orbit issues.
Conclusion
The quality of myocardial perfusion imaging was comparable among )Q‐mode, C‐mode and L‐ mode SPECT, with significantly improved image resolution and quality. This study provides new and important information about image resolution and quality using a multi‐focus fan beam collimator system )Q‐SPECT , which might be the most effective technology for myocardial perfusion SPECT imaging.
Acknowledgments
Onishi H et al Multi-focus fan beam collimation
34 Asia Oceania J Nucl Med Biol. 2013; 1(2):28-34.
References
. Jinghan Y, Xiyun S, Zuo Z, Da Silva. )terative SPECT Reconstruction Using Matched Filtering for )mproved )mage Quality. Nuclear Science Symposium conference )EEE.
; ‐ .
. (ughes T, Scherbinin S, Celler A. A multi‐ center phantom study comparing image resolution from three state‐of art SPECT‐CT systems. J Nucl Cardiol. ; : ‐ . . Onishi (, Motomura N, Fujino K, Natsume T,
(aramoto Y. Quantitative performance of advanced resolution recovery strategies on SPECT images: Evaluation with use of digital phantom models. Radio Phys Technol. ;
: ‐ .
. Myers RW, (aseman MK, Durbin M. (alftime acquisition of MP) with maintained image quality. J Nucl Cardiol. ; :S ‐ . . DePuey EG, Gadiraju R, Clark J, Thompson L,
Ansetett F, Shwartz SC. Ordered subset expectation maximization and wide beam reconstruction "half‐time" gated myocardial perfusion SPECT functional imaging: a comparison to "full‐time" filtered backprojection. J Nucl Cardiol ; : ‐
.
. Ali ), Ruddy TD, Almgrahi A, Anstett FG, Wells RG. (alf‐time SPECT myocardial perfusion imaging with attenuation correction. J Nucl Med. ; : ‐ .
. Lewin (C, (yun MC. A clinical comparison of UN upright triple‐head digital detector system to a standard supine deal‐head gamma camera [abstract]. J Nucl Cardiol
, : .
. Funk T, Kirch DL, Koss JE, Botvinick E, (asegawa B(. A novel approach to multipinhole SPECT for myocardial perfusion imaging. J Nucl Med ; : ‐ . . Vija (, Chapman J, Ray M. )QSPECT
technology White Paper. Siemens Medical Solutions USA Molecular )maging; : ‐ . . Sharir T, Ben‐(aim S, Merzon K, Prochorov V,
Dickman D, Ben‐(aim S, et al. (igh‐speed myocardial perfusion imaging: )nitial clinical comparison with Conventional dual detector anger camera imaging. J Am Coll Cardiol )maging ; : ‐ .
. Vija (, Malmin R, Yahil A, Zeintl J,
Bhattcharya M, Rempel TD, et al. A method for improving the efficiency of myocardial perfusion imaging using Conventional SPECT and SPECT/CT imaging systems. Paper represented at )EEE Nuclear Science Symposium conference ; : ‐ . . (awman PC, (aines RJ. The cardiofocal
collimator: A variable focus collimator for cardiac SPECT. Phys Med Biol ; : ‐
.
. Corbett J, Meden J, Ficaro E. Clinical validation of attenuation corrected cardiac imaging with )Q‐SPECT SPECT/CT [abstract]. J Nucl Med. ; suppl : .
. Knoll P, Kotalova D, Köchle G, Kuzelka ), Minear G, Mirzaei S, et al. Comparison of advanced iterative reconstruction methods for SPECT/CT. Z Med Phys. ; : ‐ . . Cerqueira MD, Weissman NJ, Dilsizian V,
Jacobs AK, Kaul S, Laskey WK, et al. Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart. Circulation ; : ‐ .
. Paramithas A, )rwin AG. Characterization of the )QSPECTTM cardiac SPECT system. EANM [abstract]. Eur J Nucl Med Mol )maging ; suppl : P.
. )mbert L, Poussier S, Franken PR, Songy B, Verger A, Morel O, et al. Compared performance of high‐sensitivity camera dedicated to myocardial perfusion SPECT: a comprehensive analysis of phantom and human images. J Nucl Med ; : ‐
.
. Zeintl J, Vija A(, Yahil A, (ornegger J, Kuwert T. Quantitative accuracy of clinical mTc SPECT/CT using ordered‐subset expectation maximization with ‐dimensional resolution recovery, attenuation, and scatter correction. J Nucl Med ; : ‐ .
. Liu Y(, Lam PT, Sinusass AJ, Wackers FJ. Differential effect of degrees and degrees acquisition orbits on the accuracy of SPECT imaging: Quantitative evaluation in phantoms. J Nucl Med ; : ‐ . . Goetze S, Brown TL, Lavely WC, Zhang Z,
Bengel FM. Attenuation correction in myocardial perfusion SPECT/CT: Effects of misregistration and value of reregistration. J Nucl Med ; : ‐ .