w w w . r b h h . o r g
Revista
Brasileira
de
Hematologia
e
Hemoterapia
Brazilian
Journal
of
Hematology
and
Hemotherapy
Original
article
Unrelated
hematopoietic
stem
cell
transplantation
in
the
pediatric
population:
single
institution
experience
Daniela
Hespanha
Marinho
∗,
José
Zanis
Neto,
Carmem
Maria
Sales
Bonfim,
Vaneuza
Araujo
Moreira
Funke,
Lisandro
Lima
Ribeiro
UniversidadeFederaldoParaná(UFPR),Curitiba,PR,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received4December2014
Accepted9February2015
Availableonline3June2015
Keywords:
Hematopoieticstemcell
transplantation
Bonemarrowtransplantation
Cordbloodstemcelltransplantation
Unrelateddonors
Child
a
b
s
t
r
a
c
t
Objective:Hematopoieticstemcelltransplantationhasbeensuccessfullyusedtotreatthe
pediatricpopulationwithmalignantandnon-malignanthematologicaldiseases.Thispaper
reportstheresultsupto180daysaftertheprocedureofallunrelatedhematopoieticstem
celltransplantationsinpediatricpatientsthatwereperformedinoneinstitution.
Methods:A retrospective review was performed of all under 18-year-old patients who
receivedunrelatedtransplantationsbetween1995and2009.Datawereanalyzedusingthe
log-ranktest,Coxstepwisemodel,Kaplan–Meiermethod,FineandGraymodelandFisher’s
exacttest.
Results:Thisstudyincluded118patients(46.8%)whoreceivedbonemarrowand134(53.2%)
whoreceivedumbilical cord blood transplants. Engraftmentoccurred in 89.47% ofthe
patientsthatreceivedbonemarrowand65.83%ofthosethatreceivedumbilicalcordblood
(p-value<0.001).Bothneutrophilandplateletengraftmentswerefasterinthebonemarrow
group.Acutegraft-versus-hostdiseaseoccurredin48.6%ofthepatientswithoutstatistically
significantdifferencesbetweenthetwogroups(p-value=0.653).Chronicgraft-versus-host
diseaseoccurredin9.2%ofthepatientswithahigherincidenceinthebonemarrowgroup
(p-value=0.007).Relapseoccurredin24%ofthe96patientswithmalignantdiseasewith
2-yearcumulativeincidencesof45%inthebonemarrowgroupand25%intheumbilicalcord
bloodgroup(p-value=0.117).Five-yearoverallsurvivalwas47%,withanaveragesurvival
timeof1207days,andnosignificantdifferencesbetweenthegroups(p-value=0.4666).
Conclusion:Despitedelayedengraftmentintheumbilicalcordbloodgroup,graft-versus-host
disease,relapseandsurvivalweresimilarinbothgroups.
©2015Associac¸ãoBrasileiradeHematologia,HemoterapiaeTerapiaCelular.Published
byElsevierEditoraLtda.Allrightsreserved.
∗ Correspondingauthorat:RuaGeneralCarneiro,181,AltodaGlória,80060-900Curitiba,PR,Brazil.
E-mailaddress:[email protected](D.H.Marinho).
http://dx.doi.org/10.1016/j.bjhh.2015.05.004
1516-8484/©2015Associac¸ãoBrasileiradeHematologia,HemoterapiaeTerapiaCelular.PublishedbyElsevierEditoraLtda.Allrights
Introduction
Allogeneichematopoieticstemcelltransplantation(HSCT)is
anacceptedtreatmentforanumberofinheritedandacquired
hematopoietic diseases in children, especially diseases for
whichanalternativetreatmentisnotavailableornolonger
effective.1,2
Humanleukocyte antigen(HLA)-matchedsibling donors
areavailableonlyforaround25%ofsuchchildren.However,
therehasbeensubstantialprogressoverthelastfourdecades
intheuseofalternativedonors,includingunrelatedvolunteer
donors.3–5
Theknownadvantagesofunrelatedumbilicalcordblood
(UCB) over unrelated bone marrow (BM) are well
docu-mentedandinclude:significantlyfasteravailabilityofbanked
cryopreservedUCBunits,norisktothedonor,reduced
trans-missionofviralillnessessuchascytomegalovirus(CMV)and
EpsteinBarrvirus(EBV),toleranceofHLAdisparitybetween
the donor and recipient, and reduced risk and severity of
acutegraft-versus-host disease(GVHD). However,the main
problemofusingUCBfortransplantationisthelownumber
ofhematopoieticprogenitorcells,whichresultsinincreased
riskofgraftfailure,delayedhematopoieticengraftmentand
delayedimmunereconstitution.6,7
HLAmatching,theuseofhigh-dosechemotherapyand/or
radiotherapyandtheneedofimmunosuppressivedrugs
rep-resentthecausesofonsetofmanycomplicationsfollowing
HSCT.8,9
GVHD is a common complication, mainly in unrelated
HSCTorinthepresenceofanyHLAallelemismatch.10Atan
incidenceof40%,acuteGVHDoccursintheearlyperiodafter
transplantwiththeskinbeingthemostcommonlyaffected
tissue.RiskfactorsforthedevelopmentofGVHDincludethe
recipient’sage,CMVserostatus,HSCdonorsourceand HLA
disparity.11,12ChronicGVHDisobservedin30–90%of
recipi-entsofHSCT.3
Inpatientswithmalignantdiseases,GVHDisassociated
withthegraft-versus-leukemiaeffect(GVL),thusresultingin
adecreasedincidenceofrelapse.13,14
Inrecentdecades,theoverallsurvival(OS)inchildrenwho
receivedHSCTismuchhigher,about 60%onetotwoyears
aftertransplant,dependingonthedisease,clinicalconditions
priortotransplantandcomplicationsafterthetransplant.2,6
Therefore,thisstudyaimedtoanalyzetheresultsofunrelated
HSCTinpediatricpatientsupto180daysaftertheprocedure
andtocomparethestemcellsourcesused.
Methods
Patients
This is a retrospective study. Between January 1995 and
December2009,261under18-year-oldpatientsreceived
unre-latedHSCTattheHospitaldeClínicas,UniversidadeFederaldo
Paraná(UFPR),Brazil.Ofthe261patients,ninewereexcluded
fromtheanalysisforthefollowingreasons:sevenpatientshad
insufficientdataforanalysisandtwopatientswererecipients
ofperipheralblood transplants.Onehundred andeighteen
Table1–Featuresofpatientsandtreatment.
Variable
Age–mean(range)years 8(0.2–18.0)
Diagnoses–n(%)
Malignantdisease 96(38.1)
Marrowfailure 95(37.7)
Immunedeficiency 42(16.7)
Metabolicdisorders 19(7.5)
Sourceofstemcell–n(%)
Bonemarrow 118(46.8)
Umbilicalcordblood 134(53.2)
DegreeofHLAmatch–n(%)
Withoutincompatibility(BM) 71(60.2) One/moreincompatibility(BM) 47(39.8) Withoutoroneincompatibility(UCB) 76(56.7) Two/moreincompatibility(UCB) 58(43.3)
Conditioningregimen–n(%)
CFA+FLU 106(42.4)
CFA+TBI 47(18.8)
CFA+BU 58(23.2)
CFA+BU+MEL 8(3.2)
Others 31(12.4)
ATG 174(69.0)
GVHDprophylaxis–n(%)
CsA+MTX 144(57.1)
CsA+steroids 91(36.1)
Others 17(6.7)
CFA:cyclophosphamide;FLU:fludarabine;TBI:totalbody irradia-tion;BU:busulfan;MEL:melphalan;ATG:anti-thymocyteglobulin; CsA:cyclosporine;MTX:methotrexate.
patientsreceivedBMand134UCBgrafts.Thecharacteristics ofthe252casesthatwereassessedarelistedinTable1.
HLAtypingandunrelateddonorselection
TheunitswereselectedonthebasisofbestHLAmatchingand
acriticalminimumcelldoseatthediscretionofthetreating
physiciansandsourcefromNationalandInternationalPublic
Banks.ClassItypingwasperformedbyserologicalor
molecu-lartechniquesandClassIItypingbymoleculartechniques;it
wasonlyin2008thattheanalysisofthelocusCwasfrequently
performed.
Conditioningregimenandprophylaxisagainst graft-versus-hostdisease
Theconditioning regimenand prophylaxisforacuteGVHD
variedaccordingtotheunderlyingdisease,stemcellsource
andHLAincompatibilities(Table1).
Transplantation
The units ofBM grafts used for transplantation contained
an average of4.39×108 total nucleatedcells (TNC) (range:
0.3–10.8 cells)per kilogramofrecipient’sbody weightafter
thawing.TheunitsofUCBgraftsusedfortransplantation
1.3×105CD34cells(range:0.1–11.4cells)perkilogramof
recip-ient’sbodyweightafterthawing.
Supportivecare
Central venous catheters were inserted in all patients.
Patients received acyclovir for antiviral prophylaxis,
flu-conazoleoramphotericin-Bforantifungalprophylaxis,and
trimethoprim-sulfamethoxazoleforprophylaxisagainst
Pneu-mocystiscarinii. Empiricalbroadspectrumantibiotictherapy
wasstartedatthefirstsignoffever.
Hematopoieticrecoveryandengraftment
Hematologic recovery was defined as the time when the
absolute neutrophil count was equal to or greater than
0.5×109cells/L in three consecutive laboratory counts on
different days and platelet count equal to or greater
than 20×109cells/L (after seven days without transfusion
support).6
Graft-versus-hostdisease
Patients were evaluated and considered at risk for acute
GVHDwhentherewasevidenceofneutrophilrecovery.
Diag-nosis of acute GVHD was based on clinical criteria, with
histopathologicconfirmationwhenpossible.Overallstaging
was assessed according to previously published criteria.15
Only those patients with sustained engraftment of donor
hematopoiesissurvivingformorethan90 daysafter
trans-plantwereassessedforchronicGVHDaccordingtothecriteria
describedinpreviousstudies.16
Othermeasurements
Relapse was characterized by morphological evidence of
malignant disease atany site. Transplant-related mortality
(TRM)wasdetermined tobeevery causeofdeaththatwas
notdeathbyrelapse.OSwasthetimebetween
transplanta-tionanddeathduetoanycauseorbetweentransplantation
andthedayofthelastfollow-up.
Statisticalanalysis
AllstatisticalanalyseswerecarriedoutusingtheSTATAv.12.0
computerprogram.Survivalrateswerecalculatedusingthe
Kaplan–Meiermethod.Thelog-rank testwasusedfor
test-ingdifferencesinsurvivalbetweengroupsintheunivariate
analysis.Cumulativeincidencecurveswereusedfor
engraft-mentand GVHDendpoints withdeathasacompeting risk
factor.TheCoxproportional hazards modelwithbackward
eliminationwasusedformultivariatetestingofco-variables
(statisticalsignificancewasbasedonp-values≤0.05).
Differ-encesbetweensubgroupswerecomparedusingtheFineand
Gray testwith death asa competing risk. Theassociation
betweendichotomousvariableswascalculatedusingFisher’s
exacttest.
Results
Patientcharacteristics
From1995to2009,252patientsweretreatedwithunrelated
HSCT.Themeanageofthepatientswaseightyears(range:
0.2–18 years) with 34 patients being one year old or less.
Mostpatientsweremale(61.1%).Atotalof96patientswere
transplantedformalignantdiseaseswith64(66.7%)beingat
anearlystage.Atotalof150(59.5%)donor-graftpairs were
matchedforABOand124(49.8%)werematchedforgender.
BMwasusedforHSCTin118patients(46.8%)andUCBin134
patients(53.2%).Thepatientsaswellastreatmentarelisted
inTable1.
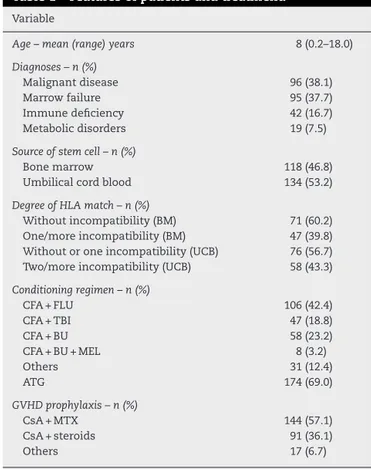
Neutrophilandplateletengraftment
Neutrophil engraftment occurred in 181 ofthe 234 (77.4%)
avaliable(18patientsdiedbefore)cases(BM:89.47%andUCB:
65.83%). Inthe BMgroup,102 ofthe118 patientsachieved
neutrophilengraftmentinanaveragetimeof24days(range:
11–1696days).ThecumulativeincidencebyDay30was80%.
IntheUCBgroup,79ofthe134patientsachievedneutrophil
engraftmentinameantimeof32days(range:14–4037days).
The cumulative incidenceby Day 42 was 62%. Inthe
uni-variateanalysis, neutrophil engraftmentwas influencedby
cellsource(p-value<0.001)andpatientage(p-value=0.002).
Inthemultivariateanalysisonlycellsource(p-value<0.001)
wasasignificantlyfavorablefactorforneutrophilengraftment
(Table2andFigure1).
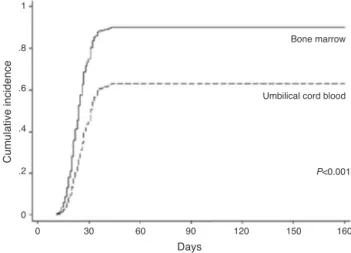
IntheBMgroup,patientsachievedplateletengraftmentin
anaverageof25days(range:10–96days).Thecumulative
inci-dencebyDay30was50%.IntheUCBgroup,patientsachieved
plateletengraftmentinameantimeof43days(range:19–158
days).ThecumulativeincidencebyDay42was38%.Inthe
uni-variateanalysis,plateletengraftmentwasinfluencedbycell
source(p-value<0.001),patientage(p-value=0.022)andHLA
match(p-value=0.027).Inthemultivariateanalysiscellsource
(p-value<0.001)andHLAmatch(p-value=0.013)were
signifi-cantlyfavorablefactorsforplateletengraftment(Table2and
Figure2).
1
.8
.6
.4
.2
0
0 30 60 90 120 150 160
Days
Cumulative incidence
P<0.001
Umbilical cord blood Bone marrow
Table2–Multivariateanalysis.
Variable Hazardratio 95%Confidenceinterval p-Value Favorablefactor
Neutrophilengraftment
Stemcellsource 0.37 0.25–0.54 <0.001 Bonemarrow
Age 0.99 0.96–1.03 0.744
Plateletengraftment
Stemcellsource 0.36 0.24–0.54 <0.001 Bonemarrow
Age 0.52 0.94–1.03 0.520
HLA 0.63 0.43–0.90 0.013 Withoutmismatch
AcuteGVHD
HLA 1.74 1.20–2.52 0.003 Withoutmismatch
ATG 0.58 0.40–0.86 0.006 Use
Relapse
Diseasestatus 3.27 1.44–7.42 0.005 CR1/2
Overallsurvival
Age(years) 1.11 1.04–1.18 <0.001 Young
GVHD 2.55 1.29–5.04 0.007 Without
ATG 1.99 1.07–3.72 0.029 Use
HLA:humanleukocyteantigen;GVHD:graft-versus-hostdisease;ATG:anti-thymocyteglobulin;CR1/2:completeremission1/2.
1
.8
.6
.4
.2
0
0 30 60 90 120 150 180 210 240 270 300 330 360
Days
Cumulative incidence
P<0.001
Umbilical cord blood Bone marrow
Figure2–Cumulativeincidenceofplateletengraftment overtime.
Graft-versus-hostdisease
AcuteGVHDoccurred in88 (48.6%)patients ofthetotal of 181(onaveragewithin90days)andwasscoredasGradeII (54.5%)andGradeIII–IV(45.5%).ByDay100after transplanta-tion,thecumulativeincidencewas44%intheBMgroupand 43%intheUCBgroup(p-value=0.653–Figure3).HLAmatch
(p-value=0.004)andATG(p-value=0.005)weresignificantin
theunivariateanalysis;thiswasthesameinthe
multivari-ateanalysis(p-value=0.003andp-value=0.006,respectively–
Table2).
ChronicGVHDoccurredin13(9.2%)patientsoutofthetotal
of142(onaveragewithin90days).Thediseasewasclassified
aslimitedinsixcases(46.2%)andextensiveinsevencases
(53.8%).Atoneyearaftertransplantation,thecumulative
inci-dencewas14%intheBMgroupand2.5%intheUCBgroup
(p-value=0.053).
1
.8
.6
.4
.2
0
0 90 180 270 360 450 540 630 720
Days
Cumulative incidence
P=0.653
Umbilical cord blood Bone marrow
Figure3–Cumulativeincidenceofacutegraft-versus-host diseaseovertime.
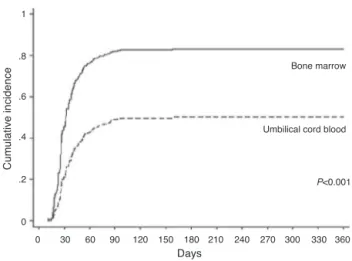
Relapse
Hematologicalrelapsewasdetectedbetween21and1228days
(mean: 181 days; 95% confidence interval:94.0–670.0) after
transplantationin23of96patientstreatedformalignant
dis-ease.Relapsewasobservedin29.31%ofthepatientsinthe
BMgroupand15.79%intheUCBgroup(p-value=0.117).The
cumulativeincidencesofrelapsewere45%intheBMgroup
and25%intheUCBgroupattwoyears(Figure4).Thestageof
thediseasewasfoundtobesignificant(p-value=0.005)
how-evercellsource,GVHD,CMVandABOincompatibilitywerenot
(Table2).
Overallsurvivalandcausesofdeath
Withanaveragefollow-uptimeofsurvivingpatientsof1207
days (3.3 years), the cumulative proportions of surviving
patientsatoneyearandfiveyearswere55%and47%,
1
.8
.6
.4
.2
0
0 365 730 1095 1460 1825
Days
Cumulative incidence
P=0.117
Umbilical cord blood Bone marrow
Figure4–Cumulativeincidenceofrelapseovertime.
yearandfiveyearsafterunrelatedHSCTwithBMwere55%and
45%,respectivelyandforUCBtheywere52%and45%,
respec-tively(p-value=0.466)(Figure5).Inamultivariateanalysis,the
factors thatmostinfluenced ofnegativeformtheOS were
higherages(p-value<0.001),non-useofATG(p-value=0.029),
andGVHD(p-value=0.007–Table2).
Inthisseries,131(52.0%)patientsdied,59(50.0%)intheBM
groupand72(53.73%)intheUCBgroup(p-value=0.614).Death
was associated with infection (n=47), graft failure (n=29),
GVHD(n=25),relapse(n=18)andothers(n=12).
Discussion
Thisretrospectivestudycomparedaheterogeneouspediatric
populationwithdifferentdiagnosestransplantedoveralong
period oftime using two different stem cell sources. This
studyaimedtoevaluatetheoutcomesofunrelatedHSCTin
thepediatricpopulationcomparingtheuseofBMandUCB;
only25%ofpatientswhoneedtransplantsfindsuitablefamily
donors.
Thecumulativeincidence(80%byDay30intheBMgroup,
and62%byDay42intheUCBgroup)andspeedofneutrophil
engraftment(onaveragebyDay24intheBMgroupandDay
0.00 0.20 0.40 0.60 0.80 1.00
0 500 1000 1500 2000 2500 3000 3500 4000 4500 5000
Time (days)
Survival
P=0.466
Umbilical cord blood Bone marrow
Figure5–Cumulativeincidenceofoverallsurvivalover time.
32 in theUCB group)were similar tothe results ofBarker
et al.17 Inother studiesonUCBtransplantations6,18,19
neu-trophilengraftmentwasfoundhigherandfasterthaninthe
currentstudy.Inthisstudy,neutrophilengraftmentwasnot
influencedbyHLAmatchingsimilartothestudyofPetterson
etal.6eventhoughKurtzbergetal.18foundthatHLAmatching
influencedneutrophilengraftment.Itisknownthat,during
thetimeofthisstudy,HLAtypingtechniqueswereimproved.
Manystudies1,6,19demonstratedthatthedoseofinfusedCD34
cells influencesneutrophilengraftment,however no
differ-ence wasseen inthecurrentstudyperhapsduetothelow
meancellcountinfused.
Thecumulativeincidence(50%byDay30intheBMgroup,
and 38% byDay 42 inthe UCBgroup)and speed (on
aver-age,Day25intheBMgroupandDay43intheUCBgroup)of
plateletengraftmentweresimilartoBarkeretal.12Petterson
etal.,6whoanalyzedpatientswhoreceivedUCB
transplanta-tion,foundthatplateletengraftmentwassimilartothisstudy.
Theauthorsreportedthatengraftmentwasnotinfluencedby
theinfusedTNCorCD34celldoseorHLAmatching.However
inthecurrentstudy,plateletengraftmentwasinfluencedby
HLAmatching.ItisagainimportanttonotethatHLAtyping
techniqueswereimprovedduringthisperiod.
Acute GVHD remains a major cause of morbidity and
mortality after HSCT. The incidence of Grade II–IV GVHD
is between 20 and 50%. The recipient’sage, CMV
serosta-tus,donorsourceandHLAdisparityhaveconsistentlybeen
foundtoberiskfactorsforthedevelopmentofacuteGVHD.12
This study demonstrates that 48.6% ofthe patients
devel-opedacuteGVHDwithGradeIIbeingfoundin54.5%ofthe
cases.Somestudies12,18,19demonstratedsimilarincidences,
howeverothers2,10foundlowerincidences,andFaracietal.20
foundanincidenceof75%.Inthisreport,aswellinBarker
etal.17andHwangetal.,21therewasnodifferenceinthe
inci-denceofacuteGVHDbetweentheBMandtheUCBgroups;
however,Wangetal.22reportedthattheincidencewaslower
intheUCBgroup.
TheincidenceofchronicGVHDrangesbetween30%and
50%andthusisamajorcauseofnon-relapsemortalityand
morbidity in long-termsurvivors.Chronic GVHD is
associ-atedwithagraft-versus-leukemiaeffect(GVL)resultingina
decreasedincidenceofrelapse.13Inthisstudy,theincidence
was9.2%versus25.0%asreportedbyZeccaetal.13Howeverin
thisstudythefollow-upwasonlyupto180daysafter
trans-plant. Similar toother reports,21,22 chronicGVHD occurred
moreintheBMgroup(p-value=0.007).Barkeretal.17reported
nodifferencebetweenthetwogroups.
Relapseoccurredin24%ofthepatientswhoreceivedHSCT
for malignant diseaseswith the incidencebeing similarin
bothgroups.Wang etal.22stated thatrelapsewaslowerin
theUCBgroup.GVHDdidnotshowaGVLeffect,similartothe
reportbyLeeetal.23;howeverYiet al.24 believethatGVHD
hasanimportantprotectiverole.Thestageofthediseasewas
importantforprognosisinthisstudyandinothers.18,25
Theestimatedone-yearOSafterHSCTintheBMandthe
UCBgroupswere58%and52%,respectively(p-value=0.466).
Thesedatawerefoundtobesimilartotheliterature.3,6,17In
the multivariateanalysis,the resultsassociatedwitha
sig-nificantlyworseOSincludedhigherage,non-useofATGand
hadworsesurvival,andWagneretal.1statedthatHLA
mis-matchinfluencedsurvivaltoo.Infectionwasthemaincause
ofdeathasinotherpublications.2,25
Conclusion
OurfindingsconfirmthatunrelatedHSCTwithBMandUCB
resultsinsimilarcomplicationsandsurvivalinthepediatric
population.Therefore,UCBisasafecellsourceforpatients
whodonothaveasiblingdonorforHSCT.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1. WagnerJE,BarkerJN,DeForTE,BakerSC,BlazarBR,EideC, etal.Transplantationofunrelateddonorumbilicalcordblood in102patientswithmalignantandnonmalignantdiseases: influenceofCD34celldoseandHLAdisparityon
treatment-relatedmortalityandsurvival.Blood. 2002;100(5):1611–8.
2. VossenJM,DonkerAE,HeemskerkBA,LieJLWT,Weel-Spiman MH,BrediusRGM,etal.Unrelateddonormarrow
transplantationinchildren:transplantpolicyandoutcomein LeidenPaediatricsSCT-Centre.BoneMarrowTransplant. 2010;45(1):87–95.
3. Yumura-YagiK,InoueM,SakataN,OkamuraT,YasuiM, SawadaA,etal.Unrelateddonorbonemarrow
transplantationfor100pediatricpatients:asingleinstitute’s experience.BoneMarrowTransplant.2005;36(4):307–13. 4. HoughR,CooperN,VeysP.Allogenichaematopoieticstem
celltransplantationinchildren:whatalternativedonor shouldwechoosewhennomatchedsiblingisavailable?BrJ Haematol.2009;147(5):593–613.
5. SmithAR,WagnerJ.Alternativehaematopoieticstemcell sourcesfortransplantation:placeofumbilicalcordblood.BrJ Haematol.2009;147(2):246–61.
6. PettersonTE,GabrielM,TiedemannK,TeagueL,ShawPJ, BakerD,etal.Outcomefollowingunrelatedcordblood transplantin136patientswithmalignantandnon-malignant diseases:areportfromtheAustralianandNewZealand children’shaematologyandoncologygroup.BoneMarrow Transplant.2009;43(3):207–15.
7. RochaV,GluckmanE.Improvingoutcomesofcordblood transplantation:HLAmatching,celldoseandothergraft-and transplantation-relatedfactors.BrJHaematol.
2009;147(2):262–74.
8. MianoM,FaraciM,DiniG,BordigoniP.Earlycomplications followinghaematopoieticSCTinchildren.BoneMarrow Transplant.2008;41Suppl.2:S39–42.
9. PerezLE.Outcomesfromunrelateddonorhematopoietic stemcelltransplantation.CancerControl.2011;18(4):216–21. 10.DvorakCC,WrightNB,WongWB,KristovichKM,Matthews
EW,WeinbergKI,etal.Safetyofhematopoieticstemcell transplantationinchildrenlessthanthreeyearsofage. PediatrHematolOncol.2008;25(8):705–22.
11.BallLM,EgelerRM.AcuteGVHD:pathogenesisand classification.BoneMarrowTransplant.2008;41Suppl. 2:S58–64.
12.MacMillanML,WeisdorfDJ,BrunsteinCG,CaoQ,DeForTE, VernerisMR,etal.Acutegraft-versus-hostdiseaseafter unrelateddonorumbilicalcordbloodtransplantation: analysisofriskfactors.Blood.2009;113(11):2410–5.
13.ZeccaM,PreteA,RondelliR,LaninoE,BalduzziA,MessinaC, etal.Chronicgraft-versus-hostdiseaseinchildren:incidence, riskfactors,andimpactonoutcome.Blood.
2002;100(4):1192–200.
14.JernbergAG,RembergerM,RindénO,WiniarskiJ.
Graft-versus-leukaemiaeffectinchildren:chronicGVHDhas asignificantimpactonrelapseandsurvival.BoneMarrow Transplant.2003;31(3):175–81.
15.PrzepiorkaD,WeisdorfD,MartinP,KlingemannHG,BeattyP, HowsJ,etal.ConsensusconferenceonacuteGVHDgrading. BoneMarrowTransplant.1995;15(6):825–8.
16.FilipovichAH,WeisdorfD,PavleticS,SocieG,WingardJR,Lee SJ,etal.NationalInstitutesofHealthConsensus
DevelopmentProjectonCriteriaforClinicalTrialsinChronic Graft-versus-HostDisease:I.DiagnosisandStagingWorking GroupReport.BiolBloodMarrowTransplant.
2005;11(12):945–55.
17.BarkerJN,DaviesSM,DeForT,RamsayNK,WeisdorfDJ, WagnerJE.Survivalaftertransplantationofunrelateddonor umbilicalcordbloodiscomparabletothatofhuman leukocyteantigen-matchedunrelateddonorbonemarrow: resultsofamatched-pairanalysis.Blood.2001;97(10):2957–61. 18.KurtzbergJ,PrasadVK,CarterSL,WagnerJE,Baxter-LoweLA,
WallD,etal.ResultsoftheCordBloodTransplantationStudy (COBLT):clinicaloutcomesofunrelateddonorumbilicalcord bloodtransplantationinpediatricpatientswithhematologic malignancies.Blood.2008;112(10):4318–27.
19.PrasadVK,MendizabalA,ParikhSH,SzabolcsP,DriscollTA, PageK,etal.Unrelateddonorumbilicalcordblood
transplantationforinheritedmetabolicdisordersin159 pediatrcpatientsfromasinglecenter:influenceofcellular compositionofthegraftontransplantationoutcomes.Blood. 2008;112(7):2979–89.
20.FaraciM,CavigliaI,BiralE,MorrealeG,GiardinoS,Garbarino L,etal.Acutegraft-versus-hostdiseasenpediatricallogeneic hematopoieticstemcelltransplantation.Single-center experienceduring10yr.PediatrTransplant.2012;16(8):887–93. 21.HwangWY,SamuelM,TanD,KohLP,LimW,LinnYC.A
meta-analysisofunrelateddonorumbilicalcordblood transplantationversusunrelateddonorbonemarrow transplantationinadultandpediatricpatients.BiolBlood MarrowTransplant.2007;13(4):444–53.
22.WangJ,ZhanP,OuyangJ,ChenB,ZhouR,YangY.Unrelated donorumbilicalcordbloodtransplantationversusunrelated donorbonemarrowtransplantationinadultandpediatric patients:ameta-analysis.LeukRes.2010;34(8):
1018–22.
23.LeeJH,YoonHS,SongJS,ChoiES,MoonHN,SeoJJ,etal.
Unrelatedhematopoieticstemcelltransplantationfor childrenwithacuteleukemia:experienceatasingle institution.JKoreanMedSci.2009;24(5):904–9.
24.YiES,LeeSH,SonMH,KimJY,ChoEJ,LimSJ,etal.
Hematopoieticstemcelltransplantationinchildrenwith acuteleukemia:similaroutcomesinrecipientsofumbilical cordbloodversusmarroworperipheralbloodstemcellsfrom relatedorunrelateddonors.KoreanJPediatr.2012;55(3): 93–9.