Structure, Morphology and Chemical Synthesis of Mg1-xZnxFe2O4 Nano-Ferrites Prepared by Citrate-Gel Auto Combustion Method
Texto
Imagem
Documentos relacionados
Em sua pesquisa sobre a história da imprensa social no Brasil, por exemplo, apesar de deixar claro que “sua investigação está distante de ser um trabalho completo”, ele
The probability of attending school four our group of interest in this region increased by 6.5 percentage points after the expansion of the Bolsa Família program in 2007 and
Este relatório relata as vivências experimentadas durante o estágio curricular, realizado na Farmácia S.Miguel, bem como todas as atividades/formações realizadas
The morphology of the materials was signiicantly altered by the synthesis method, as the KNb3 O 8 prepared by the polymeric precursor method presented a more porous
This log must identify the roles of any sub-investigator and the person(s) who will be delegated other study- related tasks; such as CRF/EDC entry. Any changes to
Despercebido: não visto, não notado, não observado, ignorado.. Não me passou despercebido
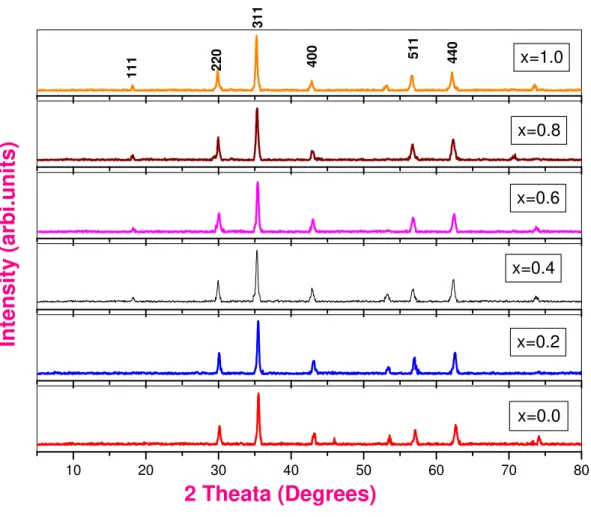
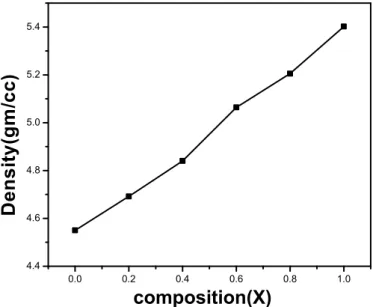
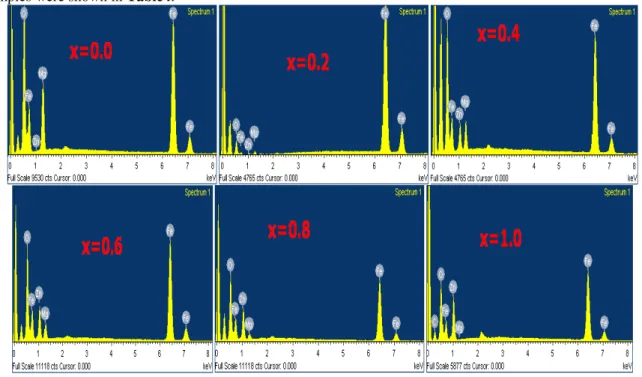
ferrites exhibits special magnetics and electrical properties, that are sensible to structure, synthesis method, particle size and cation distribution between A and B sites
In this work, zinc aluminate spinel was prepared by two methods of directly synthesis (without calcination): microwave assisted combustion and hydrothermal method.. The