Ree. Inst. Med. ttop. São Paalo 2g(7)t9-17, jøneirc-f everc¡rc, 1987
RADTOMETFTC STUDTES ON THE OX|DAT|ON OF (l-rC) FATW ACTDS BY DRUG.SUSCEPTIBLE AND DRUG.BESISTANT MYCOBACTERIA
Eilwâldo E. C/IIuáAGO, M.D., The¡ess M. KOPAffIC, GItflt¡ K. HOPKINS, N¡noy P. CANNON & Heùy N. WAGNEXI JR,, M.D.
ABSTRACT
A
radiometric assay system has been usedto
study oxidation pattems of(1-14C) fatty acid.s by drug-susceptible and drug.resistant organisms
of
the genus lì(ycobacteliüm.TV,¡o strains
of
M. tuberculosis susceptibleto all
drugs, HrRv and Erdmân,were used, Drug-resistan¿ organisms included
in
this invesftgation were M. tu.berculosis tl3zRv resistant to 5 ug,/ml isoniazid, M, bovis, M. aviul4 M.
intra߀llu-Iarc, M. kansesü and M. chelonei. Tt¡e organisms were inosulated in sterile leac,
tion vials containing liquid ?Hg medium, 10% .ADC €nrichment and 1.0 uoi of one
of
the (114C) fatty acids (butyric, hexanoic, octanoig decanoic, lauric, myristic, palmitic, stearic, oleic, linoleic, linolenic), Vials v/ere incubatedat
3?C and the laooz envolved'veas measuæd daily for 3 days with a Bactec R'301 instrument.Although each individual organism displayed a different pattern of fatty
oxi-dation, these pattems were not distinctive enough
for
identiJtcation of theorga-nism. No combination
of fatly
acids nor preferential oxidationof
long chain orof
short châinfatw
acids were ableto
separate susceptible from resistantor-ganisms. ¡'ulther investigation with a laryer number
of
drug susceptiblemyco-bacteria including assimilation studies and oxidation
of
other substætes may berequired
to
achieve a distinction between drug-susceptible and drug-resistant my-cobacteria.KEY
WORDS: lrycobâcteria; r1c-Fatfy Acids; Oxidåtion pãtterns; Radlometric system; Drug-resistance.INTBODIJCTION
fhe
ideaof
using carbon-l4 labeled fatwacids as substrates to study Í¡ycobaterial
meta-bolism resulted from an incidental finding in one
of
the
experimentswith M.
lq¡ra€mu-rium s.It
was observedthat
po\ßorbate 80markedly enìanced
the
oxidationof
(U-t4C) acetate by this organism.It
was assumed il¡ât oleic acid, which is part of the chemicalstruc-ture
of
polysorbate 80, was ¡esponsible forincreâsed oxidation rates
of
(UJ4C) acetate. Aninvestigation
of
the
oxidation rateof
(llaC) oleic acid revealed a r4CO2 ploductioncompå-rable
to
thât
of
(U-t4c) acetate and 1edto
a studyol
the entire seriesof
(lJaC) fatt¡¡ acids. Because some radiometric similarities had aheady been found with M. lelfiaemuriuIlr EndM. tuberculosis a, the study
of
the (ltac) fattyF¡om the Division of Nucleâr Meauc¡¡e, The Joh¡s Ifopki¡s Medlcal Instltutlons, Baltimorq MÊÌylaDd 212115, U.A.A.
Sùppofied in paÌt by USPHS Glarlt GM 10548.
¡ddF-ai for coEespoúdelrc€: Edfl¡aldo E. Cam.aryo, M.D., Centlo de Medicina Nucle3Ì ¿la Unive¡sialåde do São P¿ulo. CalJ€
CAMAEGO, E. E,; KOPÂJIIC, T. M.; I{OPKINS, c. K.; C¡¡ÍNON, N. p. & WAGNER JR., lr. N. - Raauomet c stucties
on the oxia¡âtion of (1l C) far¿y âciats Þy dftg.sÙsceptibte and at¡ug-rcsrsta¡r oÌ.cobacte a. Rev. rnst. Med. rrol'.
São P¡ù¡o, 29(r):9-1?, 198?.
acid series was extended to include the latter. Pleliminary expedments showed differences in the oxidation Ëtes of fatty acids between the-se organisms and led to the hypothesis that
oxi-dation patterns
of
fatty acids might provide a ba,sisfor
differentiation of mycobacteria. Forexample, a comparison of oxidation pattems of
M.
lepraæmuúum,M.
bovis, M. Jtuberculosis (It7Rv andE!ùran)
showed sig¡ificantdiffer-ences6'7. .{lso, pr€limina.ry results with isonia-zid-¡esista"nt and isoniazid-susceptible strains of
M.
tuberculosis suggested th¿t oxidationpat-terns
of fatty
acids and amino acids might helpto
identify susc€ptible and resistant oÍ-ganisms 8.The present study descdbes a seties of ex,
periments
in
which oxidationof
(1J4C) fattyacids was used
in
an attemptto
differentiatesusceptible and resistant strains
of
mycobac-teúa.MATERI,ILS AND METTIODS
Preparatlon
of
Baailli: Tt¡e organisms obtain-ed from T¡udeau Mycobacterial CulturaCollec-tion
(Saranac Lake,NY.)
includedM.
úuber-culosis II37RV (TMC 102)
fully
susceptible; M.tuberculosis H37Rv (TMC 303) lesistant
to
5uglml isoniazid
(INH);
M,
tuber€ulosis Erd-man (TMC 107) ful(y susceptible; M. bovis (TMC 602) resistant to 5 ug/ml INH; M. ¿vium (TMC 706) ¡esistant to 5ug,hl
INH, 10ug,hl
INH,10 ug,/ml para-amino salicylic
acid
(PAS), 2uglml
streptomycin (SM),5 uglml
viomysin(vM), 5
ug,hl
kanamycin (ItM), 30 uglmlcy-close¡ine (CS),
5 uglml
ethionamide (ETA),12 uglml ethambutol (EMB), 5 uglml rifampin
(RIF),50
ug/ml pyrazinamide (PZA),suscep-tible to all others; M. intr¿ae ula,re (TMC 1403)
resistant
to
5
uglmlINH,
10uglml
PAS, 2 uglml SM, 5 ug,/ml KM, 5 uglml VM, 30 ug/mlCS,6
ug/mlEMB,0.2
ug,/mlRIF,50
ug,/ml PZA,. susceptibleto all
others;M.
kansasil (TMC 1201) resistant toI
ug/ml lNH, 2 uglmlSM, 6 uglml I'MB , 0.2 ue/ml F"IF, 25 :ulg/mtP?A,
susceptible
to all
others;M.
chelonei (TMC 1542) resistant toINII,
SM, P.Á.S, VM, CS, ETÀ, P"IF, pZA, susc€ptibleto
50ug/ml
I(M.
Allorganisms were received in 3
Írl
stedle serum vials containing 2 ml of b.acterial suspension in liquid ?Hg medium (BâltimoÌe BiologicdLa-boratories, BBL). These suspensions were im. mediately trânsferred
to
stedle 250ml
eden-10
meyer tlashs
with
gauze plugs, containing 50ml of
liquid ?II9 medium ,lvith 107¿ âlbumin-dextrose.catalase (ADC,BBL)
enrichment and 0.05% polysobate 80. Tlle organisms were incu-bated at 3?"Cfor
6to
lt
days and then homo-genizedwith
a
Soryal Omnimixer (8.5 speedscale) twice
for
30 seconds. The numbetof
bacteda s'as estimated using MacFarlând
ba-lium
sulfate standards 2. The final suspensiolwas further diluted
with
sterile ?Hg mediumto yield from
1x
108 to 6 x 108 bacteriâ per ml,Part of this suspension was again diluted 1:10
with
?Hg medium, dividedinto
2ml
aliquots dispensedin
stele
plastic tubes and stored at -?tlcfor
future en)eriments.Exporimental
Media:
Liquíd
?Hg
medium\tith
lOVa ADC enrÍchment and no polysorbate \Ã'as used with all the organisrns tested. Reaction System: T'he reastion systemfol
de-tection
of
14CO2 consistedof
0.8ml
mediumin a
5.0ml
multidose sterile gl¿ss vial with0.1
ml
of bactedal suspension and 0.1ml
(1.0uci)
of
r'ro.substrate.The
following (1-r4C)fatty acids (Amersham Corporation) were used
with all organisms: butyric, hexanoic, octanoic,
decanoic,
lauric, myristic,
palmitic, stearicn oleic, linoleic, and linolenic. In preliminary ex-periments e, (1-14C)fatty
acids were dissolvedin
ethyl alcoholor
methyl alcohol (lauric). A comparisonoI
14Cq output from alcohol dis-solved and bovine sen¡m albumin (BSA)-
ca-talase complex dissolved (IJaC) fâtty acid de-monstrated a better oxidation rate when using the
latter
(Fig. 1).In
the present study, aUuci
of
(l-iac)fatw
acid u¡eÎe dissolvedin
1.0fatty
acids were prepared as follorás:a)
250uci of
(lrac) iatty
acid were dissolved in.1.0, mlof
0.01 N KOH;b)
1.0ml of
a SOE|solÍ-tion of BSA in lvrodds solution was added; c)
0-05
ml
of
0.1N
HCI were added and pHchecked betv¿een 6.5 and ?.0; d) the solutionwas
Jiltercd through a 0.22
u
pore size memblanefilter
(Milliþore Corporation);e)
0.25ml
of stedle catalase (50,000-
59,000 units/ml) were a.dded;f)
sterile vrater was added to bring thefinal
solutionto
desiled volume. AII speciflc activities were lowered to 25 mCi/rDM byaddi-tion of the sorresponding non-ladio¿ctive fatty
acid.
,As indicatorc
of
met¿bolic activityof
theorganisms, l4c-formate
(25 moi/mM)
anda
:
t
c¿.[L{RGo, E. E.; KoPAIuc, T. M.; EOPKINS, G. K.; C¡ìNON, N. P. & .WAGNER J8,., E. N.
- Raitroúet¡tc stu¿tt€s
cn tho oxi¡låLion of (1-1{C) fatiy åcids hy drug'susceplible and ùug-resistanl mycobacte a. Rev. Ist. Med. trop. Sio Paulo. 29(I):9'l?. 198?.
(U-laC) acetate (25 mCVmM) \Ã'ere used ló. Âll vials were prepaled at least
in
triplicate. Con.trol
vials were preparedin
the same way, but with autocloved bacte a added. Eachexperl-ment $'as repeated at least, twice.
Radionetric Measuremenf:
All
vials were in-cubated at 37"C.,{n ion chamber device (Bactecî
R-301, Johnston Laboratories, CocJ<eysville,
!
Md.) 'was used
to
measure bactefal metabo- ntism.
The vials were sampled daily for 3 days. -=Results \áere obtained
as
"indexunits"
(100 õunits
=
25 nanocuriesof
1aC activity). Mean gand standared devidation
of
the
cumulative ElaCO, production
for
each substrate overthe
p.entile e&erimental period wele calculated. Re.
g
sults were expressed as percent lauric acidoxi-
9dation.
¡
=
SúertlitJ¡ îesting: Sterility tests were perform- õ
ed on positive samples and consisted
of
sub-culture on chocolate-agar plates and
radiome-tric
sterility testing \Ã'ith (U-l4C) glucose 10,11,RESULTS
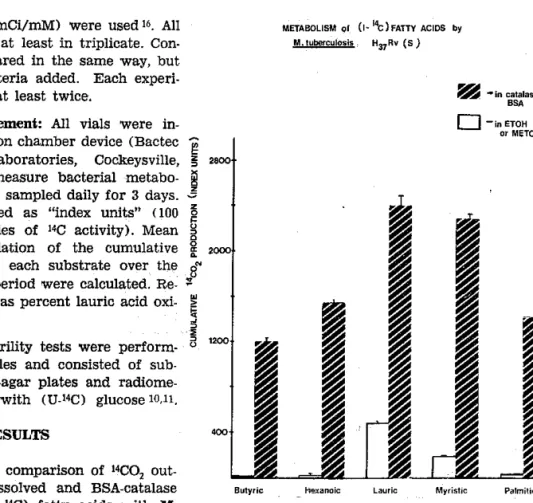
Figure 1 shows a comparison of raoq
out-put from
alcohol dissolved and BsA-catala.secomplex dissolved (1-r4C)
fatty
acidswith
M. ûrb€rculosis H3?Rv fully susceptible (TMC 102)Higher ¡aCO2 production rates were obtained
when the
fatty
acidsï'ere
dissolvedin
BS.A-catalase complex. Similar results were foundwith
M.
avftrm and M. tub€rcrdosis II3rRvre-sistant
to
5 ug/mlINH
(TMC 303).When exposed
to
the entire(1JC)
fattyacids series, M. tuberculosis H3zRv fully
suscep-tible (TMC 102) sho'.ved highest oxtdation mtes
with
octanoic, decanoic, lauric,a¡d my
sticâcids.
Low
oxidation rates v'ere found with butyric, hexanoic, linoleic and oleic acids (Fig.2, Table
I).
M. tuberculosis Erdman sho\ryed
preferen-tial
oxidationwith
hexanoic, lauric, palniticand linoleic acids. Intemediate oxidation rates
occurred Ìt/ith decanoic, myristic, stearic and
oleic a,cids. Lower oxid¿tion rates were found
with butyric, octânoic and linolenic acids. (Fig. 2, Taþle
I).
M. tub€rculosts II37RV resistant to 5 uglml
INII
(TMC 303) showedtwo
distinct sets ofconsecutive
fatty
acidsv¡ith
high oddationMEfaBolrst or (t-lt)arn ncros ly M.lubdculo.ìÉ H-Rv ls ì
Fig. 1 - Iniluence of BsA-cåtatâse complex on the oxida,
tion lates of (l.lro) fatty acrds by M. ü¡bê¡cùrosis H¡rRv
tu]ly susceptible to drugs (TMC 102) in ?Ilg ûedium.
rates: octanolc, decanoic, lauric, and myristic acids; stearic, oleic and linoleic acids.
Inter-mediate oxidation rates occr¡lred with butyric,
hexanoic, palmitic and linolenic acids (Fig. 2,
Tabþ
f).
Except
for
but¡rric, hexanoic and linolenic acids, high oxid.ation rates were foundfor
allother fatty a.cids with M. bov¡s (¡Ig. 2, Table
I),
especially decânoic and octa¡oic.M. ka,lsasti shoeíred intermediate oxidation ¡ates with palmitic, oleic, linoleic and linolenic
acids.
Iligh
oxiaf.ation rates were found \r¡ith aU other fatw aclds, especially myristic (Fig. 3, TableI).
Ifexanoic acid wasnot
tested v¡ith this o¡ganism,I
contrast,lú,
chelorei showed low oxida"CAMARGO, E. E.; KOPÁÆIC, T. M,; HOPKINS, G. K.; CANNON, N, P, & WAGNER, JR., I{. N. _ RAdiOEEiliC SIU'IIEI
on the oxialqtion of (l-1ro) fatty sctdls by ¿kug.susceptible and drug-æsist€nt ßycobacte¡la. Ree. Irst. M€d. trolÈ
SÍ¡o Påulo, 29(1):9-1?, 198?.
rAB],E
I
Oxidation sttem.s of (r.11C) fatty aci¿ls by ¡nJcoùacteri¿'
E .e ã 9¿ ã
*
EÉ
çTS
É5E
5{t
Ë8 É5
Ép
5È
t243 ! 193 7243 t l8
180¡ f 315
1?86 + 350 r?51 + 298
1821 + 158
2049 È 36S
l5?8 É 210
2294 t 280 1?51 * 88
1138 + 158
1693 È 158
609 ! 90 1918 + 248 1580 f 203 2257 t 406 813 + 90
2t44 + 35+ 1625 r l8l
l1l5 r 226 2A92 t A
1151 t t3
1529 È 202
NT
2315 ! 202 1619 I 180
2?A8 r 135 ?8? + 90 2360 ! 247
u76 ! 180
2210 ! 202 2765 ). 562 152Ð + 180
1477 :r l4l ¡fI 1398 + 141
1118 + t88
15'11 + 204
t?44 + 15?
1021 + ?9
t650 I 189
880 È 204
91t + 94 927 t 283 669 :t 173" ?96 t 133 8?9 È 5? 228 I 41
626 + 151 2541 ! tA6 993 :! 191 249 ! 21
1941 :! 23? 1008 È 265 1815 i 96 1265 f 83
2330 I 216 1592 + 212 2139 i '16 $24 i 394
2t57 ! 2t6 2653 1 133 1910 r ra4 103? t ?3
2049 :t ¡51 1645 ! 239 1986 * 26? 1193 i 93
105? a 129 2334 ! 186 1261 I 134 1213 ! 145
1553 r 86 1565 + 159 l??6 t: tît 123+ r:218 ?98 i 86 r?5t È 106 196? I 210 913 + t35 669 f 86 2945 i 239 2¡01 f 191 12?6 t ?r
lr00 + 43 1141 i E0 l0?0 ! 153 570 + 104
Butyric flexenoic Octanoic Decanoic I¿urlc ¡ls¡istio Paknttic SteÀrtc Ol€ic I,i¡olelc Linolenic
' cumulativs '{cq Foduction over 3 dsys ss met¿bollc tndex units .. Mean + stanaL$d deYfÊülon
¡It = not tested
S = Susceptible to 3ll drugs (TMC 102)
R = Resistant to 5 úg/rÞ1 isoniâztd (TMC 303)
orùúr d h,Éclh!,¡êÈ.
br ! r!¡¡E¡¡¡r. Hsrir(.) ùÈbo,rÍÍclF.q¡.ú,
ùü.,h d !,úclf¡d,^cB.
I
il
rr
[l
ll
Éllnll¡ill
nllrlllllllllllllln
"*Li.Li.lJ".l-.l.Ll_.Li".lJ,.Ll"Ll*l-.1."-u-.
Å'
¡lltldr
Il||t
n
, I ||Èll,
n
ü ü
LlilllU
ll
lll
tlll
-l
L
I
11-
l1
-l¡
l ||i| -| lt
rlll¡rl
'ritl
fr
llilllrllrl
Illl
Èt-ruuuLlLl!|Lr!uLr
o,r. ." d h-ncrF.,,,¡. d,
br !,r!.ffi H3r¡r ri,
¡'ig. 2 - Pattem of (1.LC) fatty acid oxldation by M. lúberculostl H¡1Rv futly s$ceptible to cllugF (uppe! ¡€lt), M. tu. ùorculosls EftkE¡,Ã Ouppe¡ rtght), M, tubercDrodE .IIâ|BV resistãnt to 5 ugl¡il lsonla¿id (loÌsotr ¡eft), and ¡ú, bort€
(loPe¡ right). åll organiws wero te$ed l¡r liq1lld ?Eg medfiaE with l0% ÁDC er¡¡lchEent.
CÁMARGO, E. E.; KOP.{XTIC, T. M.; HOEKTNS, G. K.: CANNON, N, P, & WAGNER JR,., H. N. _ RådiOInEIliC STUd¡E6
oD ths oxitÞtion of (l.1ac) fatty act¿s by d¡ug'susceptihle snd drug-rcsistânt mycoþacl€lia. Aev. Inst. Med. trop.
São Pauìo. 29(l):9'U, 1987.
ú[,üxI
Å
il
ilil
uu
was chosen to elimiD¿te poorly oxidized subs.
trates.
TablefI
showsthe
resultsof
thisapproach.
!'ive converdent substrates ryere chosen in Table
ff,"!r'ith
the pu4)oseof
differentlatingthe o¡ganisms used in tlds study. Ttris simpler
approach included buwric, myristic, oleic,
li-nolenic and hexanoic (Table
III).
Ho\¡rever, as can be observed in TableII, it
was not possi-ble to separate M, tuberc.ulosts H3?Rv TMC 303from
llL
bov¡s..A,s expected, t{CO2 production was not ob-seNed in the control vlals and all sterility tests
were negative.
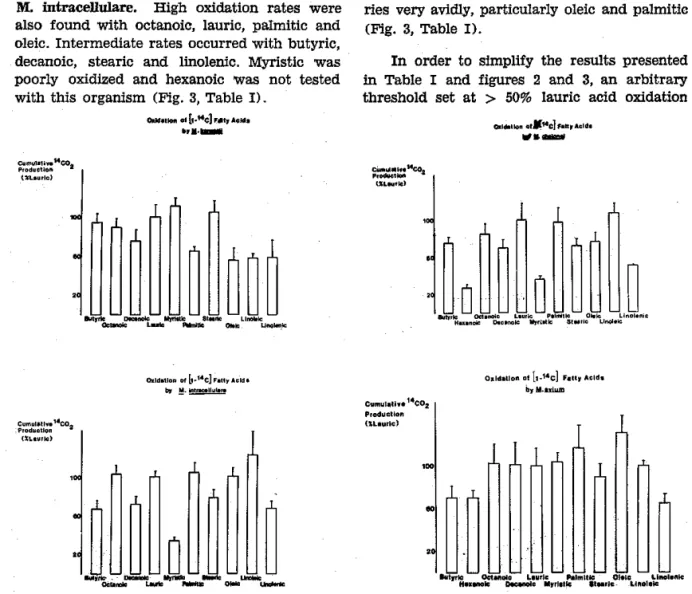
M. aviuln oxidized ihe entire fatty acid
se-ries very avidlJ, particularly oleic and palmitic (Fig. 3, Tâble
I).
In
order to stmplify the results presentedin
TableI
and figurcs 2 and 3, ¿n arbitrary threshold setsl >
50Eo lauric acid oxidationù'.'b.,FdÉ!,h0.
o,hù" d l,.rcl F.,e k,d.
DISCUSSION
Fatty aeid synthesizing en4¡me actidty h8s
been found
in
extractsof
severâl m¡¡cobate-rià,73,18,?.7,24,Æ,n. T?rese studles have shoÌgDthat a
vadetyof
long chainfatty
a.cids areformed, from 12 to 26 carbons, by addition of two carbons to the acceptor fatty acld's
carbo-ryl
end. Acetate, malonate and NADII arees-sential
for
these reactlons. Ttrereis
âlso evi. dence that M, úub€¡sulosÍs accumulates lipids durlng eqronentiel grcwtbr, with a
qmthesisof
rrycolic aßids that psrallels the grcrth
ra.te3. Some
of
the short and long chain fâttyaÆlds \Ãre¡e preferabty inco4)orated into trigly.
Linoleic acid was the best substrate with M, intracellrdare. High oxid-ation rates v¡ere
also found
with
octanoiq lauric, pÈlmitic and oleic. Intermediate rates occuûed with butyric, decanoic, stealic and linolenic. t\ilyristic was poorly oxidlzed and hexanoic \¡/as not testedwith this organism (Fig. 3, Table
I).
à!.'h dft'É4¿t¡"r'.r¡,¡rl
db,b. d L.nclrf,,¡.H.
iltl
ltilt
UL]l]
ú¡,lt
ilill]il
t]tlut]
Fig. 3 - Pattem of (liao) fatty âcld oxidabloD by M. k8!åsit (uppêr l€ft), M. ctelonel (upp€r dght), M. lDtrac€Ift¡.
la¡e (Iower reft), arl.l M. ¿vtùñ (lowe¡ ¡¡ght). All organi$r¡s were tested in liquid ?IIg mechur¡ with l0% 4ÐC e ichm€nt.
rl
ntn
CÄ¡trAEGO, E. E.; KOPÂJTIC, r. M-; HOPKINS, G. K-; CÁNNON, N P. & WAGNEE JR., I1. N. - Radiometnc $udie6
on tho oxialetion of (1.úC) f¿tiy acia¡s l¡y d¡ug.susceptible and al¡ug'¡esistânt mycobacte a. Rev. IBt. M€al. túIL Sõo P¿ùto, 29(1):9-r?, $8?.
T.{Bf,EII
(t.t{) lzlty acials with sicBiJicani oxrdâtion retes
0 0 + + + + 0 + 0 0 0
+ = > 50% acid oxrdation
0 = < $7¿ laúlc oxidafion
S: &rscep¿ible to all d¡ugs (TMC 102)
& = Resistanl ro 5 ug/Eil isoDiazid (TMc 303)
TABI,E III
OlidÂtion patteFs of selected (F¡C) fatty âctcls by
mycoþacteriå øt
332É
? ? ?
;E
AÈå
Èe
åË
Ë
Ë
Ë
å
ËÞ! =5 E: E E > E
EBuby¡ic My¡istic Oleic Linotênic qeÎânoic
(H3zRv) ând M. bovis
fißt
Ínüeased s¡ith the carbon châin lengthto a
maximum çi'it}I de-canoic and then decreasedlz. On the other hand,KONDO and I<.A.NAI 17, also studied the
bac-tericital effect
of
fatty
acids and found that myristicacid
acidhad
tlle
strongest letheleffect on M. tuberculosis H37Rv arid on M. bo.
vis: longer and shorter chain fatty acids were less toxic than
myristic.
Such toxic effects ora
possible inhibitory effect on bacterial enzy-mes involvedin
lipogenesisþ
probabll¡ wouldnot
occurwith
the small amountsof
fatwacids used
in
ou¡ exlreriments (approximately4
x
10-8M per vial). ltrelefore, one could only speculate Ìvhetheror
not the eD4¡me systemsof
the organisms includedin
our expe ments are seDsitive enough to recognize potentially to-xic substances and either transform them into non-toúc triglyceridesor
excrete them as by-products.Long chain fatty acids have been sho¡e¿n to have a stimulatory effect on oxygen uptahe by
M, tuberculosis, -á. sLnllar mechanism could be present
in
other mycobacteria and wouldex-plain the various oxidation rates observed in
our
expeliments.ft
is
also conceivable that the mâgnitudeof
the stimulatory effect vari€sfrom
one organismto
anotherfor
the sameIatty acid. Ttrerefore, the preferential oxidation
of one or more substrates over the others by
a particular mycobacteria could be due to the toxic nature of the substrate, to the stimul¿tory effect on oxygen uptâke,
to
a combinationol
both or because the subst¡ate,
for
sometea-son,
is
pa
icularly
suitablefor
the
energy needsof
the organism,Assimilation studies \Ã'ith M. lepraDn ¡rium
and M. tubolculosts H37Rv fully susceptible to
drugs 6'7 allswed us to identily fatty acids used
p
marih¡as
enerry so]Irc€s and those used primarily as ca¡bon sources. We also foundthat adsorption of the fattj¡.acids of higher
mo-lecular weight such as lauric, aJ¡ristic, palÍri.
tic
and stearic could have been a determinantof
the assimilation process.[,ith
M.tubercu-losis most of the substrates were used as both
carbon sources and energ¡ sources, whereas
lauric and formate were used chiefly to provi-de energy. lryith
i[.
IeplseEr¡Ìium, lãuric wasthe best energy source and carbon-2,
but
not carbon-l of acetate the best carbon source, Be-cBuse assimilation studies wittr râdiochlomato. Buhdc ¡Iexanolc Ðecanoic MFistic PaIEri¿ic SteaÌic Oteic I,jnoleic tinolenic0 0 0 ++
++ 0 0 NTo
NT0 + + ++
++ + + ++
++ + + ++
++ + + +0
0+ + + ++
++ + + ++
++ + + 0+
++ + + 0+
+00000+
+ 0 0 N]T 0 Nf -ft
+ + + + + + + l-(Ir¡,n,v-S) M. tuþeroulosig(EftlmåD) M. tr¡hercu¡osl8
(Hs?Rv-R)
M. Irovts
M. k¡rsesü
M. chelo¡€i
M. intrac€llül¡!€
M. ¿vium
0 + + + 0 0 0 + + + +
++0
+00
0+0
0++
+++
S = susceptible to all drugs (TMC 102)
R = ¡€sj.stent to 5 nglrEl isonisøid (TMC 303)
cerides re,
or
into phospholipids 2r arld retainedin
the cell whereas someof
them wetetmns-formed into by-ptoducts a¡d excreted into the mediumó'e.
The excretion of by-products into the
me-dium may represent ¿ mechariÍsm
of
elimina-tion of toxic substances. An example
of
such a toxic effect was observedwitlì
oteic acid: in high concentrationit
inhibited the$owth
of tubercle bacilli,but
in
low
concentrationit
enhanced growth
of
this organism, as didbu-tyric
a¡d
decanoic12.
T'his inhibitory effectcaused