Characterization of Antimicrobial Photodynamic
Therapy-Treated
Streptococci mutans
:
An Atomic Force Microscopy Study
Mary Anne Sampaio de Melo, DDS, MSc,1Juliana Paiva Marques Lima Rolim, DDS, MSc,1 Iriana Carla Junqueira Zanin, DDS, MSc, PhD,1 Eduardo Bede Barros, BS, MSc, PhD,2 Erivelton Fac¸anha da–Costa, BS, MSc, PhD,2 Lidiany Karla Azevedo Rodrigues, DDS, MSc, PhD1
Abstract
Objective:The aim of the present study was to examine the size and shape ofStreptococcus mutansbacterial cells of infected dentin substrate subjected to photodynamic therapy (PDT) using atomic force microscopy (AFM). Background data:New trends in the application of AFM have been developed in the field of dentistry, making AFM a useful technique in high resolution imaging of biological structures and processes. Materials and methods:PDT was completed using an efficient light-emitting diode source (LED -k=620–660 nm) with total light dose of 94 J/cm2in the presence of the photosensitizer toluidine blue O (TBO). Dentin specimens were immersed in brain heart infusion (BHI) broth inoculated withS. mutansfor 5 days to induce cariesin vitro. After demineralization, the samples were subjected to a series of treatments in which carious dentin infected byS. mutans was exposed to 0.9% sodium chlorite (NaCl) solution (control) for 10 min, or subjected to PDT-TBO photosensitizer followed by light exposure (energy density of 94 J/cm2).Results: Three-dimensional (3-D) im-ages and cross-sectional measurements showed rod and diplococcic cell shapes. Photoinactivated bacterial cells did not differ from the control with respect to their cross-sectional shape, but they did show a reduction in size. Conclusions: Photodynamic therapy decreased the diameter of S. mutans cells and AFM may be used as a technique for bacterial cell analysis.
Introduction
T
he relatively rapidacquisition of antibiotic resistance among pathogenic bacteria and the time consumed in developing novel antibiotics have motivated the develop-ment of alternative methods to fight bacterial infections.1,2 Photodynamic therapy (PDT) consists of a photosensitizer combined with light of a specific wavelength to generate cytotoxic elements, including singlet oxygen and free radi-cals.3These products are capable of damaging essential cell components or modifying metabolic activities in an irre-versible way, which may result in bacterial cell death.4PDT has been considered a developing, novel, convenient, and inexpensive method of killing pathogenic bacteria, and has been applied as a powerful approach against biofilm-asso-ciated infections.5Dental caries is a common, worldwide disease caused by acidogenic bacteria and their biofilms upon exposure to fermentable carbohydrates.6Specific types of acid-producing
bacteria, especially Streptococcus mutans,have a central role in the etiology of dental caries. These bacteria adhere to the enamel salivary pellicle and other plaque bacteria, creating a higher risk of developing cavities.7,8
Several studies have demonstrated that PDT is effective against oral microorganisms related to dental caries, killing a significant amount of S. mutans in planktonic cells or bio-films.9–13 Few studies have investigated the antimicrobial effects of PDT in carious dentin tissue, primarily in human dentin.14–16
It has been proposed that PDT causes harm to the cyto-plasmic membrane, which results in damage to the bacteria. This may cause leakage of cellular content or inactivation of membrane transport systems and enzymes. There is a good amount of evidence suggesting that PDT induces modifica-tions in bacterial components.17,18 Previous studies have shown cell alterations, disturbance in cell wall synthesis, the appearance of multilamellar structures near the septum of dividing cells,19,20 and the loss of bands that mark the
1Post-graduation Program, Faculty of Pharmacy, Dentistry and Nursing, Federal University of Ceara´, Fortaleza, Ceara´, Brazil. 2Physics Department, Program of Post-graduate Studies in Physics, Campus do Pici, Fortaleza, Ceara´, Brazil.
ªMary Ann Liebert, Inc. Pp.105–109
DOI: 10.1089/pho.2012.3377
boundary between "old" and "new" wall and ring the equator of the forming cell (wall bands).15
Most of these analyses are performed with optical mi-croscopy techniques such as confocal mimi-croscopy or electron microscopy. However, studying morphological structure at the molecular level by optical microscopy is difficult, because of its limited resolution and the damage incurred by samples during the pretreatment required for scanning electron mi-croscopy (SEM) imaging.21 By contrast, atomic force mi-croscopy (AFM) facilitates examination of the ultrastructure of biological samples at the molecular level using high-resolution images. This approach has also been applied to examine the morphology of S. mutans biofilm on different modified surfaces such as gold.22 AFM is a technique that requires fewer pretreatment steps, inducing little or no structural change.23To date, very little work has been done to research surface alterations on the structure of bacterial cells in a carious dentin environment after PDT.
This way, the aim of this investigation was to explore the changes on S. mutans housed within artificially deminer-alized dentin using AFM imaging, and to determine possible morphological alterations after photoinactivation.
Materials and Methods
Bacteria
AnS. mutansstrain (CTT-3440) was exposed to a micro-biological model for production of a caries-like dentinal le-sions.15S. mutanswas first grown in an overnight culture of brain heart infusion (BHI) (Difco Lab. Detroit, MI) in a 10% CO2 atmosphere (Thermo Fisher Scientific Inc., Waltham, MA) at 37C.
Specimen preparation
For substrate preparation, dentin specimens were ob-tained from impacted human third molars with more than two-thirds root formation complete. The specimens were free of apparent caries lesions and stored in a 0.01% (w/v) thy-mol solution at 4C for 1 month, then refrigerated until use.
Ten dentin specimens (5·5·2 mm3) were obtained using a
water-cooled diamond saw for cutting (IsoMet Low Speed Saw, Buehler, Lake Bluff, IL). Afterwards, all specimens were sterilized by autoclaving according to Amaechi et al24 and stored in 100% humidity until immersion in sterile BHI containing 5% sucrose (w/v).
Experimental design
The use of extracted human teeth and the study protocol was approved by the Research and Ethics Committee of Federal University institution (Process #. 143/2006).
All BHI-containing recipients were inoculated with 80lL of 1–2·108 colony forming units (CFU)/mL of standard
overnight cultures ofS. mutansadjusted to a standard optical density by spectrometry. The number of viable cells was determined with a spectrophotometer (Amersham Bios-ciences Ultrospec 1100 pro, GE Healthcare do Brazil Ltda, Sa˜o Paulo, Brazil) using a wavelength of 600 nm and an optical density of 0.3120. Inoculation of each BHI-containing recipient was performed only once on the first day of the experiment, and the dentin specimens were transferred to a fresh medium every day for 5 days.
PDT
After a 5-day experimental period, PDT was performed on carious dentin tissue after removing a significant portion of the biofilm that had formed on the specimens. This study involved two treatment conditions. In one group (n=5), dentin infected
byS. mutanswas exposed to 0.9% sodium chlorite (NaCl) so-lution for 15 min (control) in order to simulate pre-irradiation and irradiation time conditions. In the second group (n=5), the
dentin specimens were subjected to PDT (toluidine blue O [TBO] plus light-emitting diode [LED]). The specimens in the PDT group were treated by immersion in a solution of 5lL of TBO photosensitizer (Sigma-Aldrich Company Ltd. Poole, Dorset, United Kingdom) dissolved in deionized water at a final concentration of 0.1 mg/mL. After exposure to TBO, specimens were placed in darkness for 5 min (pre-irradiation time) followed by light irradiation for 10 min.14A LED (La-serbeam, Rio de Janeiro, RJ, Brazil), with a spectrum of emis-sion ranging from 620 to 660 nm and a predominant wavelength of 638.8 nm, was used as the treatment light source. A fiber optic device with a 9.5 mm cylindrical diffusing tip distributed the light. Irradiation was performed without making direct contact using a focused beam at a working dis-tance of 2.0 mm, allowing an incident energy dose of 94 J/cm2. A Lasermate power meter (Coherent Inc., Santa Clara, CA) was used to measure the maximum output power of 40 mW.
AFM
An AFM analysis was performed on infected dentin specimens in order to verify changes in theS. mutans mor-phology of treated and untreated bacterial cells. The images were obtained using a Nanoscope IIIA Bioscope (Veeco Di-gital Instruments, Santa Barbara, CA) (Fig. 1A). Information was collected through contact using sharpened silicon nitride cantilevers (OTR4 Veeco probes) with experimentally deter-mined spring constants of 0.15 nm and a nominal tip radius of <20 nm, as defined by the manufacturer (Veeco). Probes
were kept in their original sterile container as prepared during standard manufacturing techniques (Veeco) and vi-sually examined for debris before use. All measurements were obtained at room temperature (18C) with force
mea-surements recorded at a scan rate of 0.5 Hz.
The size measurement treated the point of inflection as the edge of the bacterium. Diameter of the bacteria was collected on single mechanically trappedS. mutanscells by lowering the cantilever tip toward the cell, pressing against the cell surface, and retracting the tip from the cell as schematically shown in Fig. 1B.
Statistical analysis
The qualitative analysis of the cells consisted of two diam-eter measurements made on three individual cells in each case. The image analysis system supplied by Shimadzu Co. Ltd. was used to measure the width directly from the image. The cross-sectional AFM images of bacterial cells show a sigmoidal curve at the edge (Fig. 1C). The mean values were subjected to ANOVA and Tukey tests at a 5% significance level.
Results
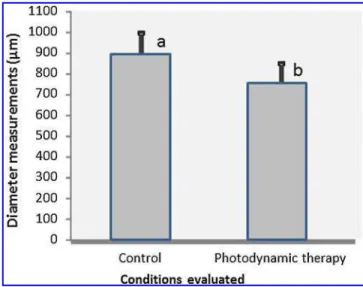
ultrastructure ofS. mutanscells at the molecular level. The morphological analysis completed using AFM revealed that the dentin specimens appeared to be infected with an abundant amount ofS. mutans, as shown in Figs. 2A and B. This measurement was performed in high resolution with a height scale of 700 nm as illustrated in three-dimensional (3-D) images. Bacteria are typically rod-shaped, with a diameter of 0.7–0.8lm (Fig. 2C and D). No morphological changes were noted in the PDT-treated bacteria; however, data from these samples revealed a significant decrease in diameter as compared with the untreated bacteria (control group), as shown in Fig. 3 (p<0.05).
Discussion
In this study, it was demonstrated that AFM analysis is a practical approach to examining the bacterial surface of PDT-treated S. mutans in a carious environment. AFM has emerged as a powerful technique for studying ultrastructure in the field of microbiology. It provides high-resolution im-ages without causing damage during sample preparation. AFM targets single bacterium and their components, in-cluding proteins and membranes, and records their physical properties, such as elasticity and adhesion forces.22, 23
Because AFM is able to perform topological analysis,25,26it is useful in examining biological samples subjected to pho-todynamic therapy. One of the primary photoinactivation mechanisms is damage to cell structures such as the cell membrane or cytoplasmic components, which can be visu-alized using AFM. Another advantage of AFM is the po-tential for bacterial size determination, as this measurement has been enabled primarily by analyzing images taken using SEM,27 which is usually a very laborious and prolonged process.
Our AFM results suggested a reduction in size of PDT-treatedS. mutans, a gram-positive bacterial species. There is a lack of research related to the alteration caused by PDT in caries-related bacteria in demineralized dentin. Similar re-sults were previously obtained27 when a bacterial size re-duction was observed inS. aureusstrains treated with 25lg/mL of hematoporphyrin monomethyl ether in combination with light illumination (power density: 200 mW/cm2).S. Aureusis also a facultative anaerobic, gram-positive, coccal bacteria. Further, the mean diameter of the PDT-treatedS. aureusalso significantly decreased compared with those of untreated
S. aureus. These data indicate that theS. aureuscell envelopes had a smoother surface texture after PDT. One might be in-clined to believe that these changes are related to structural alteration, indicating that there might be cytoplasm or other FIG. 1. (A) Image of the atomic force
mi-croscope used in this study. (B) Schematic representation of the microscope tip interact-ing with the cell surface. (C) Cross-section analysis of two diameter measurements.
FIG. 2. (A) Atomic force microscope (AFM) imaging of photodynamic therapy (PDT)-treated dentin specimens in-fected with Streptococcus mutans. (B) Image of untreated bacteria. (C) Representative image of PDT-treated bacteria for measurement analysis.(D)Representative image of un-treated bacteria for measurement analysis.
FIG. 3. Comparison of mean diameter measurements of the conditions evaluated. Data are expressed as the mean–standard deviation (n=20). Mean values represented
cellular contents leaking out of the bacteria. According to Plaetzer et al.,28the peroxidation of lipids and proteins in cell membranes causes a loss of membrane integrity and ion homeostasis, and might, therefore, be the primary explana-tion for the occurrence of necrosis.
AFM data should be analyzed and compared with sup-plemental measuring techniques to verify the reliability of size measurement by AFM. Images may appear larger than actual size because the cantilever requires a minimum mar-gin and cannot trace the surface of materials precisely at the edge.26 Combinatory imaging using AFM with confocal microscopy has been used for examination of biological materials with high resolution, and may notably contribute to the evaluation of the effects of PDT on bacteria.29
Conclusions
It was demonstrated through a high resolution imaging mode (AFM) that PDT affectsS. mutanshoused within arti-ficially demineralized dentin. Within the limitations of this study, it can be concluded that AFM analysis may be a complementary investigative approach to exploring effects on PDT-treated bacteria and cells, and that AFM may pro-vide detailed insight into understanding the process of bac-terial damage.
Acknowledgments
The authors thank the Institute of Research, Development and Innovation (IPDI) for providing the AFM equipment.
Author Disclosure Statement
No competing financial interests exist. The first author received scholarship from CNPq during this study.
References
1. Maisch, T., Hackbarth, S., Regensburger, J., et al. (2011). Photodynamic inactivation of multi-resistant bacteria (PIB) – a new approach to treat superficial infections in the 21st century. J. Dtsch. Dermatol. Ges. 9, 360–366.
2. Jori, G., and Brown, S.B. (2004). Photosensitized inactivation of microorganisms. Photochem. Photobiol. Sci. 3, 403–405. 3. Kurek, A., Grudniak, A.M., and Kraczkiewicz-Dowjat, A.,
et al. (2011). New antibacterial therapeutics and strategies. Pol. J. Microbiol. 60, 3–12.
4. Hamblin, M.R., and Hasan, T. (2004). Photodynamic thera-py: a new antimicrobial approach to infectious disease? Photochem. Photobiol. Sci. 3, 436–450.
5. Wilson, M. (2004). Lethal photosensitisation of oral bacteria and its potential application in the photodynamic therapy of oral infections. Photochem. Photobiol. Sci. 3, 412–418. 6. Marsh, P.D. (2010). Microbiology of dental plaque biofilms
and their role in oral health and caries. Dent. Clin. North Am. 54, 441–54.
7. Forssten, S.D., Bjo¨rklund, M., and Ouwehand, A.C. (2010). Streptococcus mutans, caries and simulation models. Nu-trients 2, 290–298.
8. Takahashi, N., and Nyvad, B. (2011). The role of bacteria in the caries process. Ecological perspectives. J.Dent. Res. 90, 294–303.
9. Stringer, G.J., Bird, P.S., and Walsh, L.J. (2000). Lethal laser photosensitization of Streptococcus mutans with a visible red diode laser. Aust. Dent. J. 45, Suppl. S–22.
10. Paulino, T.P., Ribeiro, K.F., Thedei, G., Jr, et al. (2005). Use of hand held photopolymerizer to photoinactivate Estrepto-coccus mutans. Arch. Oral Biol. 50, 353–359.
11. Williams, J.A., Pearson, G.J., Colles, M.J., et al. (2003). The effect of variable energy input from a novel light source on the photoactivated bactericidal action of tolu-idine blue O on Streptococcus mutans. Caries Res. 37, 190–193.
12. Bevilacqua, I.M., Nicolau, R.A., Khouri, S., et al. (2007). The impact of photodynamic therapy on the viability of Strep-tococcus mutans in a planktonic culture. Photomed. Laser Surg. 25, 513–518.
13. Costa, A.C., Chibebe Junior, J., Pereira, C.A., et al. (2010). Susceptibility of planktonic cultures of Streptococcus mu-tans to photodynamic therapy with a light-emitting diode. Braz. Oral Res. 24, 413–418.
14. Lima, J.P.M., Sampaio de Melo, M.A., Borges, F.M.C., et al. (2009). Evaluation of the antimicrobial effect of photody-namic antimicrobial therapy in an in situ model of dentine caries. Eur. J. Oral Sci. 117, 568–574.
15. Melo, M.A.S., De-Paul, D.M., Lima, J.P.M., et al. (2010).In vitrophotodynamic antimicrobial chemotherapy in dentine contaminated by cariogenic bacteria. Laser Physics. 20, 1504–1513.
16. Monteiro–Oliveira, M.P., Rodrigues, L.K.A., Sampaio de Melo, M.A., et al. (2010). Photodynamic therapy effect in carious bovine dentin—an in vitro study. J Oral Laser Applications. 10, 29–36.
17. Jori, G. (2006). Photodynamic therapy of microbial infec-tions: state of the art and perspectives. J. Environ. Pathol. Toxicol. Oncol. 25, 505–519.
18. Jori, G., and Coppellotti, O. (2007). Inactivation of patho-genic microorganisms by photodynamic techniques: mech-anistic aspects and perspective applications. Antiinfect. Agents Med. Chem. 6, 119–131.
19. Valduga, G., Breda, B., Giacometti, G.M., et al. (1999). Pho-tosensitization of wild and mutant strains of Escherichia coli by meso-tetra (N-methyl-4-pyridyl) porphine. Biochem. Biophys. Res. Commun. 256, 84–88.
20. Nitzan, Y., Gutterman, M., Malik, Z., et al. (1992). Inactiva-tion of Gram negative bacteria by photosensitized porphy-rins. Photochem. Photobiol. 55, 89–96.
21. Schwarz, H., and Humbel, B.M. (2008). Preparation of biological samples for electron microscopy. EMC. I6, 799–800.
22. Yifan, H., Zhang, J., and Ulstrup, J. (2011). Investigation of Streptococcus mutans biofilm growth on modified Au (1 1 1)-surfaces using AFM and electrochemistry. J Electroanal. 656, 41–49.
23. Van Hoogmoed, C.G., Dijkstra, R.J.B., Van der Mei, H.C., et al. (2006). Influence of biosurfactant on inter-active forces between mutans streptococci and enamel measured by atomic force microscopy. J. Dent. Res. 85, 54–58.
24. Amaechi, B.T., Higham, S.M., and Edgar, W.M. (1998). Ef-ficacy of sterilization methods and their effect on enamel demineralization. Caries Res. 32, 441–446.
25. Jung, S.H., Park, J.Y., Yoo, J.O., et al. (2009). Identification and ultrastructural imaging of photodynamic therapy-induced microfilaments by atomic force microscopy. Ultra-microscopy 109, 1428–1434.
atomic force microscopy. Appl. Microbiol. Biotechnol. 88, 761–770.
27. Boyde, A., and Williams, R.A.D. (1971). Estimation of the volumes of bacterial cells by scanning electron microscopy. Arch. Oral Biol. 16, 259–266.
28. Plaetzer, K., Kiesslich, T., Verwanger, T., and Krammer, B. (2003). The modes of cell death induced by PDT: an over-view. Med Laser Appl 18, 7–19.
29. Nishino, T., Ikemoto, E., and Kogure, K. (2004). Application of atomic force microscopy to observation of marine bacte-ria. Journal of Oceanography 60, 219–225.
Address correspondence to:
Lidiany Karla Azevedo Rodrigues Department of Dentistry Faculty of Pharmacy, Dentistry and Nursing, Federal University of Ceara, Fortaleza Cap. Francisco Pedro Street - Rodolfo Teo´filo Ceara 60430-170 Brazil