www.elsevier.pt/ge
CLINICAL
CASE
Olmesartan-Induced
Enteropathy:
An
Unusual
Cause
of
Villous
Atrophy
Marta
Eusébio
a,∗,
Paulo
Caldeira
a,
Artur
Gião
Antunes
a,
André
Ramos
a,
Francisco
Velasco
a,
Jesús
Cadillá
b,
Horácio
Guerreiro
aaGastroenterologyDepartament,CentroHospitalardoAlgarve,EPE,Faro,Portugal bPathologyDepartment,CentroHospitalardoAlgarve,EPE,Faro,Portugal
Received29July2015;accepted14September2015 Availableonline20October2015
KEYWORDS Atrophy; Gastrointestinal Diseases; AngiotensinII Receptor Antagonist/adverse effects
Abstract Wereportacaseofa63-year-old-manpresentingwithchronicdiarrheaandweight losswhileonolmesartantreatmentforhypertension.Investigationshowedmultiplenutritional deficiencies associated with diffuse intestinalvillous atrophy. Serologies for celiac disease werenegativeandothercausesofvillousatrophywereexcluded.Olmesartanasaprecipitant agentwassuspectedandwithdrawn.Clinicalimprovementoccurredindayswithnoneedfor othertherapeuticmeasures.Follow-upatthreemonthsshowedclinicalremissionandalmost completerecoveryofintestinalatrophy.
Olmesartanisanangiotensinreceptorblockercommonlyprescribedforthemanagementof hypertension. Spruelikeenteropathyassociatedwith thisdrug isarecentlydescribed entity withfewcasesreported.Itpresentswithchronicdiarrheaandintestinalvillousatrophyand shouldbe includedinitsdifferential diagnosis.This caseintendstoalert clinicians forthe possibilityofthiseventinapatientontreatmentwiththisdrug.
© 2015 Sociedade Portuguesa de Gastrenterologia. Published by Elsevier España, S.L.U. ThisisanopenaccessarticleundertheCCBY-NC-NDlicense(http://creativecommons.org/ licenses/by-nc-nd/4.0/). PALAVRAS-CHAVE Atrofia; Doenc¸as Gastrointestinais; Antanosistasdos Recetoresda Angiotensina II/efeitosadversos
EnteropatiaInduzidapeloOlmesartan:UmaCausaIncomumdeAtrofiaVilositária
Resumo Apresentamosocasodeumhomemde63anoscomdiarreia crónicaeperda pon-deral.Apresentavahipertensãoarterialtratadacomolmesartan.Ainvestigac¸ãocomplementar mostroumúltiplosdéficesnutricionaisassociadosaatrofiavilositáriaintestinaldifusa.As serolo-giasdedoenc¸acelíacaforamnegativaseoutrascausasdeatrofiavilositáriaforamexcluídas. Suspeitou-sedoolmesartancomoagenteprecipitante,sendoestesuspenso.Observou-se mel-horiaclínicaemdias,semnecessidadedeoutrasmedidasterapêuticas.Noseguimento,aos3 meses,constatou-seremissãoclínicaerecuperac¸ãoquasecompletadaatrofiaintestinal. ∗Correspondingauthor.
E-mailaddress:martaeusebio@gmail.com(M.Eusébio).
http://dx.doi.org/10.1016/j.jpge.2015.09.005
2341-4545/©2015SociedadePortuguesadeGastrenterologia.PublishedbyElsevierEspaña,S.L.U.Thisisanopenaccessarticleunderthe CCBY-NC-NDlicense(http://creativecommons.org/licenses/by-nc-nd/4.0/).
Oolmesartanéumbloqueadordosrecetoresdaangiotensina,geralmenteprescritono trata-mentodahipertensão.A enteropatia‘‘spruelike’’ associadaaestefármacoéumaentidade recentementedescrita,compoucoscasosreportados.Manifesta-sepordiarreiacrónica asso-ciadaaatrofiavilositáriaintestinal,devendoserincluídanoseudiagnósticodiferencial.Com estecaso pretende-sealertarosclínicos para apossibilidadedeste eventoem doentessob tratamentocomestefármaco.
© 2015 Sociedade Portuguesa de Gastrenterologia. Publicado por Elsevier España, S.L.U. Esteé umartigo Open Access sobalicença deCC BY-NC-ND(http://creativecommons.org/ licenses/by-nc-nd/4.0/).
1.
Introduction
The most common cause of villous atrophy is celiac disease.1,2 The villous atrophy results from injury to the
smallintestineandleadstolossofabsorptivesurfacearea, reductionofdigestiveenzymes,andconsequentialimpaired absorptionof micronutrients.3 Negativeceliacserology or
nonresponse to a gluten-free diet implies a broad and challengingdifferentialdiagnosiswhichincludesCrohn’s dis-ease,entericinfections(e.g.Giardialamblia),collagenous sprue,tropicalsprue,commonvariableimmunodeficiency, autoimmuneenteropathy, hematologicalmalignanciesand medication-associated enteropathy.2 Regarding the latter,
olmesartanmedoxomil,anangiotensinreceptorblockerfor themanagementofhypertension,hasbeenrecently recog-nizedasacauseof‘‘sprue-likeenteropathy’’.4---6
Wereportacaseofsevereenteropathyassociatedwith olmesartanuse.
2.
Clinical
case
A63-year-old manwasadmitted toour department com-plainingofprogressivediarrheaandsignificant weightloss (12kg) for one year. He reported between 5 and 7 daily episodesofbulky,wateryandnonbloodydiarrhea.Overthe precedingtwoweeks,itwasassociatedwithseverefatigue, anorexiaandvomiting.Hedeniedabdominalpain,feveror othersymptoms.Therewasnohistoryofrecenttravelsor sickcontacts.
Apartfromarterialhypertension,treatedwith olmesar-tanandhydrochlorothiazide(20/12.5mg)fortwoyears,his pastmedicalhistorywasunremarkable.
The patient had already undergone total colonoscopy andabdominalcomputed tomography withnoremarkable findings.Aglutenandlactose-freedietweretriedwithout improvement.Healsofailedinitialconservativetreatment withatrial oforal antibioticforpossiblesmallbowel bac-terialovergrowth.
Onpresentationatourdepartment,hisbodymassindex was20kg/m2(normal(N):18.5---24.99kg/m2),closetothe lowerlimitofnormal.Musclewastingwasalsoseenwithout evidentweakness.Therewasnoperipheraledemaorother relevantfindingsonphysicalexamination.
Laboratory evaluation revealed: hemoglobin 11.6g/dL (normal:13---17)withnormalmeancorpuscularvolumeand meancorpuscularhemoglobin;serumpotassium1.8mmol/L (N: 3.6---5.1), phosphorus 1.7mg/dL (N: 2.3---4.7), magne-sium1.3mg/dL(N:3.6---5.1),corrected calcium8.6mg/dL
(N: 8.8---10); albumin 2.9g/dL (N: 3.4---4.8), total protein 4.9g/dL(N:6.2---8.5),aspartateaminotransferase205UI/L (N: 5---34), alanineaminotransferase 106UI/L (N<55) and proteinC-reaction25mg/L(N:<5mg/L).Theprothrombine time(PT)wasincreased(26.6s;N:9.4---13)aswellas acti-vatedpartialthromboplastintime(aPTT)(50s;N:20---40).
Other laboratory work-up was unremarkable including leucogram, serumglucose, B12vitamin, folic acid, trans-glutaminase antibodies, serum immunoglobulins, thyroid stimulating hormone andserologyfor human immunodefi-ciency virus. Platelets,bleedingtimeandfibrinogen were alsonormal.
Other causes for hypertransaminasemia were addition-ally excluded (no alcohol consumption; bilirubin, alkaline phosphatase,gamma-glutamyltranspeptidase,serumiron, ferritin,transferrinsaturation,cholesterolandtriglycerides were normal;hepatitis Band Cserologies werenegative; antinuclearantibodiesandsmoothmuscleantibodieswere negativeandabdominalultrasoundexcludedliverorbiliary abnormalities).
Stoolexaminationnamelycultures,Clostridiumdifficile toxinassay,ovaandparasiteswasunrevealing.
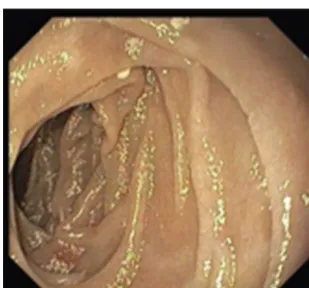
A colonoscopy was repeated and, despite all efforts, theterminalileumcouldnotbeintubated.Colonicrandom biopsies excluded microscopic colitis or other abnormali-ties.Upperendoscopyevidencedadiscreteattenuationof duodenal villous pattern without other findings (Fig. 1).
Figure1 Initialupperendoscopyshowingadiscrete attenu-ationofvillouspatternofthesecondportionoftheduodenum.
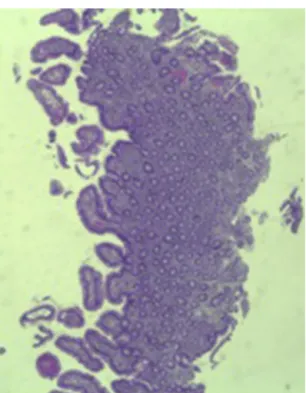
Figure2 Smallintestinalbiopsyshowingvillousatrophyand chroniclymphocyticinfiltrationofthelamina propria (hema-toxylinandeosin,4×).
Histopathological examination confirmed a partial villous atrophy and chronic lymphocytic infiltration of the lam-inapropria(Fig.2).Capsuleendoscopywasperformedand demonstrateda diffuse flattening of thesmall bowelvilli (Fig.3).
We suspected of olmesartan-associated sprue-like enteropathy.Thisdrugwasthereforewithdrawnalongwith replacementof electrolytesandvitaminKadministration. Promptimprovementwasachievedwithinafewdays.One weekafterhospitaladmission,thepatientwasdischarged withoutdiarrheaorneedfornutritional/electrolytesupport and began to gain weight. Olmesartan was switched to amlodipine.
Three months later, a complete recovery of weight (12.5kg) was seen along with full normalization of lab-oratory tests (hemoglobin, electrolytes, albumin, TP, aPTT, protein-C reaction and aminotransferases). Upper
endoscopy and capsule endoscopy (Fig. 4) were, again, performed and showed normal small bowel appearance. Histopathologicalanalysis of duodenal biopsies confirmed analmostcompleterecoveryofduodenalvilliandno lym-phocyteinfiltration (Fig.5).Atsixthmonthfollow-up,the patientremainedasymptomatic withnolaboratory abnor-malities.
3.
Discussion
Wedescribed a case of a patient presenting withchronic diarrheaandmalabsorptionasevidencedbymultiple nutri-tionaldeficitsincludingelectrolyteimbalanceandreduced serum albumin. Prolonged PT and aPTT in a patient not taking vitamin K antagonists, with no evidence of liver disease, biliary obstruction or disseminated intravascular coagulation suggested, in this clinical setting, vitamin K malabsorption. In addition, villous atrophy was present throughouttheentiresmallbowelasdemonstratedby cap-suleendoscopy,whichexplainsthemalabsoption.
Inourcase, celiacdisease,themost commoncauseof villous atrophy,1,2 was excluded by serology methods and
the lack of clinical response to a gluten-free diet. After excludingothercausesofvillousatrophy,weconsideredan olmesartan-associatedenteropathy.
Olmesartan medoxomil is an angiotensin II receptor blockerapprovedfor the treatmentof hypertension since 2002.7 A sprue-like enteropathy associated with
olmesar-tanwasfirstreportedbyRubio-Tapiaetal.4andsincethen,
similarcaseshavebeendescribed,althoughmainlyascase reportsorsmallcaseseries.6,8---15Asaresult,UnitedStates
FoodandDrugAdministrationreportedthisolmesartan asso-ciatedadverseeventviaaMedWatchalertinJuly2013.
Clinicalpresentationofthisentityincludechronic diar-rhea,vomiting,abdominalpain,bloating, weightlossand fatigue.4,6,11Moreseverecaseswithdehydration,4,9,13acute
renalfailure9andacaseofcolonicperforation11havebeen
reported.Accordingtoprevious descriptions,theduration ofexposuretoolmesartanbeforetheonsetofdiarrheahas variedbetweenseveralmonthsandyears.4,6Inourcase,it
tookoneyeartopresentsymptoms,whichisinaccordance withthetimingreported.
Laboratory investigation may show normocytic, nor-mochromicanemia,hypoalbuminemiaandoneor multiple
Figure4 Follow-upcapsuleendoscopyshowingnormalsmallbowelappearance.
electrolyteabnormalities,4asevidencedinourcase.Human
leukocyteantigen(HLA)assessment,whenperformed,may revealahigherprevalenceofDQ2or DQ8haplotypesthan expectedforthegeneralpopulation,whichsuggestsa pos-sibleroleforgeneticsinthisenteropathy.4,6
Upperendoscopymaybenormal,showanodular appear-anceofthe duodenalmucosaor flatteningof villi.6Inour
case, onlya discreteattenuation of duodenalvillous pat-ternwasobserved.Capsuleendoscopy,however,highlighted adiffuseandobviousflatteningofthesmallbowelvilli.
Themostcommonhistopathologicalfindingisintestinal villousatrophy(eithertotalorpartial),whichmaybe asso-ciatedwith variable degrees of mucosal inflammation. In contrasttoceliacdisease,flattening of villi is notalways associatedwithincreasedintra-epitheliallymphocytesand
Figure5 Histopathologicalimageshowing almostcomplete recovery of duodenal villi three months after discontinuing olmesartan(hematoxylinandeosin,4×).
inflammation.6Inaddition,involvementofthestomachand
colon withlymphocytic aggregation was also reported by someauthors,4,8,15suggestingthatthisdisordermayaffect
theentiregastrointestinaltract.
Regarding treatment, previous reports have demon-strated clinical remission in all patients after discontinu-ationof olmesartan.4,6,8---13,15 Moreover,almostall patients
have shown histological recovery of the duodenum after drugwithdrawal(althoughfollow-upintestinalbiopsieshave notbeendonesystematicallyinallpatients).4,6,10,12In
con-trast to other enteropathies, such as celiac disease that may take years to achieve histological recovery, despite adequate treatment,16,17 olmesartan-induced enteropathy
isassociatedwithaquickmucosalrecovery(medianeight months from the suspension of the drug to follow-up biopsies).4
In ourcase, thecomplete resolution of symptoms and malabsorption,theintestinal villousatrophy improvement withoutanyothertherapeutic/dietarymeasuresbesides dis-continuationofthedrugandtheabsenceofothercausesof enteropathysupportthisdiagnosis.Deliberaterechallenge testwitholmesartanwasnotperformedbecauseofthe life-threatening nature of the disease and was not clinically pertinent.
Oneadditionalinterestingfinding, inourcase,wasthe hypertransaminasemia that normalized after the discon-tinuation of the drug. After excluding other causes for this finding, we believe that this could be linked to the enteropathy, similar to that described for celiac disease. Celiacpatientswithhypertransaminasemiahavean impor-tantincreaseinintestinalpermeabilitycomparedwiththose whose liverenzymesarenormal.18 Oneproposed
explana-tion for hepaticinvolvement in thesepatients is that the increased intestinal permeability may ease the entry of toxins, antigens, and inflammatory substances (cytokines and/orautoantibodies) totheportalcirculation that, sub-sequently,playaroleinliverinjury.19Inourcaseofsevere
olmesartan-inducedenteropathywespeculatethatsimilar mechanismscouldbeonthebasisofhypertransaminasemia. Thereportofmorecasesofthisdrug-inducedenteropathy iscrucialtoclarifythesefindings.
Thepathogenesisofthisentityisstillunclear.The long intervalbetweenthebeginningofolmesartantherapyand the onset of diarrhea suggests a cell-mediated immune
mechanism.4,11 Some reports have indicated a potential
inhibitoryeffectofangiotensinreceptorblockerson trans-forminggrowthfactor,whichisresponsibleforgutimmune homeostasis.20,21 Another proposed theory is related to
a pro-apoptotic effect of angiotensin (AT) II on intesti-nal epithelial cells. Angiotensin II binds to two receptor forms, AT1 and AT2, with differentproperties and differ-entgastrointestinaldistribution.AT1receptorisexpressed throughoutthewholealimentarytract,whileAT2receptoris expressedparticularlyintheduodenumandjejunum.22Sun
etal.suggestedthatangiotensinIIthroughbindingtoAT2 receptorup-regulates pro-apoptoticproteins, suchasBax andGATA-6,inassociationwithadown-regulationofBcl-2, ananti-apoptoticprotein.23 Olmesartanshowshighaffinity
for AT1 receptors. Incase ofsaturation of thisreceptors, circulatingangiotensinIIcouldbindtoAT2receptor result-ingin apro-apoptoticeffectand,consequently,tovillous atrophy.6Furtherinvestigationsareneededtoelucidatethe
physiopathologicalmechanismsunderlyingthisentity. Another issue to clarify is if other angiotensin II receptor blockers can have a similar adverse reaction. Recently, valsartan was implicated in a case of ‘‘sprue-like’’ enteropathy.24 Nevertheless, cases of enteropathy
relatedtootherangiotensinIIreceptorinhibitorsseemtobe muchlessfrequentthanolmesartan-inducedenteropathy.5
Theselectiveroleofolmesartanmightbeexplainedbyits conversionintotheactiveformintheintestine,itslonger half-lifeandhigherefficacycomparedtoothersartans.25
In conclusion, this report intends to alert the clinical communityforthisprobablyunderreportedproblem,11since
thiscondition is serious andolmesartan is commonly pre-scribedforthetreatmentofhypertension.Negativeserology forceliacdiseaseandabsenceofresponsetoagluten-free dietonpatientstakingolmesartanshouldawareclinicians for the possibility of this condition. We show iconogra-phynamelycapsuleendoscopicimages,thatdocumentthe endoscopic findings associated with this entity and can addinformationonthehallmarksofthisentity.Increasing reports of thisdrug-induced enteropathymay leadto the identification of more cases,a better characterizationof thisentityandanearlierdiagnosis,withobviousbenefitsto patients.
References
1.FasanoA,CatassiC.Clinicalpractice.Celiacdisease.NEnglJ Med.2012;367:2419---26.
2.DeGaetaniM,TennysonCA,LebwohlB,LewisSK,AbuDayaH, Arguelles-GrandeC,etal.Villousatrophyandnegativeceliac serology:adiagnosticandtherapeuticdilemma.AmJ Gastroen-terol.2013;108:647---53.
3.ReillyNR,FasanoA,GreenPHR.Presentationofceliacdisease. GastrointestEndoscClinNAm.2012;22:613---21.
4.Rubio-TapiaA, Herman ML, LudvigssonJF, Kelly DG, Mangan TF,WuT-T,etal.Severespruelikeenteropathyassociatedwith olmesartan.MayoClinProc.2012;87:732---8.
5.MartheyL,CadiotG,SeksikP,PouderouxP,LacrouteJ,SkinaziF, etal.Olmesartan-associatedenteropathy:resultsofanational survey.AlimentPharmacolTher.2014;40:1103---9.
6.LaniroG,BibbòS,MontaltoM,RicciR,GasbarriniA,Cammarota G.Systematicreview:sprue-likeenteropathyassociatedwith olmesartan.AlimentPharmacolTher.2014;40:16---23.
7.TheseventhreportoftheJointNational Committeeon Pre-vention, Detection,Evaluation, and TreatmentofHighBlood Pressure.Bethesda,MD:NationalInstitutesofHealth,National Heart,Lung,andBloodInstitute,USDepartmentofHealthand HumanServices;2004.NIHPublicationNo.04-5230.
8.FiorucciG,PuxedduE,ColellaR,ReboldiGP,VillanacciV, Bas-sottiG.Severespruelikeenteropathyduetoolmesartan.Rev EspEnfermDig.2014;106:142---4.
9.Dreifuss SE, Tomizawa Y, Farber NJ, Davison JM, Sohnen AE. Spruelike enteropathy associated with olmesartan: an unusualcaseofseverediarrhea.CaseRepGastrointestMed. 2013:618071.
10.Halevy D, Teeuwen U, Kohlhof P. Eine neue sprue-ähnliche erkrankungalsursacheschwererdiarrhoe.DtschMed Wochen-schr.2014;139:2290---3.
11.AbdelghanyM,IiiLG,SlaterJ,BegleyC.Casereport:olmesartan associatedsprue-likeenteropathyandcolonperforation.Case RepGastrointestMed.2014:3---6,2014.
12.HeerasingN,HairC,WallaceS.Olmesartan-induced enteropa-thy.InternMedJ.2015;45:117---8.
13.ThéophileH,DavidX-R,Miremont-SalaméG,HaramburuF.Five casesofsprue-likeenteropathyinpatientstreatedby olmesar-tan.DigLiverDis.2014;46:465---9.
14.BhatN,AnupamaNK,YelsangikarA,VizhiK.Olmesartan-related sprue-likeenteropathy.IndianJGastroenterol.2014;33:564---7.
15.Ould Sidi Mohamed M, Colardelle P. Entéropathie due à l’olmesartan. Ann Cardiol Angeiol. 2015, http://dx.doi.org/ 10.1016/j.ancard.2015.04.022(inpress).
16.Wahab PJ, Meijer JWR, Mulder CJJ. Histologic follow-up of people with celiac disease on a gluten-free diet: slow and incompleterecovery.AmJClinPathol.2002;118:459---63.
17.Rubio-TapiaA,RahimMW,SeeJA,LahrBD,WuT-T,MurrayJA. Mucosalrecovery and mortalityinadultswithceliacdisease aftertreatmentwitha gluten-freediet.AmJGastroenterol. 2010;105:1412---20.
18.NovacekG,MiehslerW,WrbaF,FerenciP,PennerE,VogelsangH. Prevalenceandclinicalimportanceofhypertransaminasaemia incoeliacdisease.EurJGastroenterolHepatol.1999;11:283---8.
19.ZaniniB,BaschèR,FerraresiA,PigozziMG,RicciC,Lanzarotto F,et al.Factors that contributeto hypertransaminasemiain patientswithceliacdiseaseorfunctionalgastrointestinal syn-dromes.ClinGastroenterolHepatol.2014;12,804.e2---810.e2.
20.Macdonald TT, Monteleone G. Immunity, inflammation, and allergyinthegut.Science.2005;307:1920---5.
21.Matt P,Schoenhoff F,HabashiJ,HolmT, VanErpC,LochD, et al. Circulating transforming growth factor-beta in Marfan syndrome.Circulation.2009;120:526---32.
22.Fändriks L. The angiotensin II type2 receptor and the gas-trointestinal tract. J Renin Angiotensin Aldosterone Syst. 2010;11:43---8.
23.SunL,WangW,XiaoW,LiangH,YangY,YangH.AngiotensinII inducesapoptosisinintestinalepithelialcellsthroughtheAT2 receptor,GATA-6 and theBaxpathway. BiochemBiophysRes Commun.2012;424:663---8.
24.HermanML,Rubio-TapiaA,WuT-T,MurrayJA.Acaseofsevere sprue-likeenteropathyassociatedwithvalsartan.ACGCaseRep J.2015;2:92---4.
25.Scialom S, Malamut G, Meresse B, Guegan N, Brousse N, Verkarre V, et al. Gastrointestinal disorder associated witholmesartanmimicsautoimmuneenteropathy.PLOSONE. 2015;10:e0125024.