J. Braz. Chem. Soc. vol.21 número10
Texto
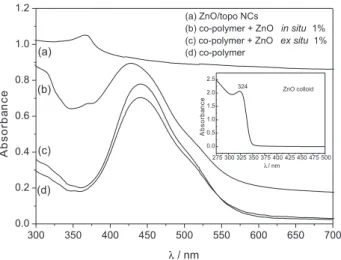
Imagem
![Figure 3. FTIR spectra of KBr pellets containing the co-polymer poly- poly-{trans-[RuCl 2 (vpy) 4 ]/sty} (a), the in situ prepared nanocomposite (b) and the ex situ prepared nanocomposite (c).](https://thumb-eu.123doks.com/thumbv2/123dok_br/18994193.461670/4.892.126.399.115.365/figure-spectra-pellets-containing-prepared-nanocomposite-prepared-nanocomposite.webp)
![Figure 5. SEM images of poly-{trans-[RuCl 2 (vpy) 4 ]/sty} (a), ZnO/](https://thumb-eu.123doks.com/thumbv2/123dok_br/18994193.461670/5.892.89.393.110.762/figure-sem-images-poly-trans-rucl-vpy-zno.webp)
![Figure S1. SEM images showing qualitative EDX mapping for ZnO/poly-{trans-[RuCl 2 (vpy) 4 ]/sty} nanocomposites prepared by ex situ (a) and in situ (b) methods.](https://thumb-eu.123doks.com/thumbv2/123dok_br/18994193.461670/7.892.102.827.537.813/figure-images-showing-qualitative-mapping-nanocomposites-prepared-methods.webp)
Documentos relacionados
Ao Dr Oliver Duenisch pelos contatos feitos e orientação de língua estrangeira Ao Dr Agenor Maccari pela ajuda na viabilização da área do experimento de campo Ao Dr Rudi Arno
Ousasse apontar algumas hipóteses para a solução desse problema público a partir do exposto dos autores usados como base para fundamentação teórica, da análise dos dados
didático e resolva as listas de exercícios (disponíveis no Classroom) referentes às obras de Carlos Drummond de Andrade, João Guimarães Rosa, Machado de Assis,
i) A condutividade da matriz vítrea diminui com o aumento do tempo de tratamento térmico (Fig.. 241 pequena quantidade de cristais existentes na amostra já provoca um efeito
The probability of attending school four our group of interest in this region increased by 6.5 percentage points after the expansion of the Bolsa Família program in 2007 and
não existe emissão esp Dntânea. De acordo com essa teoria, átomos excita- dos no vácuo não irradiam. Isso nos leva à idéia de que emissão espontânea está ligada à
Managers involved residents in the process of creating the new image of the city of Porto: It is clear that the participation of a resident designer in Porto gave a
The fourth generation of sinkholes is connected with the older Đulin ponor-Medvedica cave system and collects the water which appears deeper in the cave as permanent