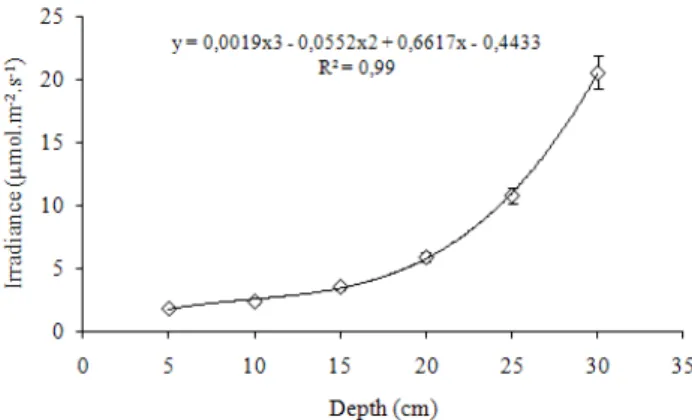
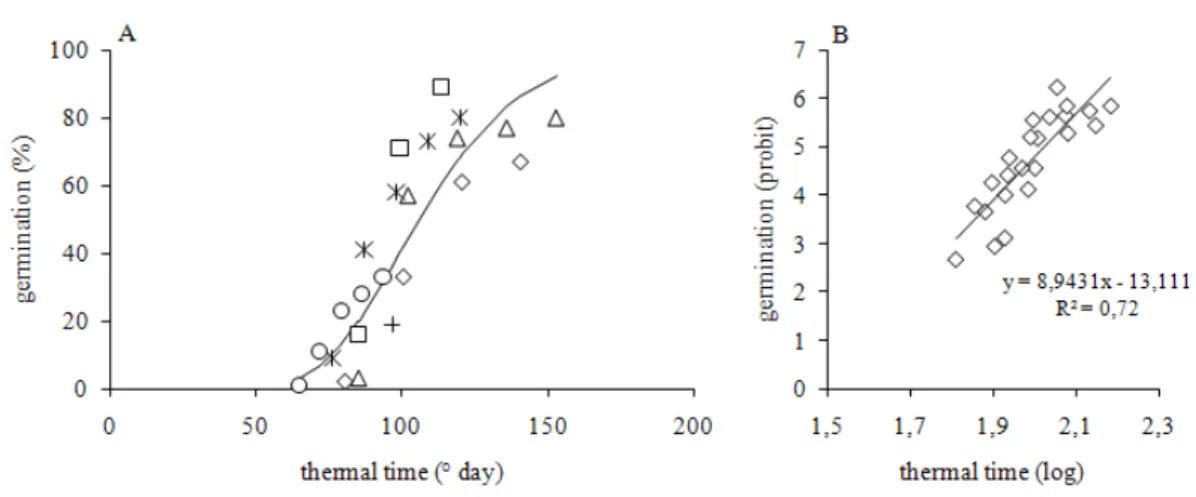
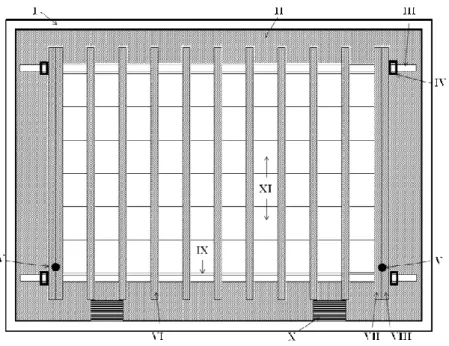
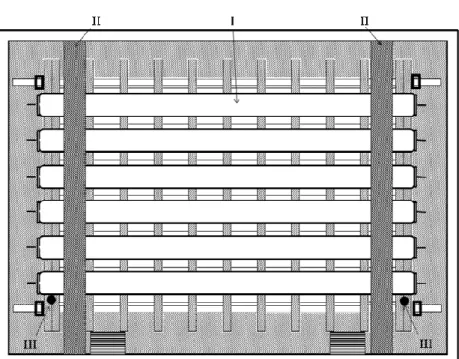
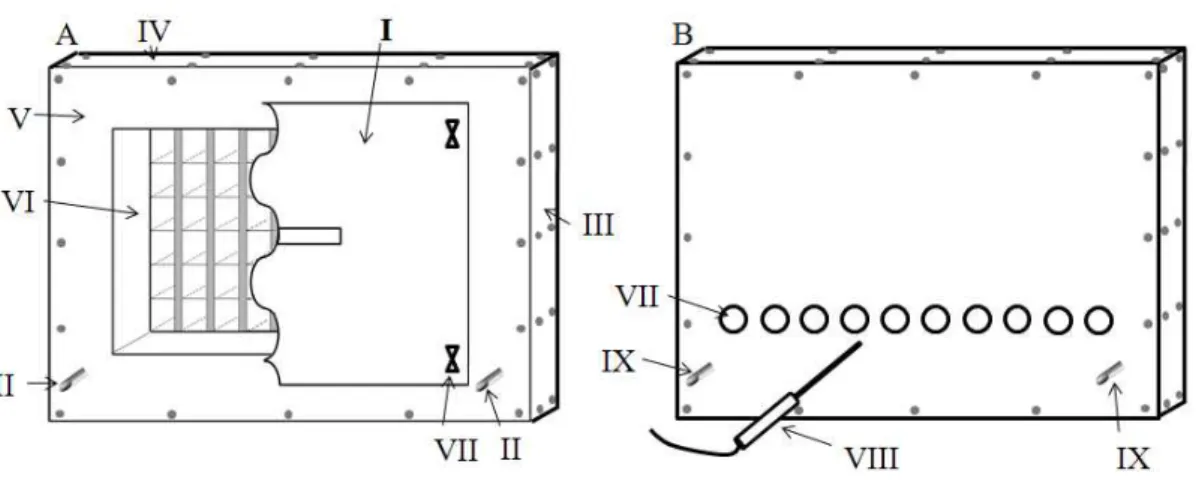
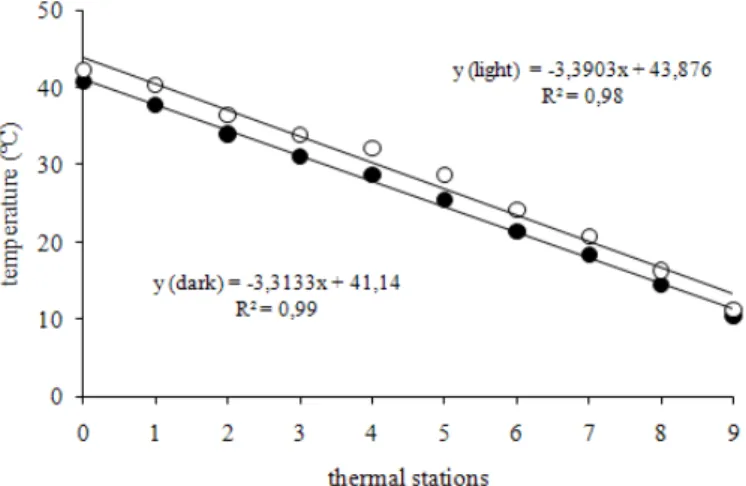
An Adapted Thermal-gradient Block for the Germination of Photoblastic Seeds
Texto
Imagem
Documentos relacionados
In the Porto series, we observed that, independently of the tumour type, 2 tumours co-expressing GLUT1, MCT4 and CD147 presented with synchronous metastasis and 2 other
Assim, como a proposta desta pesquisa, que envolveu identificar que fatores, segundo o modelo de Horner e Swarbrooke (2001), impactam sobre a decisão de compra do produto turístico
Segundo Oliveira (2010), no texto de Lessing, são evidenciadas não apenas as semelhanças como as diferenças existentes entre as.. duas artes, assim como se esboça
Considerando a importância da Classificação do Grau de Risco Familiar para o trabalho da ESF entende-se que tal ferramenta auxilia nas ações de promoção da saúde e
Riddley - Notes on the botany of Fernando Noronha.. Desmidiaeeae "in" Symbolae
The presence of a HAZ with such high hardness for the case of laser depositions can be explained by the presence of a thermal gradient from the coating/substrate interface to
In conclusion, the original version of the MGC scale, having been translated, culturally adapted, and validated into Brazilian Portuguese has proven to be a reliable instrument
Bars represent percentage of patients with non-motor symptoms (from a total of 134), as measured by the Non-Motor Symptom Scale (NMSS), MoCA (Montreal Cognitive Assessment scale)