J. Braz. Chem. Soc. vol.14 número4
Texto
Imagem


Documentos relacionados
The probability of attending school four our group of interest in this region increased by 6.5 percentage points after the expansion of the Bolsa Família program in 2007 and
CF/PAni composites, carbon fiber, and PAni powder were also characterized by Raman spectroscopy as shown.. in
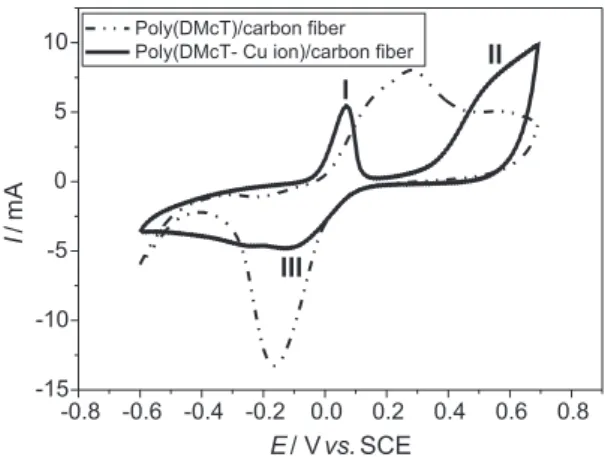
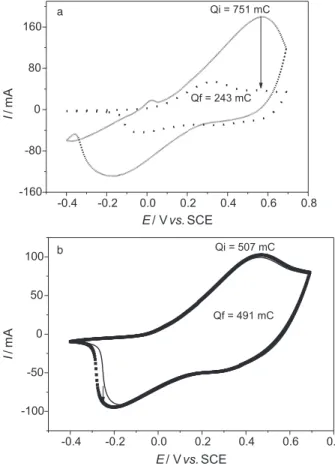
and electrochemical impedance techniques were used to understand the efect on the electrochemical properties of these ilms in relation to the presence or absence of holes on the
The electrochemical behavior of Cu-9%Al-5%Ni-2%Mn alloy in chloride media at different pHs was studied by means of open-circuit potential, cyclic voltammetry and
Comparison of the morphological, photochemical and electrochemical properties of these ilms indicated that the dansylglycine ilm electrodeposited on ITO exhibited electrochemical
In this paper, regarding the high capability of polymeric composites as a substitute of the wood in musical instruments, the acoustic properties of carbon fiber-polyester composites,
Electrochemical parameters obtained from Tafel plots for carbon steel AISI 1020, in 0.5 mol L -1 HCl in the presence and absence.. of different
Then, the LiFePO 4 /PAni composite was synthesized by chemical synthesis and its electrochemical properties as cathode material in lithium ion batteries were