ww w . r e u m a t o l o g i a . c o m . b r
REVISTA
BRASILEIRA
DE
REUMATOLOGIA
Original
article
Relapsing
polychondritis:
prevalence
of
cardiovascular
diseases
and
its
risk
factors,
and
general
disease
features
according
to
gender
Pablo
Arturo
Olivo
Pallo
a,
Maurício
Levy-Neto
a,
Rosa
Maria
Rodrigues
Pereira
b,
Samuel
Katsuyuki
Shinjo
b,∗aUniversidadedeSãoPaulo,FaculdadedeMedicina,HospitaldasClínicasHCFMUSP,SãoPaulo,SP,Brazil bUniversidadedeSãoPaulo,FaculdadedeMedicina,DisciplinadeReumatologia,SãoPaulo,SP,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received10September2016
Accepted11January2017
Availableonline15March2017
Keywords:
Autoimmunedisease
Cardiovasculardiseases
Gender
Relapsingpolychondritis
a
b
s
t
r
a
c
t
Thecomorbiditiesinrelapsingpolychondritishavebeenscarcelydescribedinthe
liter-ature.Moreover, apartfroma few relapsingpolychondritisepidemiological studies,no
studiesspecificallyaddressingrelapsingpolychondritisdistributionaccordingtogender
areavailable.Therefore,theobjectivesofthepresentstudywere:(a)toanalyzethe
preva-lenceofcardiovasculardiseasesanditsriskfactorsinaseriesofpatientswithrelapsing
polychondritis;(b)todeterminethe influenceof genderon relapsingpolychondritis.A
cross-sectionaltertiarysinglecenterstudyevaluating 30 relapsingpolychondritiscases
from1990 to 2016 wascarried out. To compare comorbidities,60 healthy individuals
matchedforage-,gender-,ethnicity-andbodymassindexwererecruited.Themeanage
ofrelapsingpolychondritispatientswas49.0±12.4years, themedian diseaseduration
6.0years,and70%werewomen. Ahigherfrequencyofarterialhypertension(53.3%vs.
23.3%;p=0.008)anddiabetesmellitus(16.7%vs.3.3%;p=0.039)wasfoundintherelapsing
polychondritisgroup,comparedtothecontrolgroup.Asanadditionalanalysis,patients
werecomparedaccordingtogenderdistribution(9menvs.21women).Theclinicaldisease
onsetfeatureswerecomparableinbothgenders.However,overthefollow-upperiod,male
patientshadagreaterprevalenceofhearingloss,vestibulardisorderanduveitisevents,
andalsoreceivedmorecyclophosphamidetherapy(p<0.05).Therewasahighprevalence
ofarterialhypertensionanddiabetesmellitus,andthemalepatientsseemedtohaveworse
prognosisthanthefemalepatientsinthefollowup.
©2017PublishedbyElsevierEditoraLtda.ThisisanopenaccessarticleundertheCC
BY-NC-NDlicense(http://creativecommons.org/licenses/by-nc-nd/4.0/).
∗ Correspondingauthor.
E-mail:[email protected](S.K.Shinjo).
http://dx.doi.org/10.1016/j.rbre.2017.02.003
2255-5021/© 2017 Published by Elsevier Editora Ltda. This is an open access article under the CC BY-NC-ND license (http://
Policondrite
recidivante:
prevalência
de
doenc¸as
cardiovasculares
e
seus
fatores
de
risco
e
características
gerais
da
doenc¸a
de
acordo
com
o
gênero
Palavras-chave:
Doenc¸aautoimune
Doenc¸ascardiovasculares
Gênero
Policondriterecidivante
r
e
s
u
m
o
Háescassezdeestudosnaliteraturasobreascomorbidadesnapolicondriterecidivante.
Alémdisso,excetoporalgunsestudosepidemiológicossobreapolicondriterecidivante,
nãoexistemtrabalhosqueanalisemespecificamenteadistribuic¸ãodapolicondrite
recidi-vantedeacordocomogênero.Portanto,osobjetivosdopresenteestudoforam:(a)analisar
aprevalênciadedoenc¸ascardiovasculareseseusfatoresderiscoemumasériedepacientes
compolicondriterecidivante;(B)determinarainfluênciadogêneronapolicondrite
recidi-vante.Fez-seumestudotransversalunicêntricoqueavaliou30casosdepolicondrite
recidi-vanteentre1990e2016.Paracompararascomorbidades,foramrecrutados60indivíduos
saudáveispareadosporidade,gênero,etniaeíndicedemassacorporal.Aidademédiados
pacientescompolicondriterecidivantefoide49,0± 12,4anos.Adurac¸ãomédiadadoenc¸a
foide6,0anose70%erammulheres.Foiobservadaumamaiorfrequênciadehipertensão
arterial(53,3%vs.23,3%,p=0,008)ediabetesmellitus(16,7%vs.3,3%;p=0,039)nogrupo
poli-condriterecidivanteemcomparac¸ãocomogrupocontrole.Emumaanáliseadicional,os
pacientesforamcomparadosdeacordocomadistribuic¸ãodegênero(novehomensversus
21mulheres).Ascaracterísticasclínicasiniciaisdadoenc¸aforamcomparáveisemambosos
sexos.Noentanto,duranteoperíododeseguimento,ospacientesdosexomasculinotiveram
maiorprevalênciadeperdaauditiva,envolvimentovestibulareeventosdeuveíteetambém
receberammaistratamentocomciclofosfamida(p<0,05).Houveumaaltaprevalênciade
hipertensãoarterialediabetesmellituseospacientesdosexomasculinoapresentarampior
prognósticodoqueaspacientesdosexofemininonoseguimento.
©2017PublicadoporElsevierEditoraLtda.Este ´eumartigoOpenAccesssobuma
licenc¸aCCBY-NC-ND(http://creativecommons.org/licenses/by-nc-nd/4.0/).
Introduction
Relapsing polychondritis (RP) is a rare systemic
autoim-mune disease characterized by recurrent inflammation of
cartilaginous structures (i.e.: ears, nasal bridge, peripheral
articulationsandtracheobronchialtree)and/ortissueswith
highproteoglycanconcentrations(i.e.:eyes,heart,kidneysand
bloodvessels).1–3Systemicmanifestationscanalsoinvolvethe
eyes,skin,joints,heartvalvesandbloodvessels.1,2
RPhasanannualincidencearoundof3.5casesper
mil-lion,andaffectsallethnicgroups,butapredominatewhite
population.4,5 Thefemale to male ratio is0.7–2.9:16–13 and
diseaseonsetoccurstypicallyinthefourthandfifthdecades.14
The few RP epidemiological studies conducted to date
showed that the most prevalent RP clinical symptoms are
auricularchondritis(65–98%ofcases)followedbyperipheral
arthritis(36–81%)andnasalchondritis(29–54%).6–13Mortality
inRPismorethantwiceofthegeneralpopulationandthe
mostfrequentcausesofdeatharerespiratorydisease,heart
conditionsandcancer.11
However, the comorbidities in RP have been scarcely
describedintheliterature.Notably,thereiscurrentlyonlyone
prospectivecohortstudy,reportingtheincidenceof
cardiovas-culardiseasesandtheirriskfactors(coronaryheartdisease,
strokeanddiabetesmellitus)inaseriesof117patientswith
RP.11 However,theauthors didnotspecificallydescribethe
prevalenceofthesecomorbidities.
Moreover,expectforafewRPepidemiologicalstudies,6–13
nostudiesspecificallyaddressingRPdistributionaccordingto
genderareavailable.Therefore,theobjectivesofthepresent
studywere:(a)toanalyzetheprevalenceofcardiovascular
dis-easesanditsriskfactorsinaseriesofpatientswithRP;(b)to
determinetheinfluenceofgenderonRP.
Materials
and
methods
Thepresentstudyisasinglecenterretrospectivecohortthat
included 30 consecutive patients with RP. To improve the
homogeneity of the sample under study, we include only
patientsfollowed upatourtertiary servicefromApril1990
toApril2016.
Allpatientsmetatleastthreeofthe6criteriaestablished
byMcAdametal.6 Patientswithage<18years,overlapping
syndrome,cancerorinfectionswereexcluded.
ThestudywasapprovedbythelocalEthicsCommittee.
Demographicsdata(ageatonsetofsymptomsand
diag-nosisofRP, gender),clinicalmanifestationsincludingfever,
fatigue, nasal involvement (saddle nose), auricular
chon-dritis, hearing loss, ocular problems (uveitis, episcleritis,
scleritis,keratitisorconjunctivitis),vestibulardisorder,
artic-ular (arthralgia or arthritis), neurological disorder (mainly
optic neuropathy, headache, seizures, hemiplegia, organic
brain syndrome, aseptic meningitis, meningoencephalitis
orcerebralaneurysms),costochondritis,subglotticstenosis,
laryngotracheitis,cardiacdisorder(mitraloraorticvalve
dis-eases), renal involvement (glomerulonephritis), body mass
index, weight, disease duration, and laboratory data were
obtainedfromasystematicreviewofthemedicalrecords.
The clinical and laboratory manifestations considered
(cumulativemanifestations).Data onbody massindexand
weightwereobtainedatthelastoutpatientvisit.
The patients were initially treated with glucocorticoid
(prednisone 0.5–1.0mg/kg/day) with subsequent tapering
of the dose according to clinical and laboratory
sta-bility. In the case of severe manifestations (i.e.: acute
hearing loss, uveitis, scleritis, vestibular disorder,
neuro-logicaldisorder,subglotticstenosis,laryngotracheitis),pulse
therapywithmethylprednisolone (1g/dayforthree
consec-utive days) was administered. For glucocorticoid tapering,
differentimmunosuppressiveswereused, aloneorin
com-binationtherapy,azathioprine(2–3mg/kg/day),methotrexate
(20–25mg/week), cyclosporine (2–4mg/kg/day),
mycophe-nolate mofetil (2–3g/day), leflunomide (20mg/day),
dap-sone(100mg/day), thalidomide(50–100mg/day),
cyclophos-phamide (0.5–1.0g/m2 of body surface area), intravenous
humanimmunoglobulin(1g/kg/day,duringtwoconsecutive
days)orbiological(tocilizumab8mg/kg,every4weeks;
abata-cept500–1000mgatweeks0,2and4,thenevery4weeks)and
non-steroidalanti-inflammatory.Drugsusedthroughoutthe
courseofthedisease,aswellasthoseprescribedinthelast
outpatientvisitofeachpatient,wereevaluated.
RP disease status was established under three groups,
based the last outpatient visit: (a) RP disease activity was
definedasthepresenceofanysymptomsand/orsigns
asso-ciatedwithRP,afterexclusionofinfectionsand/orneoplastic
causes,andofpatientsusingimmunosuppressivesand
glu-cocorticoid; (b) RP disease remission was defined as no
symptoms either signs associated to RP and without
glu-cocorticoid/immunosuppressives preceding six consecutive
months;(c)RPdiseasecontrolledwasdefinedaspatientswith
nosymptomsorsignsassociatedwithRP,butusing
immuno-suppressivesandtaperingglucocorticoid.
Surgicalintervention(tracheotomy,cochlearimplant)for
RPandmortalitywerealsoevaluated.
Thefollowingconditionswere evaluated: (a) the
cardio-vascular diseases (acute myocardial infarction, stroke and
congestiveheart failure),and(b)cardiovascularriskfactors
(arterialhypertension,type2diabetesmellitus,dyslipidemia,
smoking).
Dyslipidemia was defined as plasma total
choles-terol >200mg/dL, HDL-cholesterol <40mg/dL (male) or
<50mg/dL(female),LDL-cholesterol>130mg/dL,triglycerides
>150mg/dLordrugtreatmentforevaluatedLDL-cholesterol
ortriglycerides.14Arterialhypertensionwasestablishedwhen
thepatients werereceiving antihypertensive medicationor
when systolic pressure was ≥140mmHg and/or diastolic
pressurewas≥90mmHg.Diabetesmellituswasbasedonthe
resultsofplasmaglucosemeasurement.15
To assess the prevalence of comorbidities in patients
withRP,60consecutivehealthysubjects(controlgroup)were
included.ControlswererecruitedfromApril2013toApril2016,
andmatchedforage,sexandbodymassindex(BMI)(kg/m2)
atRPdiseaseonset.
Statisticalanalysis
The Kolmogorov–Smirnov test was used to evaluate the
distribution of each parameter. The data were expressed
as a mean±standard deviation or median (25th–75th
interquartile)forcontinuousvariablesorasfrequencies(%)
forcategoricalvariables.Themedian(25th–75thinterquartile)
was calculated for continuous variables not normally
dis-tributed.Comparisonsbetweenthepatientsandthecontrols
andbetweenthepatients(femalevs.male)weremadeusing
Student’st-testortheMann–Whitneytestforcontinuous
vari-ables.Pearson’schi-squaredtestorFisher’sexacttestwasused
toevaluatethecategoricalvariables.Themeasurementswere
expressedasanoddsratio(OR)with95%confidenceinterval
(CI).Valuesofp<0.05wereconsideredsignificant.Allofthe
analyseswereperformedwiththeSPSS15.0statisticssoftware
(Chicago,USA).
Results
Initiallyatotalof30consecutivepatientswithRPwere
com-pared with 60 healthy individuals (Table 1). As expected,
current age, gender, ethnicity and BMI were comparable
betweenbothgroups(p>0.05).However,theaverageBMIin
patients with RP was 28.0kg/m2 and, therefore, they were
overweight.ThemedianRPdiseasedurationwas6.0(2.8–15.0)
years.
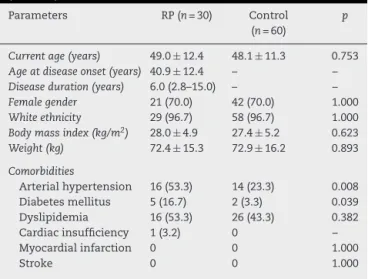
There was higher prevalence of arterial hypertension
(53.3%vs.23.3%;p=0.008,respectively)anddiabetesmellitus
(16.7%vs.3.3%;p=0.039)inpatientswithRP,whencompared
tocontrols.Thedyslipidemiaandsmokingdistributionwere
similarinbothgroups,whereasheartfailurewasfoundonlyin
patientswithRP.Therewerenocasesofmyocardialinfarction
orstrokeeventsineitherofthegroups.
On multivariate analysis, after adjusting for gender,
age and BMI, both arterial hypertension (OR 5.45; 95% CI
1.72–17.28)anddiabetesmellitus(OR6.67;95%CI1.12–39.89)
wereassociatedwithRP.
Table1–Generalfeaturesandcomorbiditiesofpatients withrelapsingpolychondritisandhealthyindividuals (control).
Parameters RP(n=30) Control (n=60)
p
Currentage(years) 49.0 ± 12.4 48.1 ± 11.3 0.753
Ageatdiseaseonset(years) 40.9 ± 12.4 – –
Diseaseduration(years) 6.0(2.8–15.0) – –
Femalegender 21(70.0) 42(70.0) 1.000
Whiteethnicity 29(96.7) 58(96.7) 1.000
Bodymassindex(kg/m2) 28.0 ± 4.9 27.4 ± 5.2 0.623
Weight(kg) 72.4 ± 15.3 72.9 ± 16.2 0.893
Comorbidities
Arterialhypertension 16(53.3) 14(23.3) 0.008 Diabetesmellitus 5(16.7) 2(3.3) 0.039 Dyslipidemia 16(53.3) 26(43.3) 0.382 Cardiacinsufficiency 1(3.2) 0 – Myocardialinfarction 0 0 1.000
Stroke 0 0 1.000
RP,relapsingpolychondritis.
Asanadditionalanalysis,patientswithRPwerecompared
according to genderdistribution (9 menvs. 21 women) as
showninTable2.
Currentpatient age,age atdiseaseonset,time between
diagnosis and symptom onset, and disease duration were
comparable between the gender groups. All initial clinical
manifestationswerealsosimilarintheRPandcontrolgroups.
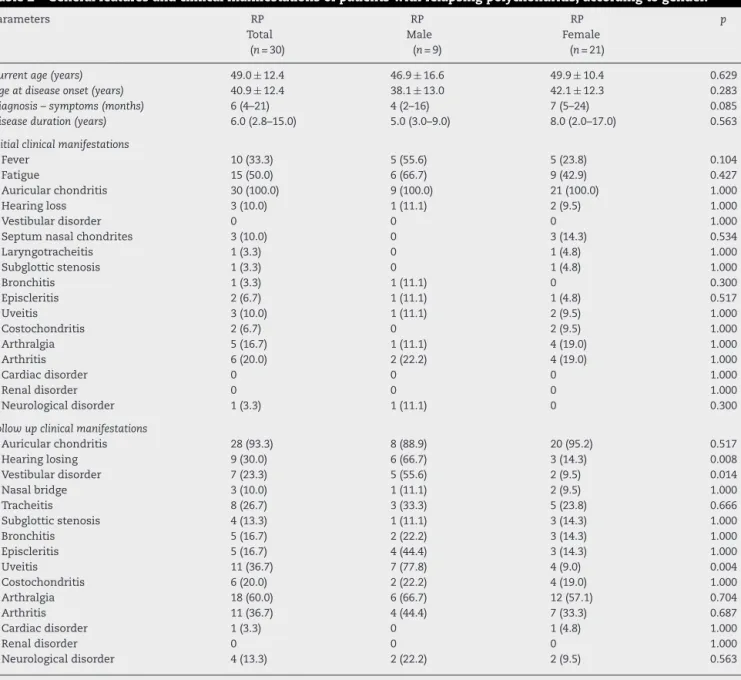
However,forfollowupclinical manifestations,therewas a
higherprevalence ofhearingloss, vestibular disorders and
uveitisinmales,comparedtofemales.
Onunivariateanalysis,afteradjustingforgender,ageand
BMI,thehearingloss(OR11.76;95%CI1.84–76.12),uveitis(OR
15.16;95%CI2.20–104.83)andvestibulardisorder(14.70;95%CI
1.59–136.14)weremorefrequentlyassociatedwithRPinmale
patientsthanfemalepatients.
Concerning cumulative treatment, there was a greater
tendencyformethylprednisolonepulsetherapyinmale,
com-paredtofemalepatientswithRP(55.6%vs.19.0%;p=0.082)
(Table 3). Male patients use significantly more
cyclophos-phamide than female patients (66.7% vs. 23.8%; p=0.042).
Use of other immunosuppressives was comparable in the
male and female groups (p>0.05). Moreover, there was no
statisticaldifferentincurrenttreatment(glucocorticoidand
immunosuppressives)betweenthetwogroups.
Surgicalcochlearimplantprocedureswereperformedin2
(22.2%)malepatientswithRP,whereastracheotomywas
car-riedoutin1(11.1%)maleand4(19.0%)femalepatientsduring
thefollowup(Table4).
Diseasestatus,remission,activityandcontrolwereequally
distributedbetweengenders.
Table2–Generalfeaturesandclinicalmanifestationsofpatientswithrelapsingpolychondritis,accordingtogender.
Parameters RP
Total (n=30)
RP Male
(n=9)
RP Female
(n=21)
p
Currentage(years) 49.0 ± 12.4 46.9± 16.6 49.9± 10.4 0.629
Ageatdiseaseonset(years) 40.9 ± 12.4 38.1± 13.0 42.1± 12.3 0.283
Diagnosis–symptoms(months) 6(4–21) 4(2–16) 7(5–24) 0.085
Diseaseduration(years) 6.0(2.8–15.0) 5.0(3.0–9.0) 8.0(2.0–17.0) 0.563
Initialclinicalmanifestations
Fever 10(33.3) 5(55.6) 5(23.8) 0.104
Fatigue 15(50.0) 6(66.7) 9(42.9) 0.427
Auricularchondritis 30(100.0) 9(100.0) 21(100.0) 1.000
Hearingloss 3(10.0) 1(11.1) 2(9.5) 1.000
Vestibulardisorder 0 0 0 1.000
Septumnasalchondrites 3(10.0) 0 3(14.3) 0.534
Laryngotracheitis 1(3.3) 0 1(4.8) 1.000
Subglotticstenosis 1(3.3) 0 1(4.8) 1.000
Bronchitis 1(3.3) 1(11.1) 0 0.300
Episcleritis 2(6.7) 1(11.1) 1(4.8) 0.517
Uveitis 3(10.0) 1(11.1) 2(9.5) 1.000
Costochondritis 2(6.7) 0 2(9.5) 1.000
Arthralgia 5(16.7) 1(11.1) 4(19.0) 1.000
Arthritis 6(20.0) 2(22.2) 4(19.0) 1.000
Cardiacdisorder 0 0 0 1.000
Renaldisorder 0 0 0 1.000
Neurologicaldisorder 1(3.3) 1(11.1) 0 0.300
Followupclinicalmanifestations
Auricularchondritis 28(93.3) 8(88.9) 20(95.2) 0.517
Hearinglosing 9(30.0) 6(66.7) 3(14.3) 0.008
Vestibulardisorder 7(23.3) 5(55.6) 2(9.5) 0.014
Nasalbridge 3(10.0) 1(11.1) 2(9.5) 1.000
Tracheitis 8(26.7) 3(33.3) 5(23.8) 0.666
Subglotticstenosis 4(13.3) 1(11.1) 3(14.3) 1.000
Bronchitis 5(16.7) 2(22.2) 3(14.3) 1.000
Episcleritis 5(16.7) 4(44.4) 3(14.3) 1.000
Uveitis 11(36.7) 7(77.8) 4(9.0) 0.004
Costochondritis 6(20.0) 2(22.2) 4(19.0) 1.000
Arthralgia 18(60.0) 6(66.7) 12(57.1) 0.704
Arthritis 11(36.7) 4(44.4) 7(33.3) 0.687
Cardiacdisorder 1(3.3) 0 1(4.8) 1.000
Renaldisorder 0 0 0 1.000
Neurologicaldisorder 4(13.3) 2(22.2) 2(9.5) 0.563
RP,relapsingpolychondritis.
Table3–Previous(cumulative)andcurrenttreatmentofpatientswithrelapsingpolychondritis,accordingtogender.
Parameters RP
Total (n=30)
RP Male
(n=9)
RP Female
(n=21)
p
Previoustreatment
PulsetherapywithMP 9(30.0) 5(55.6) 4(19.0) 0.082
Prednisone 24(80.0) 8(88.9) 16(76.2) 0.637
Cyclophosphamide 11(36.7) 6(66.7) 5(23.8) 0.042
Azathioprine 14(46.7) 6(66.7) 8(38.1) 0.236
Methotrexate 25(83.3) 7(77.8) 18(85.7) 0.622
Cyclosporine 3(10.0) 2(22.2) 1(4.8) 0.207
Mycophenolatemofetil 6(20.0) 3(33.3) 3(14.3) 0.329
Leflunomide 1(3.3) 1(11.1) 0 0.300
Dapsone 2(6.7) 0 2(9.5) 1.000
NHAI 18(60.0) 8(88.9) 10(47.6) 0.490
Talidomide 2(6.7) 1(11.1) 1(4.8) 0.517
IVIg 4(13.3) 2(22.2) 2(9.5) 0.563
Biological 5(16.7) 2(22.2) 3(14.3) 0.622
Currenttreatmentprednisone
Currentuse 8(26.7) 3(33.3) 5(23.8) 0.666
Dose(mg/day) 15.0(8.2–20.0) 15(15–20) 10(6–30) 0.571
Immunosuppressives
None 14(46.7) 3(33.3) 11(52.4) 0.440
One 15(50.0) 6(66.7) 9(42.9) 0.427
Two 1(3.2) 0 1(3.2) –
IVIg,intravenoushumanimmunoglobulin;MP,methylprednisolone;RP,relapsingpolychondritis. Resultsexpressedaspercentage(%),median(25th–75thinterquartilerange).
Therewasnostatisticaldifferenceincardiovascular dis-easeanditsriskfactorsdistributionaccordingtogender.The
mostcommonparametersinmenwerearterialhypertension
anddyslipidemia,followedbydiabetesmellitusand
myocar-dialinfarction,heart failure,and smoking.Amongwomen,
themostcommonparameterswerearterialhypertensionand
dyslipidemia,followingbydiabetesmellitusandmyocardial infarction,andsmoking.
Therewerenocasesofstrokeormortalityineitherofthe groups.
Discussion
Inthepresentstudy,ahighprevalenceofarterialhypertension anddiabetesmellituswereobservedinpatientswithRP. More-over,themalepatientsappearedtohaveaworseprognosis thanfemalepatientsinthefollowup.
AlthoughRP is a rare disease, we performed an
analy-sisinasampleof30 consecutive patientswithdefinedRP. Thepatients were recruited from asinglecenter, reducing inter-examinerfollow-upvariability.Inaddition,toevaluate theprevalenceofcardiovasculardiseaseanditsriskfactors, patientswere matchedwith60 healthyindividualsforage,
genderandbodymassindex.
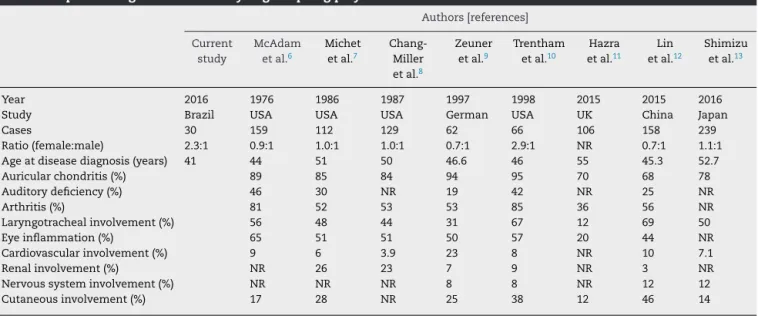
Themeanageofpatientsatdiseaseonsetwas49years,
comparabletopatientageinthemajorityofotherstudies,7–13
butcontrastingwiththestudiesofMcAdametal.6andHazra
etal.11thatreportedmeanagesof44and55years,respectively
(Table5).
AlthoughmoststudiesshowthatRPaffectbothgenders
similarly,6–9ourresultsfoundthatRPpredominantlyaffected
women. Ethnicity distribution in RPremains controversial.
AlthoughRPaffectsallracialgroupsequally,someseries
stud-ieshavefoundpredominanceinthewhitepopulation.1Our
resultscorroboratedthisfinding.
Inthepresentstudy,nocasesofmyocardialinfarctionor
strokewerefound.However,therewasahighprevalenceof
arterialhypertensionanddiabetesmellitus.Inthese
condi-tions,thecausescanbemulti-factorial(i.e.:associatedwith
RP,smokingand/orchronicuseofdrugs,particularly
gluco-corticoid).Infact,previousstudieshaveshownthatpatients
with chronic inflammatory rheumatic conditions, such as
systemiclupuserythematosus,rheumatoidarthritis,
spondy-loarthritis (psoriatic arthritis and ankylosing spondylitis),
inflammatory myopathies and juvenile idiopathic arthritis,
areatincreasedriskofdevelopingprematurecardiovascular
disease.16–25
ThemostcommoninitialclinicalmanifestationofRPisthe
auricularcondritis,6–13coincidingwithourstudy,foundinall
patientswithoutdifferencebetweengenders.Ofnote,
auric-ular chondritiscouldbemistakenbyaninfectious etiology
ortrauma.Otherclinicalmanifestationsarehighlyvariable,
in other series ofcase and ourstudy found: nasal
condri-tis,arthralgiawith/withoutsynovitis,laryngotracheitis,ocular
inflammation.Andthelesscommonfindingswere:cardiac,
renalandneurologicalinvolvement.6–13
Inadditionalanalysis,clinicalcourseoftheRPandtheirs
comorbitieswereevaluated,accordingtogenderdistribution.
Inourobservation,malegenderhadworseprognosiswhen
comparedtofemalegender,withhighprevalenceofuveitis,
hearinglossand vestibular disorder(55.6%).Tocorroborate
Table4–Surgery,diseasestatusandcomorbiditiesinpatientswithrelapsingpolychondritis,accordingtogender.
Parameters RP
Total (n=30)
RP Male
(n=9)
RP Female
(n=21)
p
Surgery
Cochlearimplant 2(6.6) 2(22.2) 0 –
Tracheostomy 5(16.7) 1(11.1) 4(19.0) 1.000
Diseasestatus
Remission 14(46.7) 3(33.3) 11(52.4) 0.440
Activity 8(26.7) 3(33.3) 5(23.8) 0.666
Controlled 8(26.7) 3(33.3) 5(23.8) 0.666
Comorbidities
Arterialhypertension 16(53.3) 5(55.6) 11(52.4) 1.000
Dyslipidemia 16(53.3) 5(55.6) 11(52.4) 1.000
Diabetesmellitus 5(16.7) 2(22.2) 3(14.3) 0.622
Heartfailure 1(3.2) 1(11.1) 0 1.000
Myocardialinfarction 0 2(22.2) 3(14.3) 0.622
Stroke 0 0 0 1.000
Mortality 0 0 0 1.000
RP,relapsingpolychondritis. Resultsexpressedaspercentage(%).
Table5–Epidemiologicalstudiesanalysingrelapsingpolychondritis.
Authors[references]
Current study
McAdam etal.6
Michet etal.7
Chang-Miller etal.8
Zeuner etal.9
Trentham etal.10
Hazra etal.11
Lin etal.12
Shimizu etal.13
Year 2016 1976 1986 1987 1997 1998 2015 2015 2016
Study Brazil USA USA USA German USA UK China Japan
Cases 30 159 112 129 62 66 106 158 239
Ratio(female:male) 2.3:1 0.9:1 1.0:1 1.0:1 0.7:1 2.9:1 NR 0.7:1 1.1:1
Ageatdiseasediagnosis(years) 41 44 51 50 46.6 46 55 45.3 52.7
Auricularchondritis(%) 89 85 84 94 95 70 68 78
Auditorydeficiency(%) 46 30 NR 19 42 NR 25 NR
Arthritis(%) 81 52 53 53 85 36 56 NR
Laryngotrachealinvolvement(%) 56 48 44 31 67 12 69 50
Eyeinflammation(%) 65 51 51 50 57 20 44 NR
Cardiovascularinvolvement(%) 9 6 3.9 23 8 NR 10 7.1
Renalinvolvement(%) NR 26 23 7 9 NR 3 NR
Nervoussysteminvolvement(%) NR NR NR 8 8 NR 12 12
Cutaneousinvolvement(%) 17 28 NR 25 38 12 46 14
methyprednisolone pulse therapy and also received more
cyclophosphamidepulses.
Conductivehearinglossdevelopsinupto46%ofpatients
and sensorineural hearing loss and vestibular dysfunction
mayoccur.Itcouldbesecondarytocartilagedestructionwith
closureoftheexternalauditorymeatus,serousotitismedia
oreustachiantubeobstruction,orserousotitismedia.4,26,27
Theetiologyofsensorineuralhearinglossandvestibular
dysfunctionmaybeduetoconductivehearingloss,to
vas-culitis ofthe branches ofthe internal auditory artery28 or
toautoantibodiesagainst labyrinthinebindingsites with a
local inflammatory response and subsequent apoptosis of
labyrinthinecells.29Thesensorineuralhearinglossrelatedto
vascularmechanismgenerallyispermanent,whereas
periph-eralvestibulardysfunctionaregenerallyreversible.26
OcularinflammationinRPmayaffectanypartoftheeye
andoccursbetween20and60%ofthecases.27–30Inourstudy
uveitisoccursinapproximately36.7%ofpatientswithRP.
In the present study, 16.7% of patients were submitted
totracheotomy (11.1%maleand 19.0%female). Respiratory
tractinvolvementisseeninupto38%ofpatientswithRPat
presentation,andinaboutonehalftotwothirdsofpatients
throughout the course of the disease.6,7,10 Airway
involve-mentisgenerallyconsideredominousandhasbeenreported
toportendapoorprognosis.7Tracheobronchomalacia,dueto
lossofthesupportivecartilaginousscaffoldingoftheupper
respiratoryairways,canbeseenasachronicsequelaeofRP
duetorecurrentinflammation.31,32 Respiratorycompromise
stemming from fixed airway obstruction or hyperdynamic
Inthepresentstudy,comorbiditydistributionanddisease
statuswerecomparablebetweenthegendergroups.Thereare
nodataonthedistributionofcomorbiditiesbetweengenders
butourstudyfoundnodifferencebetweenmalesandfemales.
Dataondiseasestatusisalsolimitedandinoursample46.7%
caseswereinremission,26.7%activityand26.7%controlled.
Itisalsodifficulttodeterminethefactorsassociatedwiththis
status,whichmaybeattributabletodiseaseseverity,
hetero-geneityofclinicalsymptoms,absenceoftreatmentprotocols
duetoalackofcontrolledclinicaltrials,adherenceto
treat-ment,orgeneticcomponents.
TheleadingcauseofmortalityinRPisairwayobstruction
secondary topneumonia, respiratory failure or progressive
cardiovascularinvolvement.34Inthepresentstudy,nodeaths
occurredduringpatientfollow-up.
Thisstudyhassomelimitations.Themajorlimitationsare
aretrospectivecohortstudydesign.Additionally,theinclusion
ofpatientssolelyfromatertiarycarecentermaynotrepresent
thefullRPspectrumandmighthaveresultedin
overestima-tionofdisease ordrugcomplicationsinthesemoresevere
cases.Finally,othercardiovascularriskfactorswerenot
ana-lyzed,suchastobacco,unhealthydiet,physicalinactivity,low
socioeconomicstatus.
In conclusions, there was a high prevalence of arterial
hypertensionanddiabetesmellitusinRP,andmalepatients
appearedtohaveaworseprognosisduringthefollowupthan
femalepatients.Furtherepidemiologicalstudiesareneeded
toconfirmourresults.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1. SharmaA,GnanapandithanK,SharmaK,SharmaS. Relapsingpolychondritis:areview.ClinRheumatol. 2013;32:1575–83.
2. GergelyPJr,PoorG.Relapsingpolychondritis.BestPractRes ClinRheumatol.2004;18:723–38.
3. AnanthakrishnaR,GoelR,PadhanP,MathewJ,DandaD. Relapsingpolycondritis–caseseriesfromSouthIndia.Clin Rheumatol.2009;28Suppl.1:S7–10.
4. KentPD,MichetCJJr,LuthraHS.Relapsingpolychondritis. CurrOpinRheumatol.2004;16:56–61.
5. PeeboBB,PeeboM,FrennesonC.Relapsingpolychondritis:a rarediseasewithvaryingsymptoms.ActaOphthalmolScand. 2004;82:472–5.
6. McAdamLP,O’HanlanMA,BluestoneR,PearsonCM. Relapsingpolychondritis:prospectivestudyof23patients andareviewoftheliterature.Medicine(Baltimore). 1976;55:193–215.
7. MichelCJJr,McKennaCH,LuthraHS,O’FallonWM.Relapsing polychondritis:survivalandpredictiveroleofearlydisease manifestations.AnnInternMed.1986;104:74–8.
8. Chang-MillerA,OkamuraM,TorresVE,MichetCJ,Wagoner RD,DonadioJVJr,etal.Renalinvolvementinrelapsing polychondritis.Medicine(Baltimore).1987;66:202–17.
9.ZeunerM,StraubRH,RauhG,AlbertED,ScholmerichJ,Lang B.Relapsingpolychondritis:clinicalandimmunogenetic analysisof62patients.JRheumatol.1997;24:96–101.
10.TrenthamDE,LeCH.Relapsingpolychondritis.AnnIntern Med.1998;129:114–22.
11.HazraN,DreganA,CharltonJ,GullifordMC,D’CruzDP. IncidenceandmortalityofrelapsingpolychondritisintheUK: apopulation-basedcohortstudy.Rheumatology.
2015;54:2181–7.
12.LinDF,YangWQ,ZhangPP,LvQ,JinO,GuJR.Clinicaland prognosticcharacteristicsof158casesofrelapsing polychondritisinChinaandreviewoftheliterature. RheumatolInt.2016;36:1003–9.
13.ShimuzuJ,OkaH,YamanoY,YudohK,SuzukiN.Cutaneos manifestationsofpatientswithrelapsingpolychondritis:an associationwithextracutaneouscomplications.Clin Rheumatol.2016;35:781–3.
14.DeBackerG,AmbrosioniE,Borch-JohnsenK,BrotonsC, CifkovaR,DallongevilleJ,etal.ThirdJointTaskForceof EuropeanandOtherSocietiesonCardiovascularDisease PreventioninClinicalPractice.Europeanguidelineson cardiovasculardiseasepreventioninclinicalpractice.Third JointTaskForceofEuropeanandOtherSocietieson CardiovascularDiseasePreventioninClinicalPractice.Eur HeartJ.2003;24:1601–10.
15.TheExpertCommitteeonthediagnosisandclassificationof diabetesmellitus.ReportoftheExpertCommitteeonthe diagnosisandclassificationofdiabetesmellitus.Diabetes Care.1997;20:1183–97.
16.MovahediM,BeauchampM,AbrahamowiczM,RayM, MichaudK,PedroS,etal.Riskofincidentdiabetesmellitus associatedwiththedosageanddurationoforal
glucocorticoidtherapyinpatientswithrheumatoidarthritis. ArthritisRheumatol.2016;68:1089–98.
17.DiDalmaziG,PagottoU,PasquiliR,VicennatiV.
Glucocorticoidsandtype2diabetes,fromphysiopatology.J NutrMetab.2012;2012:525093.
18.WalkerBR,EdwardsCR.Newmechanismsfor corticosteroid-inducedhypertension.BrMedBull. 1994;50:342–55.
19.deMoraesMT,deSouzaFH,deBarrosTB,ShinjoSK. Analysisofmetabolicsíndromeinadultdermatomyositis withafocusoncardiovasculardisesase.ArthritisCareRes. 2013;65:793–9.
20.deSouzaFH,ShinjoSK.Thehighprevalenceofmetabolic syndromeinpolymyositis.ClinExpRheumatol.2014;32: 82–7.
21.PereiraRM,deCarvalhoJF,BonfaE.Metabolicsyndromein rheumatologicaldiseases.AutoimmunRev.2009;8:415–9.
22.BruceIN.‘Notonly,butalso’:factorsthatcontributeto acceleratedatherosclerosisandprematurecoronaryheart diseaseinsystemiclupuserythematosus.Rheumatology (Oxford).2005;44:1492–502.
23.Avi ˜na-ZubietaJA,ChoiHK,SadatsafaviM,EtminanM,Esdaile JM,LacailleD.Riskofcardiovascularmortalityinpatients withrheumatoidarthritis:ametaanalysisofobservational studies.ArthritisRheum.2008;59:1690–7.
24.PetersMJ,vanderHorst-BruinsmaIE,DijkmansBA,
NurmohamedMT.Cardiovascularriskprofileofpatientswith spondylarthropathies,particularlyankylosingspondylitisand psoriaticarthritis.SeminArthritisRheum.2004;34:585–92.
25.CoulsonEJ,NgW-F,GoffI,FosterHE.Cardiovascularriskin juvenileidiopathicarthritis.Rheumatology(Oxford). 2013;52:1163–71.
27.IsaakBL,LiesegangTJ,MichetCJ.Ocularandsystemic findingsinrelapsingpolychondritis.Ophthalmology. 1986;93:681–9.
28.YuEN,JurkunasU,RubinPA,BaltatzisS,FosterCS. Obliterativemicroangiopathypresentingaschronic conjunctivitisinapatientwithrelapsingpolychondritis. Cornea.2006;25:621–2.
29.ChowMT,AndersonSF.Relapsingpolychondritis.OptomVis Sci.2000;77:286–92.
30.LetkoE,ZafirakisP,BaltatzisS,VoudouriA,Livir-RallatosC, FosterCS.Relapsingpolychondritis:aclinicalreview.Semin ArthritisRheum.2002;31:384–9.
31.BachorE,BlevinsNH,KarmodyC,KühnelT.Otologic manifestationsofrelapsingpolychondritis.Reviewof literatureandreportofninecases.AurisNasusLarynx. 2006;33:135–41.
32.SuzukiS,IkegamiA,HirotaY,IkusakaM.Feverandcough withoutpulmonaryabnormalitiesonCT:relapsing
polychondritisrestrictedtotheairways.Lancet.2015;385:88.
33.AdliffM,NgatoD,KeshavjeeS,BrenamanS,GrantonJT. Treatmentofdiffusetracheomalaciasecondarytorelapsing polychondritiswithcontinuouspositiveairwaypressure. Chest.1997;112:1701–4.