www.rbceonline.org.br
Revista
Brasileira
de
CIÊNCIAS
DO
ESPORTE
ORIGINAL
ARTICLE
Effects
of
supplementation
with
L-glutamine
and
L-alanine
in
the
body
composition
of
rats
submitted
to
resistance
exercise
Audrey
Yule
Coqueiro
∗,
Raquel
Raizel,
Thaís
Menezes
Hypólito,
Julio
Tirapegui
UniversidadedeSãoPaulo,FaculdadedeCiênciasFarmacêuticas,DepartamentodeAlimentoseNutric¸ãoExperimental, SãoPaulo,SP,Brazil
Received26January2016;accepted13June2017 Availableonline9August2017
KEYWORDS Glutamine; Alanine;
Resistanceexercise; Bodycomposition
Abstract We investigatedthe effects ofglutamine and alanine supplementation on body compositionofratssubmittedtoresistanceexercise.Wistarratsweresubmittedtoeight-week ofresistanceexercise,whichconsistedofclimbingaladderwithprogressiveloads(25---100%of bodyweight).Inthelast21daysoftraining,animalsweresupplementedwithL-glutamineand L-alanine,asadipeptideorintheirfreeform(DIP,GLN+ALAandALAgroups),orwater(SED andCTRLgroups).REattenuatedbodyweightgainandlipidcontentsofCTRLgroup(p<0.05 vs.SED)andDIPsupplementationpromotedanincreaseintibialismuscleweight,aswellasin proteincontent(p<0.05vs.CTRL).Takentogether,ourdataindicatedthatresistanceexercise improvesbodycompositionanddipeptidepotentiatedthemusclehypertrophiceffect. ©2017Col´egioBrasileirodeCiˆenciasdoEsporte.PublishedbyElsevierEditoraLtda.Thisisan openaccessarticleundertheCCBY-NC-NDlicense( http://creativecommons.org/licenses/by-nc-nd/4.0/).
PALAVRAS-CHAVE Glutamina;
Alanina;
Exercícioresistido; Composic¸ãocorporal
Efeitosdasuplementac¸ãocomL-glutaminaeL-alaninanacomposic¸ãocorporalderatos submetidosaoexercícioresistido
Resumo Foram investigados os efeitos da suplementac¸ão com glutamina e alanina na composic¸ão corporal de ratos submetidos a exercício resistido. Ratos Wistar foram sub-metidos, durante oito semanas, ao exercício resistido, que consistia em subiruma escada com cargas progressivas (25 a 100% do peso corporal). Nos últimos 21 dias de treina-mento, os animais foram suplementados com L-glutamina e L-alanina, como dipeptídeo ou em sua forma livre (DIP, GLN+ALA ALA e grupos) ou água (grupos SED e CTRL). Exercício resistido atenuou o ganho de peso corporal e conteúdo lipídico do CTRL (p < 0,05 vs. SED) e o DIP promoveu aumento no peso do músculo tibial, bem como no
∗Correspondingauthor.
E-mail:[email protected](A.Y.Coqueiro). http://dx.doi.org/10.1016/j.rbce.2017.06.003
teordeproteína(p<0,05vs.CTRL).Osnossosdadosindicamqueoexercícioresistido,melhora acomposic¸ãocorporaledipeptídeopotencializaoefeitohipertróficomuscular.
©2017Col´egioBrasileirodeCiˆenciasdoEsporte.PublicadoporElsevierEditoraLtda.Este ´e umartigoOpenAccesssobumalicenc¸aCCBY-NC-ND(http://creativecommons.org/licenses/ by-nc-nd/4.0/).
PALABRASCLAVE
Glutamina; Alanina; Ejerciciode resistencia;
Composicióncorporal
EfectosdelasuplementaciónconL-glutaminayL-alaninaenlacomposicióncorporal deratonessometidosaejercicioderesistencia
Resumen Seinvestigaronlosefectosdelaglutaminaylaalaninaenlacomposición corpo-ralderatonessometidosaejercicioderesistencia.AlgunosratonesWistarfueronsometidos a8semanasdeejercicioderesistencia,queconsistía ensubirunaescalera concargas pro-gresivas(del25al100%delamasacorporal).Enlosúltimos21días,losanimalesrecibieron unsuplementodeL-glutaminayL-alanina,enformadedipéptidooensuformalibre(grupos DIP,GLN+ALAyALA)oagua(gruposSEDyCTRL).Elejercicioderesistenciaredujoelaumento demasacorporalylaconcentracióndelípidosdelCTRL(p<0,05vs.SED).Lasuplementación conDIPpromovióunaumentodepesodelmúsculotibial,asícomoenelcontenidodeproteína (p<0,05frenteaCTRL).Nuestrosresultadosindicanqueelejercicioderesistenciamejorala composicióncorporalyelDIPpotenciaelefectohipertrófico.
©2017Col´egioBrasileirodeCiˆenciasdoEsporte.PublicadoporElsevierEditoraLtda.Estees unart´ıculoOpenAccessbajolalicenciaCCBY-NC-ND(http://creativecommons.org/licenses/ by-nc-nd/4.0/).
Introduction
Body composition is influenced by geneticfactors, physi-calactivity,nutrition,diseasesandothers.Changesinbody composition,suchasraisinginfatandreductionofmuscle tissue,areassociatedwiththedevelopmentofpathologies (Miller et al., 2013). Fat accumulation, characteristic in overweight andobesity, increases the risk of chronic dis-eases, such as diabetes, hypertension and cardiopathies. Similarly,decreased levelsofmusclemass interfere nega-tivelyintherestingmetabolicrate,reducephysicalcapacity andlifequality(Dalyetal.,2014).
Thesedentarylifestyleandhighcaloriefoodintake con-tributetotheincreaseinbodyfat,throughpositiveenergy balance(Ekelundetal.,2014)anddisturbancein homeosta-sisofhormonesinvolvedinappetiteandweightregulation (StieglerandCunliffe,2006).Musclemass gainis compro-mised in asedentary lifestyle due reducedstimulation of muscle protein synthesis (Shahar et al., 2013). Similarly, low-caloriedietspromotereductioninmusclemass, indicat-ingtheimportanceof balanceddietsfor weightreduction programs(Milleretal.,2013).Dietaryprogramsandphysical exercisearethemaininterventionsusedforpreventionand treatmentofmetabolicdisordersrelatedtobody composi-tiondisturbance(Kreideretal.,2010).
Studies demonstratethe effectivenessofaerobic exer-ciseinreducingbodyweightandadiposity(Sillanpaaetal., 2009;Aadland etal., 2014;Miller etal.,2014). However, thereareevidencesthatresistanceexercisepromotes sim-ilareffects(Arnarsonetal.,2014;Villanuevaetal.,2014; Youngetal.,2014),andthemainhypothesisisrelatedtoan increaseinmusclemass,increaseinrestingmetabolicrate andenergyexpenditure(StieglerandCunliffe,2006), reduc-tion in appetite and energy consumption (Rogero et al., 2005), aswell increasedlipid oxidation after an exercise session(DeFeo,2013).
Muscle hypertrophy, induced by resistance exercise, is promoted by myofibrilsreplication intothemuscle fibers. Thismechanismpromotesgrowthofthefibersandincrease in the production of muscle strength (Villanueva et al., 2014). The protein synthesis and muscle hypertrophy are enhanced by supplemental protein and essential amino acids.Thesenutrientsprovidesubstratesforanabolic reac-tions and repair after a physical exercise session.Hence, aminoacids supplementationisaverypopularalternative (Hartmanetal.,2007).
Glutamine,themostabundantaminoacidinthebody,is apotentiallyusefulsupplementforathletes(Cruzatetal., 2010).Besidesitsimportantroleforimmunefunction, glu-tamine has been related to hypertrophy. The hypothesis thatglutamineoptimizeproteinsynthesisandhypertrophy induced by exercise is based on the increase in cell vol-ume(Kreideretal.,2010),aswellasthroughoptimizingthe energystores,preventingproteolysis(Fontanaetal.,2003) andmodulationoftheimmunesystem,reducingtherelease of cytokines,which can impair muscle mass gain (Cruzat etal.,2010).However,theeffectivenessofthehypertrophic potentialofglutamineisnotwellelucidated(Kreideretal., 2010).
Oral or enteral supplementation with glutamine has reduced influence on glutaminemia and tissue glutamine, due to the high metabolism of this nutrient in the intes-tine. Inthis sense,administration withdipeptideL-alanyl L-glutaminehasbeenconsideredanalternativetoimprove glutamine absorption through to intestinal oligopeptide transporter (Pept-1), which is more efficient in transport dipeptidethanfreeaminoacids(Cruzatetal.,2014).
betweentheinterventionscouldimprovethebody composi-tion,reducinglipidcontent,increasingfat-freemassaswell asmuscleproteincontent.
Materials
and
methods
Animals
Forty adultmaleWistar ratswereprovidedby theanimal houseoftheUniversityofSãoPauloforuseinthisstudy. Ani-malswerehousedthreepercageinacontrolenvironment at 22±2◦Candrelativeair humidityof55±10%, undera 12-hlight/12-hdarkcycle(lightson4PM,lightsoff4AM)for aperiodofeightweeks.Animalsweredistributedintofive groups:sedentary(SED)andtrainedcontrol(CTRL);trained and supplemented withL-alanine (ALA), L-glutamineplus L-alanine in their free form (GLN+ALA), and the dipep-tide L-alanyl-L-glutamine (DIP). Rats had free access to waterandstandardchow(NUVILABCR1,NuvitalNutrients, Curitiba,Brazil). Foodintakeand bodyweightwere mea-suredthreetimesperweekandwaterintakewasregistered daily.AllprocedureswereapprovedbytheEthics Commit-teeonAnimalUseoftheUniversityofSãoPaulo(protocol: CEUA/FCF/428).
Resistanceexerciseprotocol
The exercise protocol has been adapted from Hornberger andFarrar(2004)andSchefferetal.(2012).Physicaltraining wasperformed duringeightweeksandconsistedof climb-ingaverticalladder(1.1m×0.18m,2cmgrid,80◦inclined) withweightaffixedtothebaseofrattail.Setsconsistedof eightladderclimbsandrest of 2min. Thisprocedure was repeatedonceat thefirsttwoweeks(adaptationperiod), withloadequalto5%ofbodyweight(BW).Afterthe adap-tationperiod,theexercisetrainingstartedwithloadof25% ofBW,withthreesets,duringtwoweeks.Subsequent exer-cisesessionswereincreasedtofoursets(50%ofBW)forone week,fivesets(75%ofBW)foroneandahalfweekand,in thelastsessions,animalsperformedsixsetscarrying100% of BW. Each day of exercise was considered one session, conductedevery48h(Fig.1).
Supplementation
Supplementswereadministeredinthelast21daysof exper-iment, diluted to 4% in drinking water and provided ad
libitum.Thesupplementintakewasdailyassessed.Amino acids amount wascalculated based in commercial dipep-tideconcentration(Dipeptiven® solutionconsistsof 20mg ofL-alanyl-L-glutaminedissolvedin100mLofwater,which equals8.2gofL-alanineand13.46gof L-glutamine).Free L-glutamine and free L-alanine were manufactured and supplied by Labsynth (Synth, São Paulo, SP, Brazil) and L-alanyl-L-glutaminewas manufactured by Fresenius Kabi S.A.(BadHomburg,HE,Germany).
Tissuemeasurements
Ratsweresacrificedbydecapitation1hafterthelastsession ofresistance exercise. Epididymal adiposetissue and tib-ialismuscleweresurgicallyexcisedaftersacrifice,weighted andaliquotswerestoredat−80◦Cforfurtheranalysis. Mus-clesamples(1g)werehomogenizedin2mLof lysisbuffer andproteinextractioncontaining20mMTris---HCl(pH7.4), 150mMNaCl,2% NonidetP-40, 1mM EDTA(pH 8.00),10% glycerol,20mMsodium fluoride, 30mM sodium pyrophos-phate,0.2% SDS,0.5% sodiumdeoxycholate andultrapure water.Was addedtothebuffer 1mMPMSF, manufactured bySigma---Aldrich.
Forhomogenizationwasusedelectrictypehomogenizer Polytron(IKAT10basic),keepingsampleschilledtoreduce theactivityofproteolyticenzymesandphosphatases.After this process, the samples were centrifuged for 15min at 14,000rpm at −4◦C temperature. The supernatant was dilutedtentimesindemineralizedwaterandhomogenized byvortexing.
Protein contentwasquantifiedin tibialis skeletal mus-cleusing BCA Protein Assaykit (Thermo Fisher Scientific, Massachusetts,USA, BCAProtein AssayKit). Briefly, 10L ofeach standard curve point and10L ofdiluted sample werepipettedintriplicateina96wellsplate,then200L ofworkingsolutionwasaddedintoeachwell.Theplatewas incubatedat37◦Cfor30minandabsorbancewasmeasured in450nmbyMicroplateReader340---750nmUV/vis(Biorad Benchmark--- California,EUA).
Bodyfat
Accordingtoanalyticalstandards ofthe AdolfoLuts Insti-tute (1985), after drying of carcasses, the lipid fraction was extracted with ethyl ether for 48h in extractor of soxhlet(InfratecMultiTE188).Afterextraction,the mate-rialremainedintheincubatorat105◦Cforanhourandthen
Physical training initiation
Supplementation initiation
Adaptation load 5%
1 sets
Load 25% 3 sets
Load 50% 4 sets
Load 75% 5 sets
Load 100% 5 sets
Sacrifice
1* 2* 3* 4* 5* 6* 7* 8*
Table1 Foodintake,bodyweightgain,bodyfatandpercentageofepididymaladiposetissueinratssubmittedtoresistance exerciseandsupplementationwithL-glutamineandL-alanine.
SED CTRL ALA GLN+ALA DIP
Foodintake(g/day) 62.08±4.39 54.13±1.63a 54.00±1.56 53.96±2.29 54.13±0.91 Bodyweightgain(%) 43.48±2.47 35.89±1.14a 35.91±1.88 35.94±1.70 36.98±0.85 Bodyfat(%) 24.83±2.28 14.53±2.41a 19.21±2.48 18.86±2.70 16.41±2.93 Epididymaladiposetissue(%) 1.34±0.19 0.94±0.20a 0.69±0.18 1.00±0.08 0.97±0.13
SED,sedentarygroupreceivedwater;CTRL,controlgroupreceivedwater;ALA,supplementedwithL-alanine;GLN+ALA,supplemented withL-alanine plusL-glutamine;DIP, supplementedwithL-alanyl-L-glutamine. Supplementsweredilutedindrinking waterina4% solutionandofferedadlibituminthelast21daysoftheexperiment.Eight-weekexerciseprotocolconsistedofclimbingaladderwith progressiveloadincrease.Theweightoftheepididymaladiposetissuewasdeterminedbythepercentageoftotalbodyweight.Data arepresentedasmean±SDM(n=8pergroup).
ap<0.05vs.SED(ttest).
wascooled in the desiccator prior tofinal weighing. The resultwasobtainedbycalculating:
Calculation: B
−A C
×100=%Lipids
Aistheweightoftheemptyballoon;Bistheweightof theballoonwithsample;CistheCarcassweight.
Statisticalanalysis
To test normality was used Kolmogorov---Smirnov test. To parametric data was used Student’s T-test for compari-son between SED and CTRL groups and One-way ANOVA, with post-test Tukey HSD (Honestly Significant Differ-ences), was used to compare CTRL and supplemented groups. Mann---Whitney test and One-way ANOVA followed byKruskal---Walliswereperformedtoanalyzenonparametric data.Differenceswithpvalues<0.05wereconsidered sta-tisticallysignificant. Analyseswereperformed usingGraph PadPrism5.0anddatawereexpressedasmean±standard deviationofthemean(SDM).
Results
Foodintakeandbodyweight
Food intake was statistically higher in sedentary rats (62.08±4.39g/day) compared to animals submitted to resistance training (54.13±1.63g/day). Resistance exer-cise promoted reduced body weight in CTRL group (35.89±1.14%) compared to sedentary (43.48±2.47%) (p<0.05vs.CTRL).ThedifferencesbetweenSEDandCTRL groups,concerningfoodintakeandbodyweightgain, con-sistof 15% and 21%,respectively. These data supportthe efficacyofprogressiveresistancetraininginreducingfood intakeandbodyweightgain.However,supplementsdidnot influencetheseparameters(Table1).
Bodyfat
AsshowedinTable1,resistanceexercisewasabletoreduce the lipid content of trained animals. Both body fat and
SED CTRL ALA GLN+ALA DIP
0.00 0.05 0.10 0.15 0.20 0.25
*
# #
% o
f BW
Figure2 Percentageoftibialismuscleweightrelativetothe totalfinalWistarratsweightsubmittedtoresistancetraining, exceptSEDgroup.Animalssupplementedfor21 dayswith4% solution,containingdipeptide(DIP),L-glutamineandL-alanine intheirsfreeforms(GLN+ALA)orL-alanine(ALA).SEDandCTRL receivedfilteredwater.Dataarepresentedasmean±SDM(n=8 pergroup).*p<0.05vs.SED(ttest);#p<0.05vs.CTRL(ANOVA, Tukey’sHSD).
weightofepididymaladiposetissuewerehigherin seden-taryanimals(24.83±2.28%and1.34±0.19%,respectively), comparedtotrainedgroup(14.53±2.41%and0.94±0.20%) (p<0.05vs.SED).Thesevaluesrepresentdifferenceof71% and 43% amongsedentary and trainedgroups, concerning bodyfat andweightofepididymaladipose tissue, respec-tively.Nodifferencewasfoundamonggroupssupplemented withaminoacids.
Musclemassgainandintramuscularprotein
SED CTRL ALA GLN+ALA DIP 0
10 20 30 40
*
#
ug
/u
L
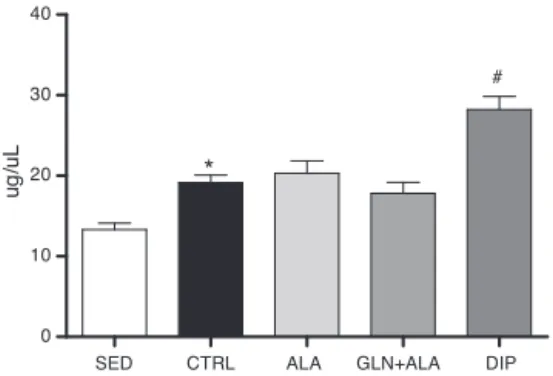
Figure3 Intramuscular protein intibialismuscleof Wistar ratssubmittedtoresistancetraining,exceptSEDgroup.Animals were supplemented for 21 days with 4% solution,containing dipeptide(DIP),L-glutamineandL-alanineintheirsfreeforms (GLN+ALA)orL-alanine(ALA).SEDandCTRLreceivedfiltered water. Data are presented as mean±SDM (n=8 per group). *p<0.05vs.SED(ttest);#p<0.05vs.CTRL,ALAandGLN+ALA (ANOVA,Tukey’sHSD).
Discussion
Progressiveresistanceexercisereducedfoodconsumption, body weight gain and lipid content in trained rats, con-firmingtheeffectivenesstochangethebodycomposition. Interestingly, L-glutamine administrationin the free form alongwithL-alanineorasdipeptideimprovedtheREeffects enhancingtibialismuscleweightintrainedanimals.Similar effectwasobservedinintramuscularproteincontent,which wasenhancedbyprogressiveresistanceexercise,however improvedonlybydipeptideadministration.
Intensephysicalexercisesareknowntoreduceappetite and food intake (Kreher and Schwartz, 2012) by increas-ingcytokinesreleaseandactivationofhypothalamicnuclei (Rogeroetal.,2005).Thedecreaseinenergyconsumption and increase in energy expenditure promoted by physi-cal exercise originate the negative energy balance state (Ekelundetal.,2014;StieglerandCunliffe,2006).Themain contributingfactoroftotalenergyexpenditureisthe exer-cise,correspondingto30---40%.Musclemassincreaseinduces high energy demands and contributes to the energy bal-anceandincreasedmetabolicrate,consequently reducing fatmass(StieglerandCunliffe,2006).
Thelipidcontentscanbemodifiedbytheresistance exer-cisethroughvariousmechanisms.Subsequenttoanexercise session,lipidoxidationisoptimizedassubstratetoprovide energy(DeFeo,2013).Theoptimizationofmetabolicrate, energyexpenditureandreducedfoodconsumptionarealso responsiblefor increaseduseof lipidsasenergysubstrate (Stiegler andCunliffe, 2006; Kreher andSchwartz, 2012). Furthermore,studiesindicatethatresistanceexercise pro-moteschangesin theprofileofplasmaticlipoproteinsand these findings were consistent with the reduction of adi-posetissueandimprovementofbodycomposition(Arnarson etal.,2014).
Resistance exercises can promote muscle hypertrophy, which is understood as increasingin the thicknessof the fibres, both due to the myofibrils accumulation, by the contractileproteins(actinandmyosin),asforaccumulation of non-contractilestructures,such aswaterand glycogen
(Buccietal.,2005).Themusclehypertrophycausedby exer-cise occurswhen there aresuccessive periods of positive muscleproteinbalance, which indicates proteinsynthesis higher than degradation (Hartman et al., 2007). Myofib-rilsreplicationwithinthemusclefibres,resultingfromthe hypertrophy,promotesgrowthoffibresandincreasein pro-ductionofmusclestrength(Villanuevaetal.,2014).
In thisstudy, therewasan increase of protein synthe-sis,evidenced byhigherlevelsofintramuscularproteinin animals subjected to progressive resistance exercise and supplementedwith dipeptide.This result,combined with theincrease oftheweightofmuscletissue,alsohigherin theexercisingandsupplementedgroup(GLN+ALAandDIP), correspondstotheaforementionedhypertrophyconcept.
Arnarson et al. (2014) subjected individuals to resis-tance exercise and found increased body weight, due to the increased muscle mass, as well decrease of approxi-mately1%ofbodyfat(Arnarsonetal.,2014).Theresultsof bodyweightdifferfromthepresentstudy,possiblydueto intensityoftheappliedexercise,sinceexhaustiveexercise reducesappetite,foodintakeand,therefore,bodyweight (Rogeroetal.,2005).
Aadland etal.(2014) subjected individualsto predom-inantly aerobic exercises. There was reduction of body weight and body fat of 10% and 16%, respectively. How-ever, the authorsalso observed reductionin muscle mass (Aadlandetal.,2014).Contrarytoaforementionedstudies, Youngetal. (2014) submittedrowers tosessionsof resis-tanceand aerobic exercise.There was reductionof body weightandbodyfat,aswellasanincreaseinmusclemass (Youngetal.,2014).
As a result of the benefits promoted by aerobic and anaerobicexercises, physical trainingcovering both types can provide better results on body composition parame-ters, regarding thereduction in body fat throughaerobic exercise, and potent elevation of the muscle mass, due toresistance exercise (Young et al.,2014). However,the exerciseprotocolappliedinthisstudywaseffectivein pro-motingimprovement in body composition, especially due toitsintensity,whichallowedbothbodyfatreductionand musclemassincrease.Musclehypertrophycausedby exer-ciseisenhancedbysupplementalproteinandaminoacids, sinceitprovidesthe substratesneeded for anabolic reac-tions (Hartman et al., 2007). In addition, protein intake contributestothepositiveproteinbalanceandinhibits pro-teindegradation(Villanuevaetal.,2014).
thecell andis an anabolic signal for hypertrophy (Cruzat etal.,2010).
Candow et al. (2001) subjected young adults to glu-taminesupplementationandresistanceexercise,however, differentfromtheresults presentedin thisstudy, supple-mentation didnot attenuate muscleprotein degradation, andtherewasnosignificantdifferenceinperformanceand bodycomposition(Candowetal.,2001).Thesefindingscan beexplainedby thesupplementationprotocol used,since thefree glutamine administrationhas reduced effects on glutaminemiaandtissueglutamine.
Supplementation with glutamine through dipeptide L-alanyl L-glutamine or a solution containing these amino acids in their free form, asapplied in the present study, havebeenprioritized,sinceitsabsorptionismoreefficient than absorptionof independentamino acids in free form (Cruzatetal.,2014).Previousstudiescorroboratethis find-ing(Cruzat andTirapegui,2009,2010; Petryetal.,2014). In this study, we observed a higher influence of supple-mentation withdipeptide L-alanyl L-glutamine onmuscle protein,comparedtothegroupssupplementedwitha solu-tion containing L-glutamine and L-alanine in their free forms.
Conclusion
We conclude that eight weeks of progressive resistance training resulted in improvements on body composition evidencedbyreductionofbodyweightandbodyfat,aswell asenhanced intramuscular proteincontent and increased musclemass.Supplementationwiththedipeptide L-alanyl-L-glutaminewasable tooptimizethe hypertrophic effect promotedbyexercise.
Funding
The present workwasfunded by SãoPaulo State Founda-tionforResearchSupport(FAPESP,Process:2012/21087-4). ThestudentshipswereprovidedbyBrazilianNational Coun-cilforScientificandTechnologicalDevelopment(CNPq)and theHigherEducationandTrainingCoordination(CAPES).
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
References
AadlandE,JepsenR,AndersenJR,AnderssenSA.Differencesinfat lossinresponsetophysicalactivityamongseverelyobesemen andwomen.JRehabilMed2014;46:363---9.
ArnarsonA, Ramel A, Geirsdottir OG,JonsoonPV, Thorsdottir I. Changesinbodycompositionanduseofbloodcholesterol low-eringdrugspredictchangesinbloodlipidsduring12weeksof resistanceexercise training inoldadults. AgingClin ExpRes 2014;26:287---92.
BucciM,VinagreEC,CamposGER,CuriR,Pithon-CuriTC.Efeitosdo treinamentoconcomitantehipertrofiaeendurancenomúsculo esquelético.RevCiêncMov2005;13:17---28.
CandowDG,ChilibeckPD,BurkeDG,DavisonKS,Smith-PalmerT. Effectofglutaminesupplementationcombinedwithresistance traininginyoungadults.EurJApplPhysiol2001;86:142---9. CruzatVF,TirapeguiJ.Effectsoforalsupplementationwith
glu-tamine and alanyl-glutamine on glutamine, glutamate, and glutathionestatusintrainedratsandsubjectedtolong-duration exercise.Nutrition2009;25:428---35.
CruzatVF,RogeroMM,TirapeguiJ.Effectsofsupplementationwith freeglutamineandthedipeptidealanyl-glutamineon parame-tersofmuscledamageand inflammationinratssubmittedto prolongedexercise.CellBiochemFunct2010;28:24---30. CruzatVF,KrauseM,NewsholmeP.Aminoacidsupplementationand
impactonimmunefunctioninthecontextoofexercise.JIntSoc SportsNutr2014;11:61.
DalyRM,MillerEG,DunstanDW,KerrDA,SolahV,MenziesD,etal. Theeffectsofprogressiveresistancetrainingcombinedwitha whey-proteindrinkandvitaminDsupplementationonglycaemic control,bodycompositionand cardiometabolicriskfactorsin olderadultswithtype2diabetes:studyprotocolfora random-izedcontrolledtrial.Trials2014;15:431.
De Feo P. Is high-intensity exercise better than moderate-intensityexercise for weight loss? NutrMetabCardiovasc Dis 2013;23:1037---42.
EkelundU,HildebrandM,CollingsPJ.Conferenceon‘Nutritionand healthyageing’The2013SummerMeetingSilverMedalWinner Physicalactivity,sedentarytimeandadiposityduringthefirst twodecadesoflife.ProcNutrSoc2014;73:319---29.
Fontana KE,ValdesH,Valdissera V.Glutamina comosuplemento ergogênico.RevBrasCiêncMov2003;11:91---6.
HartmanJW,TangJE,WilkinsonSB,TarnopolskyMA,LawrenceRL, Fullerton AV, et al. Consumption of fat-free fluid milk after resistanceexercisepromotesgreaterleanmassaccretionthan doesconsumptionofsoyorcarbohydrateinyoung,novice,male weightlifters.AmJClinNutr2007;86:373---81.
HornbergerTAJr,FarrarRP.PhysiologicalhypertrophyoftheFHL musclefollowing8weeksofprogressiveresistanceexercisein therat.CanJApplPhysiol2004;29:16---31.
Instituto AdolfoLuts.NormasanalíticasdoInstituto AdolfoLutz. Métodosquímicos efísicospara análisesde alimentos.v.1,3a ed.,SãoPaulo;1985.
Kreher JB, Schwartz JB. Overtraining syndrome. Sports Health 2012;4:128---38.
KreiderRB,WilbornCD,TaylorL,CampbellB,AlmadaAL,Collins R,etal.ISSNexercise&sportnutritionreview:research& rec-ommendations.JIntSocSportsNutr2010;7:7.
MillerCT,FraserSF,LevingerI,StraznickyNE,DixonJB,Reynolds J,etal.Theeffectsofexercisetraininginadditiontoenergy restriction on functional capacities and body composition in obeseadultsduringweightloss:asystematicreview.PLoSOne 2013;8:e81692.
Miller MB,PearceyGEP,Cahill F,Mccarthy H,StrattonSBD, Nof-tallJC,etal.Theeffectofashort-termhigh-intensitycircuit trainingprogramonworkcapacity,bodycomposition,andblood profilesinsedentaryobesemen:apilotstudy.BiomedResInt 2014;2014:191797.
Petry ER, Cruzat VF, Heck TG, Leite JSM, Bittencourt PIH Jr, TirapeguiJ.Alanyl-glutamineandglutamineplusalanine supple-mentsimproveskeletalredoxstatusintrainedrats:involvement ofheatshockproteinpathways.LifeSci2014;64:130---6. Rogero MM,MendesRR,TirapeguiJ. Aspectosneuroendócrinose
nutricionaisematletascom overtraining.Arq BrasEndocrinol Metabol2005;49:359---68.
Scheffer DL, Silva LA, Tromm CB, Rosa GL, Silveira PCL, Souza CT, etal.Impactofdifferentresistancetrainingprotocolson muscularoxidativestressparameters.ApplPhysiolNutrMetab 2012;37:1239---46.
interventiononbodycomposition,functionalfitness,and oxida-tivestressamongelderlyMalayswithsarcopenia.ClinIntervent Aging2013;2013:1365---75.
Sillanpaa E, Laaksonen DE, Hakkinen A, Karavirta L, Jensen B, Kraemer WJ,et al. Bodycomposition,fitness,and metabolic healthduringstrengthandendurancetrainingandtheir com-binationinmiddle-agedandolderwomen.EurJApplPhysiol 2009;106:285---96.
StieglerP,CunliffeA.Theroleofdietandexerciseforthe mainte-nanceoffat-freemassandrestingmetabolicrateduringweight loss.SportsMed2006;36:239---62.
VillanuevaMG,HeJ,SchroederET.Periodizedresistancetraining with and without supplementation improve body composi-tion and performance in older men. Eur J Appl Physiol 2014;114:891---905.