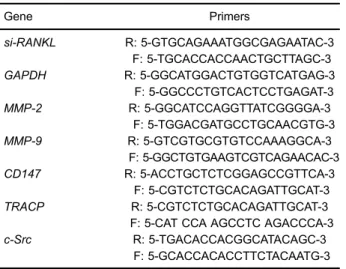
Up-regulated expression of CD147 gene in malignant bone tumor and the possible induction mechanism during osteoclast formation
Texto
Imagem
Documentos relacionados
Assim, ao constatar um crescente número de estudos com base nas duas teorias motivacionais, com algum destaque para o modelo do clima motivacional criado pelo treinador, acrescido
As tecnologias da informação (TI), por sua vez, têm dado um enorme contributo ao desenvolvimento e à diversidade de sistemas de suporte à decisão de crédito. Não obstante
Therefore, our study supports the hypothesis that volunteers with CP have RANKL/OPG imbalance during osseointegration, characterized by high levels of RANKL/OPG expression,
In this study, we investigated the effect of geraniin on OLC- induced bone resorption in vitro and the mRNA and protein expression of OPG and RANKL in rat osteoblasts.. MATERIAL
We plan to compare participants who are engaged in Housing First programs to participants engaged in traditional services to determine whether they differ on service and
In the glucocorti- coid-induced osteoporosis the RANK/RANKL/ /OPG role was described: glucocorticoids stimu- late RANKL expression by osteoclasts and inhibit OPG synthesis,
Receptor activator of nuclear factor kappa B ligand (RANKL) expres- sion is induced in osteoblasts, activated T-cells, synovial fibroblasts and bone marrow stromal cells, and
The RAW 264.7 cells were pretreated with 150 m g/mL titanium particles and different concentrations of BMP-2 for 1 h, then treated or not with 30 ng/mL RANKL for 7 days.. Data