w w w . j c o l . o r g . b r
Journal
of
Coloproctology
Original
Article
Extensive
colectomy
in
colorectal
cancer
and
hereditary
nonpolyposis
colorectal
cancer
–
long-term
results
Marisa
D.
Santos
a,b,c,∗,
Cláudio
Silva
a,
João
Oliveira
a,
Pedro
Brandão
a,b,c,
Mónica
Sampaio
a,b,c,
Ana
Cristina
Silva
a,b,c,
Anabela
Rocha
a,b,c,
Eduarda
Matos
d,
Ricardo
Marcos-Pinto
c,eaCentroHospitalarUniversitáriodoPorto(CHUP),Servic¸odeCirurgia,Porto,Portugal
bCentroHospitalarUniversitáriodoPorto(CHUP),UnidadedeCirurgiaColorretal,Porto,Portugal cUniversidadedoPorto,InstitutodeCiênciasBiomédicasAbelSalazar,Porto,Portugal
dUniversidadedoPorto,InstitutodeCiênciasBiomédicasAbelSalazar,DepartamentodeSaúdeComunitária,Porto,Portugal eCentroHospitalarUniversitáriodoPorto(CHUP),Servic¸odeGastroenterologia,Porto,Portugal
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received1December2018 Accepted21April2019 Availableonline24June2019
Keywords: Colorectalcancer HNPCC
Lynchsyndrome Instability
Mismatchrepairgenes Totalcolectomy
a
b
s
t
r
a
c
t
Background:Colorectalcancersurvivalisbetterinhereditarynonpolyposiscolorectalcancer patientsthaninsporadiccolorectalcancerpatientsandevenforhereditarynonpolyposis colorectalcancerwithcolorectalcancerisnotconsensualthatextensivecolectomyis prefer-abletopartialcolectomy.Thisstudyanalyzesandcomparesthelong-termresultsofthese twogroupsofpatientssubmittedtocurativesubtotalcolectomyortotalcolectomy. Methods:Between2002and2018,68patientswithcolorectalcancerwithoutfamilial ade-nomatouspolyposisweresubmittedtoatotalor subtotalcolectomyinasingletertiary center.Thepatientsweredividedintwogroups:hereditarynonpolyposiscolorectalcancer patients(withAmsterdamcriteria)andsporadiccolorectalcancerpatients(theothers).The presenceofAmsterdamcriteriaforhereditarynonpolyposiscolorectalcancerandgermline mutationformismatchrepairgeneswasconfirmedbyclinicalrecords.Resultsandsurvival wereanalyzedfollowingsurgery.
Results:Weobtainedasporadiccolorectalcancergroupwith31patientsandahereditary nonpolyposiscolorectalcancergroupwith37patients.Thetwogroupsdifferinagebutnot ingender,tumorstageorsurgicalmorbidity.Theoverallsurvivalanddisease-freesurvival weregoodinbothgroupsbutevenbetterforhereditarynonpolyposiscolorectalcancergroup withstatisticalsignificancewhencomparingthetwogroups.
Conclusion: Totalorsubtotalcolectomyforcolorectalcancerprovidesagoodsurvival.These surgicalproceduresshouldbeconsideredthefirstoptionforcolorectalcancerinyoung
∗ Correspondingauthor.
E-mail:marisadsantos@gmail.com(M.D.Santos). https://doi.org/10.1016/j.jcol.2019.04.005
2237-9363/©2019SociedadeBrasileiradeColoproctologia.PublishedbyElsevierEditoraLtda.ThisisanopenaccessarticleundertheCC BY-NC-NDlicense(http://creativecommons.org/licenses/by-nc-nd/4.0/).
hereditarynonpolyposiscolorectalcancerpatients.Inthosecases,theyprovidegood long-termresults,avoidingtheriskofmetachronouscolorectalcancerandthesurveillanceis restrictedonlytotheremainingneedforrectum.
©2019SociedadeBrasileiradeColoproctologia.PublishedbyElsevierEditoraLtda.Thisis anopenaccessarticleundertheCCBY-NC-NDlicense(http://creativecommons.org/ licenses/by-nc-nd/4.0/).
Colectomia
extensa
em
cancro
colorretal
e
cancro
colorretal
hereditário
sem
polipose
–
resultados
a
longo
prazo
Palavras-chave: Cancrocolorretal HNPCC
SíndromedeLynch Instabilidade
Genesdereparac¸ãodoADN Colectomiatotal
r
e
s
u
m
o
Introduc¸ão: Asobreviv ˆenciadocancrocolorretalémelhorempacientescomcancro col-orretalhereditárionãoassociadoapoliposedoqueempacientescomcancrocolorretal esporádico.Mesmoemcasosdecancrocolorretalhereditáriosempolipose,apreferência pelacolectomiatotalemrelac¸ãoàparcialnãoéconsensualnaliteratura.Esteestudo anal-isaecomparaosresultadosalongoprazodestesdoisgruposdepacientessubmetidosà colectomiacurativasubtotaloutotal.
Métodos: Entre2002e2018,68pacientescomcancrocolorretalsempoliposeadenomatosa familiarforamsubmetidosacolectomiatotalousubtotalemumúnicocentroterciário.Os pacientesforamdivididosemdoisgrupos:aquelescomcancrocolorretalhereditáriosem polipose(deacordocomoscritériosdeAmsterdão)eoscomcancrocolorretalesporádico (osdemais).OscritériosdeAmsterdãoparacancrocolorretalhereditáriosempoliposeea presenc¸ademutac¸ãogerminativaparaosgenesdereparac¸ãodeADNforamconfirmados porconsultadosregistrosclínicos.Osresultadoseasobrevivênciaforamanalisadosapós acirurgia.
Resultados: Nopresenteestudo,31pacientesforamincluídosnogrupodecancrocolorretal esporádicoe37nogrupodecancrocolorretalhereditáriosempolipose.Diferenc¸as signi-ficativasforamobservadasemrelac¸ãoàidade,masnãoaog ´enero,estadiodotumorou morbilidadecirúrgica.Asobreviv ˆenciaglobaleasobreviv ˆencialivrededoenc¸aforamboas emambososgrupos,masosresultadosforamaindamelhoresnogrupodecancrocolorretal hereditáriosempolipose,comsignificadoestat´istico.
Conclusão: Acolectomiatotalouacolectomiasubtotalparaocancrocolorretal propor-cionamumaboasobreviv ˆenciaedevemserconsideradasaprimeiraopc¸ãodetratamento em pacientesjovenscomcancrocolorretalhereditáriosem polipose.Nestespacientes, umacirurgiac ´olicamaisextensapermiteaobtenc¸ãodebonsresultadosalongoprazo; reduzoriscodecancrocolorretalmet ´acronoerestringeavigilânciaendosc ´opicaaoreto remanescente.
©2019SociedadeBrasileiradeColoproctologia.PublicadoporElsevierEditoraLtda.Este ´eumartigoOpenAccesssobumalicenc¸aCCBY-NC-ND(http://creativecommons.org/ licenses/by-nc-nd/4.0/).
Introduction
Hereditary nonpolyposis colorectal cancer (HNPCC, Lynch syndrome)isageneticdiseaseofautosomaldominant inheri-tanceduetogermlinemutationsinmismatchrepair(MMR) genesMLH1, MSH2, MSH6, PMS2or epigenetic silencingof MSH2 via inherited EPCAM mutations,1,2 which carries a
cumulative risk by age 70 years of 26%–80% for colorec-tal cancer (CRC).3 Mutation carriers also are at risk for several extra-colonic cancers, particularly cancers of the endometrium,ovary,urothelium,smallbowel,stomachand brain.4
Itsprevalenceinthegeneralpopulationisabout1in500, anditcausesabout2%–3%ofallcolorectalcancers.The muta-tioninoneoffourgenesoftheDNAmismatchrepairsystem
alsoconfersamarkedlyincreasedriskforothertypesof can-cer,namelyofendometrium.5,6
ThepresenceofHNPCCissuspectedwhenapatient devel-opscanceratanunusuallyyoungageorbecauseoffamilial clustering.UsuallypatientswithCCRwhomeetthe Amster-dam criteria(Table1)areHNPCCpatientsbydefinition.7 In
familiesthatmeettheAmsterdamcriteria,theprobabilityof havingamutationinMLH1,MSH2genesishigh(50%–92%).8,9
CurrentlytheAmsterdamcriteriaalsostillcoversfamilies withnoevidenceofaDNArepairdefectinatumor,inwhich the increasedtumorriskisprobablydueto geneticcauses that have not yetbeen identified.10 The familial natureof
coloncancerisalsocaused,toanunknownextent,by sim-plecoincidence.HNPCCpatientsalsoincludethosewhomeet theweakercriteriaoftheBethesdaGuidelines11(Table1)and
Table1–AmsterdamIICriteriaandRevisedBethesdaGuidelines.
AmsterdamIICriteria(Vasenetal.7) Allcriteriamustbemet:
Threeormorerelativeswithhistologicallyconfirmedcolorectalcancerorcanceroftheendometrium,smallbowel,ureter,orrenalpelvis, oneaffectedrelativebeingafirst-degreerelativeoftheothertwo;FAPshouldbeexcluded;
Twoormoresuccessivegenerationsareaffected;
Atleastonerelativewasdiagnosedbeforetheageof50years RevisedBethesdaGuidelines(Rodriguez-Bigasetal.11) Oneormoreofthefollowingcriteriamustbemet: Colorectalcancerbeforetheageof50years;
SynchronousormetachronouscolorectalcancerorotherHNPCC-relatedtumorsa,regardlessofage; ColorectalcancerwithMSI-highmorphologybbeforetheageof60years;
Colorectalcancer(regardlessofage)andafirst-degreerelativewithcolorectalcanceroranHNPCC-relatedtumorbeforetheageof50years; Colorectalcancer(regardlessofage)andtwoormorefirst-orsecond-degreerelativesdiagnosedwithcolorectalcanceroranHNPCC-related
tumor(regardlessofage)
FAP,familialadenomatouspolyposis;HNPCC,hereditarynonpolyposiscolorectalcancer;MSI,microsatelliteinstability.
a HNPCC-relatedtumorsincludecolorectal,endometrial,stomach,ovarian,pancreas,ureterandrenalpelvis,biliarytract,andbrain(usually
glioblastomaasseeninTurcotsyndrome)tumors,sebaceousglandadenomasandkeratoacanthomasinMuir–Torresyndrome,andcarcinoma ofthesmallbowel.
b Presenceoftumor-infiltratinglymphocytes,Crohn’s-likelymphocyticreaction,mucinous/signet-ringcelldifferentiation,ormedullarygrowth
pattern.
haveahighersensitivitybutlowerspecificitythanthe Amster-damcriteriaregardingevidenceofamutationinanMMRgene. Allpatientscarryingacancer-causinggermlinemutationin anMMRgene (almost halfofHNPCC patients)canalso be saidto haveLynchsyndrome. However,inclinical practice theterms“HNPCC”and“Lynchsyndrome”areusuallyused synonymously.12
UsuallyforLynchsyndromeconfirmation,thetumortissue isanalyzedforevidenceofdeficientmismatchrepair.Ifsuch evidenceisfound,ageneticmutationissought.The identifi-cationofapathogenicmutationbygenetictestconfirmsthe diagnosisinthepatientandenablespredictivetestingofother familymembers.13
Yet,theidentificationofMMRgenemutationisnotalways achievedandthediagnosis,thetherapeuticplanand surveil-lancearemadetakingintoaccounttheothercardinalfeatures besides the earlier average age of cancer onset, including CRCmainlyinrightcolon,acceleratedcarcinogenesis,high riskformetachronouscolorectalcancers,specificpathology featuresforHNPCCcolorectal cancers(theyare moreoften poorlydifferentiatedandhaveanexcessofmucoidandsignet cell features, a Crohn-like reaction and an excess of infil-tratinglymphocyteswithinthetumor)14andimportantly,an
increased survivalwhen compare with sporadic colorectal cancerpatients.
These aspects have clinical implications for HNPCC patientswithCRCmanagementnamely,extensionof surgi-calresection,theneedofchemotherapyinCRCStageIIIand frequencyofscreeningcolonoscopieswhenpartialcolectomy isdonetodetrimentofatotalorsubtotalcolectomy.15Infact,
theconceptthatmoreextensivecolonicresectionintheCRC andinparticularintheHNPCCdoesprovidesurvival advan-tageisnotprovenandthesurgicalpolicyvariesindifferent centers.Forthisspecificreasonwecarryoutthepresentstudy whichaimstoanalyzelong-termresultsaftertotalorsubtotal colectomyincolorectalcancer,especiallyconcerningpatients withAmsterdam criteria and based on resultsachieved to
determinate the most appropriate extension of surgical resectioninthisgroupofpatients.
Methods
Databasedesign,patientpopulationandinclusioncriteria Thestudywasanopen observationalstudy withouta con-trol group. All patients were 20 years of age or older at inclusion.Age atinclusion was thepatient age whenthey weresubmittedtoelectivetotal/subtotalcolectomyor colec-tomytotalization.Weincludedallpatientswithcoloncancer without familialadenomatouspolyposis (FAP)submittedto a curative total, subtotal colectomy or colectomy totaliza-tion between December 2002 and April 2018 in a single tertiaryreferralcenter.Otherdiagnosedneoplasia,presence of rectal cancer, clinical Stage IV and R1/R2 surgery were exclusioncriteriabutnottheclinicalpatienthistoryof pre-viouscolonoscopywithpolypectomyoraprevioussurgeryfor CRC.
Thepatientsweredividedintwogroups:HNPCCpatients (patients withAmsterdam criteria) and SCRC patients (the others).Allthepatientshaveacoloncancerdiagnosis with-outotherneoplasm.Allpatientsweresubjectedtofollow-up accordingtointernationalguidelines.Thepatientswere fol-loweduntilthelastupdateofinformation,andscoredasalive withoutdisease,alivewithdiseaseanddead.
Thefollowinginformationwasusedforthisreport: gen-der, presenceof Amsterdam criteria, geneticvariant when identified,ageatsurgery,ageandstageofpreviousCRCand surgery type and date if any, surgery extension and date, surgery morbimortality cancer stage, numbers of months betweensurgeryandlastobservationordead.When calculat-ingsurvival,eachpatientwasscoredonceonly,irrespective of how many surgeries for cancer the patient might have had.
Diagnosis
Total colonoscopy with a biopsy-proven adenocarcinoma, chest,abdominalandpelviccomputedtomographicscan,and serumcarcinoembryonicantigen(CEA)level.
Surgicalprocedures
Surgeryconsistedmainlyoftotalorsubtotalcolectomy.For patientswithpreviouspartialcolectomythesurgeryrealized wastotalizationofcolectomy.
Thetermof“subtotalcolectomy”isusedfortheauthors onlyfortheextensivecolectomyinwhichpartofthesigmoid andsigmoidarteriesweresparedinaneartotalcolectomy.
For authors the synonymous of“partial colectomy” are “segmentalcolectomy”.Thistermincludes“rightcolectomy”, “leftcolectomy”,“sigmoidectomy”or“transversecolectomy”. Regarding the selection of the operative procedure, we consideredthepresenceofCRCwithAmsterdamcriteria, syn-chronousCRC, metachronousCRC or CRC withpolyps not removablebycolonoscopy.
Resectedspecimensamples–H&Estaining;analysisfor deficientofMMR
Standard pathological tumorstaging ofthe resected spec-imen was performed in accordance with the guidelines of the American Joint Committee on Cancer (http://www. cancerstaging.net).
Tumor tissuewas analyzedfor evidenceofloss of mis-matchrepairproteinexpressionbyimmunohistochemistryor bymicrosatelliteinstabilitytest.Whenpositivethepathogenic mutationbygenetictest(denaturinghighperformanceliquid chromatographyanalysisandDNAsequencing)wasrealized. Adjuvantchemotherapyprotocol
Post-surgerypatientswithpathologicalStageIIIwere admin-istered adjuvant chemotherapy protocol for 6 months performed preferably with 5-FU or a combination of 5-FU and oxaliplatin(one ofthe followedregimens:mFolFOX6– 200mg/m2folinicacid(FA)day1,400mg/m25-FUbolusday1,
continuedinfusionfor46hof2400mg/m25-FUand85mg/m2
oxaliplatin,14/14daycycle;CapeOx:1000mg/m2capecitabine
twiceaday,days1–14,130mg/m2oxaliplatinday1,21/21day
cycle;5-FU/FA: 200mg/m2 FA day1, 400mg/m2 5-FU bolus
day1,continuedinfusionfor46hof2400mg/m25-FU,14/14
daymFolFOX6orCapeOXwerethepreferredregimens.When theadministrationofoxaliplatinisnotpossibleduetoside effectsofthedrugor thecomorbiditiesofthepatient, one offollowedregimenswasused:5-fluorouracil/folinicacid (5-FU/FA)200mg/m2folinicacid(1hinfusionpriorto5-FU)and
400mg/m2 5-FU per day intravenously once daily×5every
5weeksor1000mg/m2capecitabinetwiceaday×5every5 weeks.
Survivalanddiseaserecurrencedefinitions
Disease recurrence was evaluated according to location: locoregional (LR), systemic (DR) or mixed. All surviving
patients were followed-up and their current status was confirmed. None ofthe patients were lost from follow-up. Disease-free survival (DFS) was calculated from the date ofsurgerytothe dateofprogression(local ordistant), and overallsurvival(OS)wascalculatedfromthedateofsurgery tothedateofdeathorlastfollow-up.
Statisticalanalysis
TheHNPCCgroupandtheSCRCgroupwerecomparedin rela-tiontoage(Student’st-test)andinrelationtosex,stageand morbidity(Chi-squaretest).
Overallsurvivalanddisease-freesurvivalwereestimated inthe68patientsstudiedandalsointhetwogroupsusingthe Kaplan–Meiermethod.
Thedifferenceinsurvivalratesbetweenthetwogroups wastested forsignificanceusingthelog-ranktest.Survival wascomparedwiththelogranktest.
ThestatisticalanalysiswasmadewithSPSSstatistical soft-ware(version 21.0forWindows; SPSSInc., Chicago,IL).All statisticaltestswereconductedatatwo-sidedlevelof signif-icanceof0.05.
Ethicsapproval
ThisprojectwasapprovedbytheResearchEthicsHealth Com-mittee. Patientinformed consentfortheuseofclinicaland geneticinformationwasobtained.
Results
Descriptionofstudypopulationandclinicalparameters Thiscohortstudygathered72consecutivepatientswithCRC treatedwithtotal,subtotalcolectomyortotalizationof colec-tomy atonesingleUniversity Hospital.Afterthe exclusion of4patients withpStage IV, 68patients were includedin thepresentanalysis,withamedianageofyears58.7(range 30–86years).Themaletofemaleratiowas1.72:1.Patientswere dividedintwogroups–HNPCCgroup(patientswith Amster-damcriteria):37andSCRCgroup(theothers):31.Theclinical parametersaresummarizedinTable2.
Surgery
Total colectomy was performed in 42 (61.7%) patients; subtotal colectomy in 17 (25%) patients and totalization of colectomy in 9 (13.2) patients. In 26 (38.2%) patients the surgical approach was laparoscopic. The perioperative morbidity of the series was 23.5%, with 5 (7.3%) abdom-inal or pelvic abscesses, 5 (7.3%) anastomotic leaks, that included8(11.7)reoperations(duetofiveleakagesandthree abdominal abscesses), without re-admissions or mortality (Table3).
Table2–Clinicalparameters.
Entiresample(n=68) HNPCCgroup(n=37) SCRCgroup(n=31) p
Age 58.7 52.3 66.5 <0.001 Male 43 25 18 0.59 PreviousCRC 9 7 2 SynchronousCRC 20 12 6 CRC+polyps 43 21 22 CRClocation Rightcolon 31 18 13 Transversecolon 13 5 8 Leftcolon 19 11 8 Sigmoidcolon 14 6 8
Timebetweendiagnosisandsurgery(days) 42 41 43
Table3–Surgicalparameters.
Entiresamplen=68(%) HNPCCgroupn=37(%) SCRCgroupn=31(%) p Surgery Total/colectomy 42 23 19 Subtotal/colectomy 17 7 10 Totalization/of/colectomy 9 7 2 Morbidity Postoperativeileus 16(23.5%) 9(24.3%) 7(22.5%) Abdominal/infection 6 4 2 Abscess 3 1 2 Anastomoticleak 2 1 1 Reintervention 0.87
Withtemporarystoma 5 3 2
Withabdominal/washout 3 1 2
Readmission 0 0 0
Posteriorsurgeryforstomaclosure 5 3 2
Mortality 0 0 0
p=0.87(includesmorbidity,reintervention,readmissionandposteriorsurgeryforstomaclosure),notonlyreintervention.
Table4–Pathologicandgeneticparameters.
Entiresample(n=68) HNPCCgroup(n=37) SCRCgroup(n=31) p
Tumorgrade G1 3 0 3 G2 60 33 27 G3 5 4 1 Crohn-likereaction 10 8 2 Mucinoustumor 15 9 6
“Ringsignetcells”presence 3 3 0
Tumorstage 0.70 StageI 24 12 12 StageII 23 12 11 StageIII 22 13 8 MMRgenemutation MLH1 1 1 0 MSH2 8 8 0 MSH6 3 3 0 PMS2 0 0 0 EPCAMmutation 2 2 0
Pathologyofresectedspecimensandgenetictest
StagedistributionisshowninTable4.Theaveragenumber ofdissectedlymphnodesinthesurgicalspecimenswas23 (range18–55).AllresectedspecimenswereR0.
Geneticmutationwasidentifiedin14patientsof37with Amsterdamcriteria.
Clinicaloutcome
Table5showslong-termclinicaloutcome,relapseofdisease andsurvival.
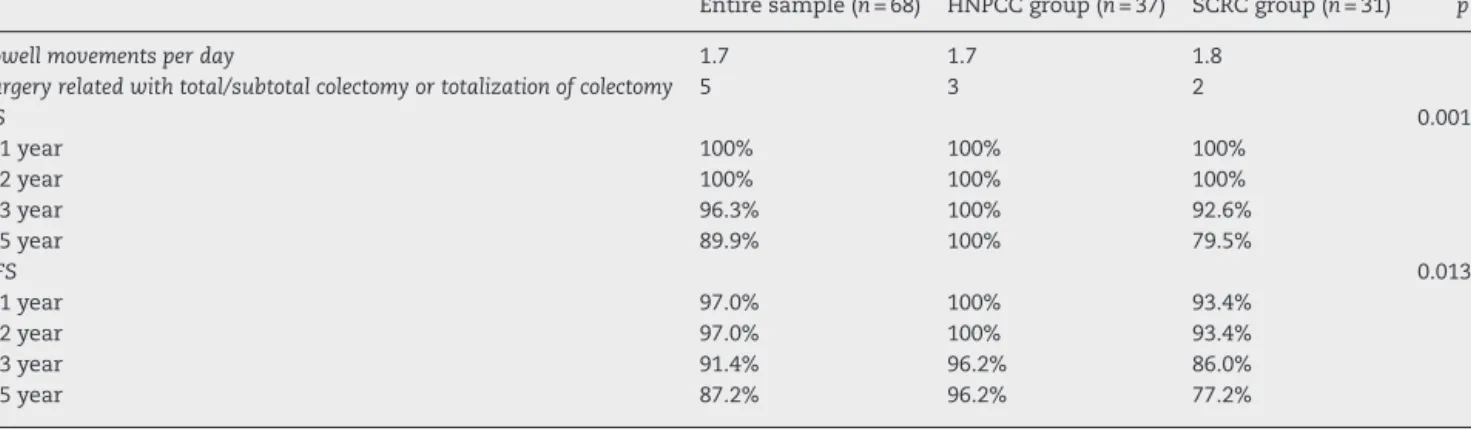
Themeanofpatientsfollow-upwas79.5months(range 6–190).
Table5–Long-termresultsafterextensivecolectomyinCRC.Meanoffollow-up:months79.5(range6–190).
Entiresample(n=68) HNPCCgroup(n=37) SCRCgroup(n=31) p Bowellmovementsperday 1.7 1.7 1.8
Surgeryrelatedwithtotal/subtotalcolectomyortotalizationofcolectomy 5 3 2
OS 0.001 1year 100% 100% 100% 2year 100% 100% 100% 3year 96.3% 100% 92.6% 5year 89.9% 100% 79.5% DFS 0.013 1year 97.0% 100% 93.4% 2year 97.0% 100% 93.4% 3year 91.4% 96.2% 86.0% 5year 87.2% 96.2% 77.2%
Concerning functional results: these patients have not
dietary restriction, regular medication for stoolregulation, nighttimedefecation,useofpaddaytimeoratnight,soiling orseepageduringdaytimeornightorurgencyofdefecation.
Also,theyhavenotanysocialhandicap. The24hstoolfrequencywas1.7.
Fiveofthe68patientshadothersurgicalproceduresrelated withthetotal/subtotalcolectomyortotalizationofcolectomy. Forexample,intestinalreconstruction(HNPCC-3andSCRC-2). Forglobalsample5yearOSwas89.9%and5yearDFSwas 87.2%.
WhencomparedOSandDFSforHNPCCandSCRCgroups,
survivalwassignificantlyhigherinHNPCCgroup(p=0.001and
p=0.013).
Fromtotalsample(68patients)onlyninepatients(13.2%) developeddistant metastases(3 hepatic metastases,3
pul-monarymetastases;1hepaticandpulmonarymetastasesand
1hepaticandretroperitoneallymphnodemetastases):7died and2werealivewithpulmonarymetastases.
Discussion
CCRisoneofthemostfrequentworldwidecancers.Inthecase ofnon-metastaticcoloncancer,segmentalcolectomy contin-uestobethetreatmentplancornerstonefollowedbyadjuvant chemotherapyfortumorStagesIII.16MostofCRCaresporadic
cancersbutcirca3%areHNPCCwithamorefavorablesurvival. So,inthosecasestherapy,surveillanceandgeneticcounseling arenecessarilydifferent.
ThefirstproblemthatarisesistheHNPCCidentification in population withCRC. Weknow that a large number of cliniciansare stillnotsensitized forscreeningand inmost centersthesystematicdetectionofmicrosatelliteinstability inpatientswithcoloncancerandageunder70yearsisnot yetimplemented.Intheotherhand,about15%ofsporadic cancershowsmicrosatelliteinstabilityandtherearepatients withAmsterdamcriteriainwhichmicrosatelliteinstabilityis negative.Forthatreason,inourcenterthepolicyadoptedfor patientswithAmsterdamcriteriaregardlesstheidentification ornotofthegeneticmutationthathavethe firstCRCwas amoreextensivecolectomyinsteadasegmentalcolectomy. Thisexplainswhy aconsiderable number ofpatients with Amsterdamcriteriaandmicrosatelliteinstabilitynegativetest weresubmittedtoamoreextensivecolectomyinoursample.
Thesecond issue isrelatedwith theconcept thatmore extensive colonicresectioninthe CRC andinparticularin the HNPCCdoes providesurvivaladvantage.Surgical man-agement ofcoloncancerforpatientswithLynchSyndrome whocarryamismatchrepairgenemutationiscontroversial. Actually,currentrecommendationsintheUSA,suggestthat personswithLynchsyndromeundergoingsurgicalresection of acolon cancer should beoffered toextensive resection rather than asegmentalresection, eventhoughthis policy hasnotpreviouslybeenproventobesuperiortoapolicyof1–2 yearlycolonoscopicsurveillance.17Despitethis
recommenda-tion,theextentofresectionperformedvariesbetweencenters in the USA. For example, the Cleveland Clinic performed total colectomyfor16of33 CRCpatientsfrom Amsterdam criteria-positive families compared with seven of 60 from clinics elsewhere in the USA.18 In Europe, on the basisof
adecisionanalysisstudy9 andthedocumentedhighriskof
ametachronous cancer,current guidelines recommendthe optionofextensiveresectionbediscussedwithpatients, par-ticularlythoseundertheageof50years.19Infact,thereare
nocontrolledstudiesavailablethataddressthequestionof whetherradicalsurgeryisappropriate.Anydecisionon radi-calsurgery,uptoandincludingcolectomy,wouldneedtotake into accountthe risk ofsurgery,the patient’s ageand sex, long-term medicalprognosis, and thequality and capacity todoannualcolonoscopysurveillancewithpreventive ade-nomaremoval.Infact,thedecisiontoremovemoreorlessof thecoloninvolvestheconsiderationofarelativelyhighrisk ofmetachronousColorectalCancer(CRC)withtheimpactof moreextensivesurgeryqualityoflife.20 Therelatively high
riskofmetachronousCRC,i.e.primaryCRCdiagnosedmore than12monthsafterthefirstdiagnosisofprimarycolon can-cer,(16%after10years)21supportsamoreaggressiveprimary
surgicalapproachinvolvingtheremovalofall,oratleastmost, ofthecolonafterdiagnosis.22Thefunctionalconsequenceof
anincreaseinbowelfrequencyandpossiblenegativeimpact onqualityoflifemightbebalancedagainstthereductionin riskofmetachronousCRCaffordedbymoreextensivesurgery, particularlyifthepersonisagedlessthan60yearsatthetime ofsurgery.23Ontheotherhand,surveillanceoftheremaining
colonandrectumwillberequiredaftermostsurgeryandthe inconvenience ofyearlycolonoscopy(withthe requirement forstandardbowelpreparationratherthanenema prepara-tion) may be offset bythe better functional outcome after segmentalsurgery.Thisclinicalequipoiseisreflectedinworld
surgicalopinion4,24andouropinionisinfavorofamore
exten-sivesurgery.
Basedon theseassumptionsweanalyzedfirst the long-termoverallresultsofoursampleandthenthesamesample dividedintotwogroups:patientsintwogroupswith Amster-dam criteria that we named HNPCC group and the other patientswithCRCbutwithoutcriteriaofAmsterdamwhere the surgery was more extensive due to the presence of metachronousneoplasmsorthepresenceofpolypsthatcould notbeendoscopicallyeasilyremoved.
Asthefirstanalysisoftheoverallsamplewecanconsider asgood the resultsofpatientstreated withamore exten-sivesurgery.Themorbidityofthesurgerywastheexpected, withoutdefinitivestomasandwithoutmortality.Themeanof movementsbowelnumberperdaywere1.7,withoutdietary restriction,needofmedicationforstoolregulationorsocial handicap; there were few subsequent abdominal surgeries relatedtothe total/subtotal colectomy;the overall survival anddisease-freesurvivalweregood(5year,OS:89.9%;5year, DFS:87.2%).So,inourseriesmorbidityandfunctionalquality ofamoreextensivesurgerywereacceptablewithouta nega-tiveburden.
In the other hand, the division ofthe sample into two groupsallowedustoobtaintwogroupswithdifferencesinage asexpectedbutwithoutsignificantdifferencesinsex,surgery, surgicalcomplicationsandstage,whichallowsustostatethat thesurvivalandsurvivalofthefirstgroupissignificantly bet-terthanthatofthesecondgroup.Thisisinaccordancewith thestatementthatCRCinHNPCCpatientshavebetter progno-sisandprobablyisnotfundamentaltocharacterizedpatient genemutationtoadoptamoreextensivesurgerysincepatient showsAmsterdamcriteria.
Finally, the third aspect the CRC treatment of HNPCC patient: patients with microsatellite-unstable tumors have abetterprognosisthan thosewithstabletumors(probably due to an immune response to tumor cells). This means thatthere maybeless benefitfrom adjuvant therapy. Sev-eral retrospective studies have shown that patients with microsatellite-unstableStageII andIII coloncancer donot benefitfrom adjuvant5-FU-basedchemotherapy.25Ongoing
researchisinvestigatingwhether thisisalsotrue forcolon cancersinHNPCCpatients.Thisaspectwasnotconsideredin ourseries.AllthepatientswithtumorStageIIIweretreated withadjuvantchemotherapywhichdoesnotinterferewith theresultspresented.
The strength of our study includes long mean follow-up, dataand surgeryrealized inasingle center.There are somelimitations inherenttothis study:the small number of patients and a small number of patients with genetic mutationconfirmation.Thisisatechnicaldifficultythatwe faceinthelastyearsinourcenter.Weintendinthefuture revisedallFormalin-FixedParaffin-Embeddedblocksoftumor resectedspecimens(FFPEtissuesample)fromthePathology DepartmentatourHospitalandrepeated themicrosatellite instabilityanalysisbyimmunohistochemistryandbyPCR.In meantimethemissingknowofgeneticmutationstatushas notpreventedthatitmaybeconsideredagoodpolicytoadopt amoreextensivecolicsurgerywheneverthepatientpresents Amsterdamcriteria.
Thevirtueofthisstudyiswarningtheneedforlarge-scale studies to confirmthe advantageof more extensive colon surgeryforpatientswithCRC,especiallyforthefirstCRCin patientswithAmsterdamcriteria.
Conclusion
Total or subtotal colectomy in CRC provides a good sur-vival.Thesesurgicalproceduresshouldbeconsideredthefirst optionforcolorectalcancerinyoungHNPCCpatients.Inthose cases,theyprovidegoodlong-termresults,avoidingtherisk ofmetachronousCRCandsurveillanceisrestrictedonlytothe remainingrectum.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1.LynchHT,LynchPM,LanspaSJ,SnyderCL,LynchJF,Boland CR.ReviewoftheLynchsyndrome:history,molecular genetics,screening,differentialdiagnosis,andmedicolegal ramifications.ClinGenet.2009;76:1–18.
2.TutlewskaK,LubinskiJ,KurzawskiG.Germlinedeletionsin theEPCAMgeneasacauseofLynchsyndrome–literature review.HeredCancerClinPract.2013;11:9.
3.BonadonaV,BonaïtiB,OlschwangS,GrandjouanS,HuiartL, LongyM,etal.Cancerrisksassociatedwithgermline mutationsinMLH1,MSH2,andMSH6genesinLynch syndrome.JAMA.2011;305:2304–10.
4.VasenHF,BlancoI,Aktan-CollanK,GopieJP,AlonsoA,Aretz S,etal.Revisedguidelinesfortheclinicalmanagementof Lynchsyndrome(HNPCC):recommendationsbyagroupof Europeanexperts.Gut.2013;62:812–23.
5.StoffelEM.Lynchsyndrome/hereditarynon-polyposis colorectalcancer(HNPCC).MinervaGastroenterolDietol. 2010;56:45–53.
6.D ˛ebniakT,GromowskiT,ScottRJ,GronwaldJ,HuzarskiT, ByrskiT,etal.Managementofovarianandendometrial cancersinwomenbelongingtoHNPCCcarrierfamilies: reviewoftheliteratureandresultsofcancerriskassessment inPolishHNPCCfamilies.HeredCancerClinPract.2015;13:3. 7.VasenHF,MecklinJP,KhanPM,LynchHT.TheInternational
CollaborativeGrouponHereditaryNon-PolyposisColorectal Cancer(ICG-HNPCC).DisColonRectum.1991;34:424–5. 8.CouraRdosS,Ashton-ProllaP,ProllaJC.Hereditary
non-polipomatouscolorectalcancer:hereditary
predisposition,diagnosisandprevention.ArqGastroenterol. 2005;42:99–106.
9.HendriksYM,deJongAE,MorreauH,TopsCM,VasenHF, WijnenJT,etal.Diagnosticapproachandmanagementof Lynchsyndrome(hereditarynonpolyposiscolorectal carcinoma):aguideforclinicians.CACancerJClin. 2006;56:213–25.
10.SteinkeV,EngelC,BüttnerR,SchackertHK,SchmiegelWH, ProppingP.Hereditarynonpolyposiscolorectalcancer (HNPCC)/Lynchsyndrome.DtschArzteblInt.2013;110:32–8. 11.Rodriguez-BigasMA,BolandCR,HamiltonSR,HensonDE,
JassJR,KhanPM,etal.Anationalcancerinstituteworkshop onhereditarynonpolyposiscolorectalcancersyndrome: meetinghighlightsandBethesdaguidelines.JNatlCancer Inst.1997;89:1758–62.
12.KravochuckSE,KaladyMF,BurkeCA,HealdB,ChurchJM. DefiningHNPCCandLynchsyndrome:what’sinaname?Gut. 2014;63:1525–6.
13.BonneauC,RouzierR,GeylC,CortezA,CastelaM,LisR,etal. Predictivemarkersofchemoresistanceinadvancedstages epithelialovariancarcinoma.GynecolOncol.2015;136:112–20. 14.SmyrkTC,WatsonP,KaulK,LynchHT.Tumor-infiltrating
lymphocytesareamarkerformicrosatelliteinstabilityin colorectalcarcinoma.Cancer.2001;91:2417–22.
15.delaChapelleA,HampelH.Clinicalrelevanceof microsatelliteinstabilityincolorectalcancer.JClinOncol. 2010;28:3380–7.
16.DosSantosLV,FariaTM,LimaAB,AbdallaKC,deMoraesED, CruzMR,etal.Timingofadjuvantchemotherapyin colorectalcancer.ColorectalDis.2016;18:871–6.
17.LindorNM,PetersenGM,HadleyDW,KinneyAY,MiesfeldtS, LuKH,etal.Recommendationsforthecareofindividuals withaninheritedpredispositiontoLynchsyndrome:a systematicreview.JAMA.2006;296:1507–17.
18.VanDalenR,ChurchJ,McGannonE,FayS,BurkeC,ClarkB. Patternsofsurgeryinpatientsbelongingto
Amsterdam-positivefamilies.DisColonRectum. 2003;46:617–20.
19.VasenHF,MösleinG,AlonsoA,BernsteinI,BertarioL,Blanco I,etal.GuidelinesfortheclinicalmanagementofLynch syndrome(hereditarynon-polyposiscancer).JMedGenet. 2007;44:353–62.
20.MaedaT,CannomRR,BeartRWJr,EtzioniDA.Decisionmodel ofsegmentalcomparedwithtotalabdominalcolectomyfor coloncancerinhereditarynonpolyposiscolorectalcancer.J ClinOncol.2010;28:1175–80.
21.deVostotNederveenCappelWH,NagengastFM,GriffioenG, MenkoFH,TaalBG,KleibeukerJH,etal.Surveillancefor hereditarynonpolyposiscolorectalcancer:along-termstudy on114families.DisColonRectum.2002;45:1588–94. 22.ParryS,WinAK,ParryB,MacraeFA,GurrinLC,ChurchJM,
etal.Metachronouscolorectalcancerriskformismatch repairgenemutationcarriers:theadvantageofmore extensivecolonsurgery.Gut.2011;60:950–7.
23.deVostotNederveenCappelWH,BuskensE,vanDuijvendijk P,CatsA,MenkoFH,GriffioenG,etal.Decisionanalysisinthe surgicaltreatmentofcolorectalcancerduetoamismatch repairgenedefect.Gut.2003;52:1752–5.
24.BernsteinIT,Lindorff-LarsenK,TimshelS,BrandtCA, DinesenB,FengerM,etal.Biomedicalinformaticsassupport toindividualhealthcareinhereditarycoloncancer:the DanishHNPCCsystem.HumMutat.2011;32:551–6. 25.RibicCM,SargentDJ,MooreMJ,ThibodeauSN,FrenchAJ,
GoldbergRM,etal.Tumormicrosatellite-instabilitystatusas apredictorofbenefitfromfluorouracil-basedadjuvant chemotherapyforcoloncancer.NEnglJMed.2003;349: 247–57.