w w w . r b h h . o r g
Revista
Brasileira
de
Hematologia
e
Hemoterapia
Brazilian
Journal
of
Hematology
and
Hemotherapy
Original
article
A
new
index
to
discriminate
between
iron
deficiency
anemia
and
thalassemia
trait
Januária
F.
Matos
a,b,∗,
Luci
M.S.
Dusse
b,
Karina
B.G.
Borges
b,
Ricardo
L.V.
de
Castro
c,
Wendel
Coura-Vital
d,
Maria
das
G.
Carvalho
baInstitutoFederaldeMinasGerais(IFMG),OuroPreto,MG,Brazil
bUniversidadeFederaldeMinasGerais(UFMG),BeloHorizonte,MG,Brazil
cHospitalOdilonBehrens,BeloHorizonte,MG,Brazil
dUniversidadeFederaldeOuroPreto(UFOP),OuroPreto,MG,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received12March2016 Accepted19May2016 Availableonline22June2016
Keywords:
Newindex
Irondeficiencyanemia Thalassemiatrait
a
b
s
t
r
a
c
t
Background:Themostcommonmicrocyticandhypochromicanemiasareirondeficiency
anemiaandthalassemiatrait.Severalindicestodiscriminateirondeficiencyanemiafrom thalassemiatraithavebeenproposedassimplediagnostictools.However,someofthebest discriminativeindicesuseparametersintheformulasthatareonlymeasuredinmodern countersandarenotalwaysavailableinsmalllaboratories.
Thedevelopmentofanindexwithgooddiagnosticaccuracybasedonlyonparameters derivedfromthebloodcellcountobtainedusingsimplecounterswouldbeusefulinthe clinicalroutine.Thus,theaimofthisstudywastodevelopandvalidateadiscriminative indextodifferentiateirondeficiencyanemiafromthalassemiatrait.
Methods:Todevelopandtovalidatethenewformula,bloodcountdatafrom106(thalassemia
trait:23andirondeficiency:83)and185patients(thalassemiatrait:30andirondeficiency: 155)wereused,respectively.Irondeficiency,-thalassemiatraitand␣-thalassemiatraitwere confirmedbygoldstandardtests(lowserumferritinforirondeficiencyanemia,HbA2>3.5%
for-thalassemiatraitandusingmolecularbiologyforthe␣-thalassemiatrait).
Results:Thesensitivity,specificity,efficiency,Youden’sIndex,areaunderreceiveroperating
characteristiccurveandKappacoefficientofthenewformula,calledtheMatos&Carvalho Indexwere99.3%,76.7%,95.7%,76.0,0.95and0.83,respectively.
Conclusion:Theperformanceofthisindexwasexcellentwiththeadvantageofbeingsolely
dependentonthemeancorpuscularhemoglobinconcentrationandredbloodcellcount obtainedfromsimpleautomaticcountersandthusmaybeofgreatvalueinunderdeveloped anddevelopingcountries.
©2016Associac¸ ˜aoBrasileiradeHematologia,HemoterapiaeTerapiaCelular.Published byElsevierEditoraLtda.ThisisanopenaccessarticleundertheCCBY-NC-NDlicense
(http://creativecommons.org/licenses/by-nc-nd/4.0/).
∗ Correspondingauthorat:InstitutoFederaldeMinasGerais,CampusOuroPreto,RuaPandiáCalógeras,898,Bauxita,35400-000Ouro
Preto,MG,Brazil.
E-mailaddress:[email protected](J.F.Matos).
http://dx.doi.org/10.1016/j.bjhh.2016.05.011
Introduction
Anemiaaffectsabout800millionchildrenandwomen world-wide.Themostcommoncausesofanemiaareirondeficiency anemia(IDA)andthalassemiatrait(TT).1–3AccordingtoWorld
HealthOrganization(WHO)estimatesin2004,IDAresultedin 273,000deathsandthelossof19.7milliondisability-adjusted lifeyears,accountingfor1.3%ofthe globaltotal,with97% occurringinlow-andmiddle-incomecountries.3,4
Differentialdiagnosisofmicrocyticanemiasisofgreat clin-icalimportancesinceprognosisandtreatmentare distinct. Thefirststeptodiagnosemicrocyticanemiasisbythe micro-scopicanalysisofbloodfilmanddeterminationofredblood cell(RBC)indicesusingcell counters.Consideringthegreat similaritybetweenIDAandTT,complementarylabmethods areneededbesidestheroutinebloodexam.Currently, diag-nosisofIDAisobtainedbyevaluatingtheironmetabolism, includingserumiron,totalserumironbindingcapacityand serumferritinmeasurements.Diagnosisofthe-thalassemia trait(-TT)isusuallymadebyhemoglobinelectrophoresisand HbA2 levels (>3.5%).5,6 Onthe other hand,diagnosis ofthe
␣-thalassemiatrait(␣-TT)isconfirmedbyinvestigating muta-tionsinthe␣gene.Despitetheirgreatutility,goldstandard testsforthediagnosisofthesemicrocyticandhypochromic anemiasinvolvetime-consumingmethodologies,highcosts andareinaccessibletopoorerpopulations.5–8
Inanattempttosimplifythedifferentialdiagnosisbetween IDA and TT, several indicesusing blood cell count param-eters have been suggested.9–16 The formula developed by
Green&King{[(MCV2)
×RDW]/(Hb×100)whereMCVismean
corpuscular volume, RDW is RBC distribution width and Hb ishemoglobin}has, accordingto many studies,agood performance.16–18However,thisformulaisdependentonthe
RDW,aparameterthatisnotprovidedbyallautomatic coun-ters.Furthermore,thecurrentlyexistingindexesreportedin theliteraturehavenotbeendevelopedusingmolecular biol-ogytodiagnose␣-thalassemia,whichisnecessarytoexclude concomitantdiseases.
Theaimofthisstudywastodevelopandvalidateanew indexthatdiscriminatesbetweenIDAandTTemployingvery simpleparametersthatareprovidedbyallautomaticblood counters.Thisstudyusedmoleculartechniquestodiagnose TTandexcludethepresenceofthisdisorderinIDApatients.
Methods
Populationevaluatedtodevelopthenewindex
Twogroupsofpatientswereselectedtodevelopthisindex, namelyover18-year-oldindividualswithIDAorTT.For inclu-sionintheIDAgroup,patientshadhemoglobinlevelsbelow 12g/dLforwomenand13g/dLformen,19MCVandmean
cor-puscularhemoglobin (MCH) below80fL and 27pg for both genders,respectively,20andferritin<6ng/mLforwomenand
28ng/mLformen,therebycharacterizingIDA.Exclusion crite-riafortheIDAgroupwerethepresenceofmalignanciesand inflammatory/infectious diseases or presenceof mutations associatedwith␣-TT(3.7,4.2,20.5,MED,SEA,THAI,FILand
Hph).Patientswiththesemutationswereexcludedsothatthis groupdidnothaveindividuals presentingthe twodiseases simultaneously.
For inclusion in the TT group, patients had the MCV below 80fL. -TT carriers were confirmed by hemoglobin electrophoresisatpH8.6andHbA2 levels>3.5%.5 ␣-TTwas
confirmed by the presenceofmutations. All patients with malignancies orinflammatory/infectious diseasesbased on clinicaldataandpersonalinformationobtainedfrom medi-calrecordswereexcludedfromthisgroup,aswerepatients sufferingfromirondeficiencyidentifiedbymeasuringferritin serumlevels.
Samplecollection,hematologicalandbiochemical evaluations
Peripheral blood (5mL) was collected in tubes containing ethylenediaminetetraaceticacid(EDTA)atafinal concentra-tionof1mg/mL.Bloodcellcountresultswereobtainedusing automaticcell counters(ABXPentraDX-120,HoribaMedical DF® in IPSEMG and Sysmex® XE-2100 in Hospital
Univer-sitário).HemoglobinelectrophoresiswasperformedatpH8.6 using asemi-automated system(Hydrasys® ofSebia®) and
HbA2wasmeasuredbytheelutionmethodusingacellulose
acetatestrip.5Inaddition,thesesampleswereusedto
investi-gateeightmutationsthatcause␣-TT(3.7,4.2,20.5,MED,SEA, THAI,FILandHph).Anothersampleoftheperipheralblood (5mL)wascollectedwithoutanticoagulanttoevaluateferritin values.SerumferritinlevelsweredeterminedbyImmulite®
DPC® usingthechemiluminescentimmunoassaytechnique
andImmulite 2000DPC® kits(DiagnosticProducts
Corpora-tion, LosAngeles,CA, USA)withreference valuesbetween 28and397ng/mLformenandbetween6and159ng/mLfor women.
Molecularanalysis
Genomic DNA was extracted from peripheralblood leuko-cytesusingtheGentraPuregeneBloodKit(Qiagen®,Germany)
accordingtothemanufacturer’sinstructions.Investigationof the deletionalmutations (3.7, 4.2, MED,20.5, SEA, FIL and THAI)wasperformedbymultiplexpolymerasechainreaction (PCR)accordingtothemethodologydescribedbyTanetal.21
OligonucleotidesweresynthesizedbyPromega® (Promega®,
USA),whosesequenceswerealsodescribedbyTanetal.21A
control,theLIS1genelocatedonchromosome17,wasusedin thereactiontodetectthesemutationsinordertoverifythe successofamplification.Afteramplification,thePCRproduct wassubjectedtoelectrophoresison1.5%agarosegel.
Developmentofthenewindex
A mathematical formula to discriminate betweenIDA and TT wasdevelopedwiththe supportofastatistician ofthe Universidade Federalde MinasGerais, Brazil, using values of the hematological parameters of 23 patients with -TT and83patientswithIDAconfirmedusinggoldstandardtests (HbA2>3.5%in-TTandlowserumferritininIDA).5
employedtocreatethisnewindex.Basedonthelargestarea underthecurve,theformulawiththebestperformancewas theonethattookintoaccounttheRBCcountandmean cor-puscularhemoglobinconcentration(MCHC)parameters.
The adjusted formula of the new index developed in this study is Matos & Carvalho Index (MCI)=(1.91×RBC)+
(0.44×MCHC).
AccordingtotheROCcurve,theMCIpresentedacut-off pointwithavalueof23.85todiscriminatebetweenIDAandTT. Iftheindexis<23.85,thepatientisclassifiedasanIDApatient, whilevalues>23.85classifytheindividualasaTTcarrier.
ValidationoftheMatos&CarvalhoIndex
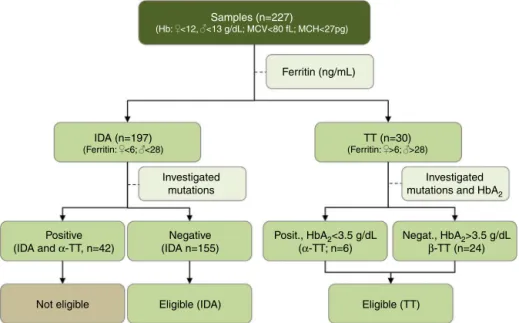
In order to validate the MCI, a cross-sectional study was conductedfrom2009to2011intwohospitals(Hospital Gov-ernadorIsraelPinheiro–IPSEMGandHospitalUniversitário) inBeloHorizonte,MinasGerais,Brazil.Atotalof227 outpa-tientswereincludedinthisstudy.Classificationofpatientsas havingIDAorTTwascarriedoutaccordingtothe aforemen-tionedcriteria.From227patients,197patientspresentedIDA and30patientswereTTcarriers.Ofthe197patientswithIDA, 42hadmutationsrelatedto␣-thalassemiaandwereexcluded
(Figure1).Thus,theIDAgroupwascomposedof155patients.
IntheTTgroup,24patientshad-TTandtheothersixpatients
had␣-TT(Figure1).
Statisticalanalysis
StatisticalanalysiswasperformedusingtheGraphPadPrism 5.0software(Graph-PadSoftwareInc.,LaJolla,CA).Normality oftheCBCdatawasassessedusingtheKolmogorov–Smirnoff test.Consideringthe nonparametricnatureofalldatasets, theMann–Whitneytestwasusedtoinvestigatedifferences betweenthetwogroups.Differenceswereconsidered signifi-cantwhenthep-value<0.05.
Thesensitivity,specificity,efficiencyandYouden’sIndex werecalculatedtoinvestigatetheperformanceoftheMCIto differentiatebetweenIDAandTT.Besidesthecalculationof theseparameters,theROCcurveofdiscriminantformulaswas plottedanditsareadeterminedusingGraphPadPrism5.0.The largestareaunderthecurveindicatestheindexmostlikelyto correctlydiscriminatepatientswithoneoftheseanemias.
ThevalidityoftheMCIwascomparedtotheresultsfrom moleculartechniques(␣-TT),HbA2measurement(-TT)and ferritin measurement (IDA) asreference standards, respec-tively.Inordertoinvestigatethedegreeofagreementbetween the results, theKappa coefficient wascalculated using the softwareOpenEpiversion2.3.1.InterpretationofKappawas according to the following scale: excellent 1.00–0.81; good 0.80–0.61;moderate0.61–0.40;weak0.40–0.21;andabsenceof agreement0.20–0whencomparedtothegoldstandardsinthe diagnosisofIDAandTT.22
Ethicalstatement
ThisstudywasapprovedbyboththeEthicsCommitteesofthe UniversidadeFederaldeMinasGerais(protocoln◦344/09)and
theHospitalGovernadorIsraelPinheiro(protocoln◦361/09)in
BeloHorizonte,Brazil.Patientswereinformedoftheobjectives ofthe research and signedinformed consentformsbefore bloodsamplinganddatacollection.
Results
Hematologicalparameters
ThemedianandinterquartilerangesofCBCparametersfor the 185patients (TT and IDA groups) enrolled in the vali-dationofMCIwerecalculated.RBC,Hb,hematocritandMCHC valuesforTTpatients weresignificantly higherthan those observedforIDApatientswhereasMCV,MCH,RDWandthe
IDA (n=197) (Ferritin: <6; <28)
Ferritin (ng/mL)
Investigated mutations
Positive
(IDA and α-TT, n=42)
Negative (IDA n=155)
Eligible (IDA) Eligible (TT)
Posit., HbA2<3.5 g/dL
(α-TT; n=6)
Negat., HbA2>3.5 g/dL
β-TT (n=24)
Investigated
mutations and HbA2
Not eligible
Samples (n=227)
(Hb: <12, <13 g/dL; MCV<80 fL; MCH<27pg)
TT (n=30) (Ferritin: >6; >28)
Table1–Hematologicalparametersofpatientswithirondeficiencyanemiaandthalassemiatraitusedtovalidatethe Matos&CarvalhoIndex.
Parameter Irondeficiencyanemia(n=155) Thalassemiatrait(n=30) p-value
RBC(106/L) 4.4(4.2–4.7) 5.4(5.1–5.7) <0.0001
Hb(g/dL) 10.0(9.3–10.7) 10.9(10.5–11.8) <0.0001
Ht(%) 32.2(30.3–33.8) 34.6(33.2–36.2) <0.0001
MCV(fL) 73.0(67.0–76.0) 63.8(61.6–68.5) <0.0001
MCH(pg) 22.7(20.8–24.1) 20.4(19.7–21.6) <0.0001
MCHC(%) 31.2(30.4–31.8) 31.8(31.2–32.6) 0.0003
RDW(%) 17.9(16.6–19.4) 15.9(15.3–16.9) <0.0001
PLT(103/L) 319.0(257.0–390.0) 233.5(197.5–275.3) <0.0001
RBC:redbloodcount;Hb:hemoglobin;Ht:hematocrit;MCV:meancorpuscularvolume;MCH:meancorpuscularhemoglobin;MCHC:mean corpuscularhemoglobinconcentration;RDW:redbloodcelldistributionwidth;PLT:plateletcount.
Thedataarepresentedasmediansandinterquartilerange.
Table2–PerformanceofMatos&CarvalhoIndexinthediscriminationbetweenirondeficiencyanemiaandthalassemia trait.
Group IDA TT
Sensitivity%(95%CI) 99.3(96.4–99.9) 76.7(59.1–88.2)
Specificity%(95%CI) 76.7(59.1–88.2) 99.3(96.4–99.9)
Accuracy(%) 95.7(91.7–97.8)
Youden’sIndex 76.0
AUC(95%CI) 0.95(0.90–1.00)
Kappacoefficient(95%CI) 0.83(0.68–0.97)
IDA:irondeficiencyanemia;TT:thalassemiatrait;95%CI:95%confidenceinterval;AUC:areaunderreceiveroperatingcharacteristiccurve.
plateletcountweresignificantlylowerthanthoseobservedfor IDApatients.Hematologicaldataofthetwostudygroupsare showninTable1.
Validationoftheindex
MCIvalueswerecalculatedforallpatientsusingCBCdata. MCIcorrectlydiagnosed154(99.3%)ofthe155patientswith IDAand23(76.7%)ofthe30patientswithTT.MCIperformance wasanalyzedbycalculatingsensitivity[99.3;95%confidence interval(95%CI):96.4–99.9],specificity(76.7;95%CI:59.1–88.2), accuracy(95.7;95%CI:91.7–97.8),areaunderROCcurve(0.95; 95%CI:0.90–1.00),Youden’sIndex(76.0)andthekappa coeffi-cient(0.83;95%CI:0.68–0.97)(Table2).Figure2showstheROC curvefortheMCI.Thisindexshowedhighaccuracyandhas revealedexcellentagreementwiththegoldstandard diagnos-tictechniquesaswellasgoodclinicalapplicabilityasamethod ofscreening.
Discussion
Inthisstudy,anewindex(MCI)wasdevelopedusingdetailed statistical analysiswith the aimof accurately discriminat-ing between IDA and TT. This formula produced excellent diagnosticaccuracyandtheparameters,suchassensitivity, efficiency,Youden’sIndex,andareaundertheROCcurvewere quitehigh.MCI showed excellentagreement withthegold standardmethodsofdiagnosisfortheseanemias, showing goodapplicabilityasascreeningtoolintheclinicalpractice.
100
50
0
0 50
100%-specificity%
Sensitivity
, %
100
Figure2–Receiveroperatingcharacteristiccurveforthe Matos&CarvalhoIndex.
Furthermore,theMCIwasdevelopedusingamolecular biol-ogytechniquetodiagnose␣-thalassemia,whichisnecessary to exclude concomitant diseases. The indexes currently described in the literature were not developed using this technique.
thosethatcorrectlydiagnoseTT.Thisisbecauseonemonth ofiron supplementation to a patient mistakenly classified ashavingIDAwhoinfacthasTT,causeslessdamagethan thelackofthissupplement toapatientwithIDAwhowas mistakenlyclassifiedasaTTcarrier.Ironanderythropoietin arerequiredfortheformationofhemoglobin.5Furthermore,
iron isa component requiredin critical cellularprocesses suchasthetransportandutilizationofoxygen,productionof adenosinetriphosphate(ATP),DNAsynthesis,metabolismof catecholamines,mitochondrialelectrontransportandother physiologicalprocesses.23–25Thus,biologicalsystems
includ-ingtheimmuneandneurologicalsystems,areallaffectedby thelackofiron.23Forthesereasonsthelackoftreatmentfor
IDAwouldbeveryharmfultotheindividual.
Despite the moderate sensitivity for detecting patients withthalassemia,applicationoftheMCIisstilladvantageous ifthereissuspicionofthisdisorder.Inthiscase,thepatient whopresentedavalueofMCI>23.85shouldbereferredfor confirmationbyconventionalmethods.Nevertheless,in prac-ticaltermsthis isanadvantage,becauseforthis group,an investigationofironlevelscanbeconsideredasecondoption. The MCI represents a breakthrough in discriminating between IDA and TT, with potential for wide application considering its advantage ofbeing dependent only on the numberofRBCsandtheMCHC.Itshouldbenotedthatthese twoparametersareobtainablefrommostsimplecellcounters, thereforesophisticatedautomaticcountersarenotrequired. Hence,theMCIcanbeappliedinareaswhereadvanced tech-nologiesarenotavailableinclinicallabs.
Despitetheadvantagesandsimplicityofthe implementa-tionoftheMCIinthelaboratorypractice,thereisalimitation ofMCIandotherdiscriminatingformulassincetheyarenot abletodifferentiateallcasesofIDAfromTT.Inlightofthis, twosituationscanoccurthatdeservespecialattention:(i)the indexindicatedTT,butthepatienthadIDAand(ii)the appli-cationoftheindexindicatedIDA,butthepatientwasaTT carrier.Inthe firstcase,patientfollow-upisnecessaryand willindicate,overtime,asignificantreductionincirculating hemoglobinlevels,promptingthephysiciantorequest inves-tigativetestsofironmetabolism.Inthelattersituation,patient follow-upisalsoneededwhichwillshowtheneedfor medi-calprocedures.Inthiscase,theprescriptionofironwouldnot increasethehemoglobinlevelduetothegeneticdisorder.In casesofconcomitantdiseases,monitoringcanalsoclarifythe bestmedicalapproach.Therefore,MCIisausefultoolin guid-ingthephysicianregardingtheinitialconducttobeadopted; however,itdoesnoteliminatetheneedofafollow-upthat eventuallymayrequireconfirmatorytests.
Conclusion
TheMCIhas anexcellentperformanceand may be poten-tiallyusefulforscreeningpatientswithmicrocyticanemias. TheroutineuseoftheMCIprovidedgreatdiagnosticaccuracy, andcould contributedecisivelytoguidethe choiceof con-firmatorylaboratorytestsleadingtoacorrectdiagnosisand treatment.Thiswouldresultinasignificantcostsavingforthe health system, especially advantageous in underdeveloped anddevelopingcountrieswithlimitedfinancialresources.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1.KassebaumNJ,JasrasariaR,NaghaviM,WulfSK,JohnsN,
LozanoR,etal.Asystematicanalysisofglobalanemia
burdenfrom1990to2010.Blood.2014;123(5):615–24.
2.PasrichaSR.Anemia:acomprehensiveglobalestimate.Blood.
2014;123(5):611–2.
3.WorldHealthOrganization.Theglobalprevalenceofanaemia in2011.Availablefrom:http://www.who.int/nutrition/
publications/micronutrients/globalprevalenceanaemia
2011/en/.
4.MathersC,StevenG,MascarenhasM.Globalhealthrisks:
mortalityandburdenofdiseaseattributabletoselectedmajor
risks.Geneva,Switzerland:WorldHealthOrganization;2009.
5.RodakBF,FritsmaGA,KeohaneEM.Hematology:clinical
principlesandapplications.4thed.ElsevierSaunders;2011.
6.MartinA,ThompsonAA.Thalassemias.PediatrClinNorth
Am.2013;60(6):1383–91.
7.CaoA,GalanelloR.Beta-thalassemia.GenetMed.
2010;12(2):61–76.
8.HandinRJ,LuxSE,StosselTP,editors.Blood:principlesand
practiceofhematology.2nded.Philadelphia:Lippincott
Williams&Wilkins;2003.
9.MentzerWCJr.Differentiationofirondeficiencyfrom
thalassaemiatrait.Lancet.1973;1(7808):882.
10.SrivastavaPC.Differentiationofthalassaemiaminorfrom
irondeficiency.Lancet.1973;2(7821):154–5.
11.ShineI,LalS.Astrategytodetectbeta-thalassaemiaminor.
Lancet.1977;1(8013):692–4.
12.RicercaBM,StortiS,d’OnofrioG,ManciniS,VittoriM,Campisi
S,etal.Differentiationofirondeficiencyfromthalassaemia
trait:anewapproach.Haematologica.1987;72(5):409–13.
13.GreenR,KingR.Anewredcelldiscriminantincorporating
volumedispersionfordifferentiatingirondeficiencyanemia
fromthalassemiaminor.BloodCells.1989;15(3):481–91,
discussion492–5.
14.JayaboseS,GiavanelliJ,Levendoglu-tugalO,SandovalC,
OzkaynakF,VisintainerP.Differentiatingirondeficiency
anemiafromthalassemiaminorbyusinganRDW-based
index.JPediatrHematolOncol.1999;21(4):314.
15.EhsaniMA,ShahgholiE,RahiminejadMS,SeighaliF,Rashidi
A.Anewindexfordiscriminationbetweenirondeficiency
anemiaandbeta-thalassemiaminor:resultsin284patients.
PakJBiolSci.2009;12(5):473–5.
16.SirdahM,TaraziI,AlNajjarE,AlHaddadR.Evaluationofthe
diagnosticreliabilityofdifferentRBCindicesandformulasin
thedifferentiationofthebeta-thalassaemiaminorfromiron
deficiencyinPalestinianpopulation.IntJLabHematol.
2008;30(4):324–30.
17.ShenC,JiangYM,ShiH,LiuJH,ZhouWJ,DaiQK,etal.
Evaluationofindicesindifferentiationbetweeniron
deficiencyanemiaandbeta-thalassemiatraitforChinese
children.JPediatrHematolOncol.2010;32(6):e218–22.
18.JanelA,RoszykL,RapatelC,MareynatG,BergerMG,
Serre-SapinAF.Proposalofascorecombiningredbloodcell
indicesforearlydifferentiationofbeta-thalassemiaminor
fromirondeficiencyanemia.Hematology.2011;16(2):123–7.
19.WorldHealthOrganization.Guidelinesfortheuseofiron
supplementstopreventandtreatirondeficiencyanemia;
20.WorldHealthOrganization.Irondeficiencyanaemia:
assessment,preventionandcontrol.Aguideforprogramme
managers;2001.p.114.
21.TanAS,QuahTC,LowPS,ChongSS.Arapidandreliable
7-deletionmultiplexpolymerasechainreactionassayfor
alpha-thalassemia.Blood.2001;98(1):250–1.
22.SzkloM,NietoJ.Epidemiology:beyondthebasics.2nded.
Jones&BartlettLearning;2007.
23.NobiliB,PerrottaS,MatareseSMR,ConteML,GiudiceEM.
EvaluationofbodyironstatusinItaliancarriersof
beta-thalassemiatrait.NutrRes.2001;21(1/2):55–60.
24.BeardJL,DawsonH,PineroDJ.Ironmetabolism:a
comprehensivereview.NutrRev.1996;54(10):295–317.
25.CohenAR,GalanelloR,PennellDJ,CunninghamMJ,Vichinsky
E.Thalassemia.HematologyAmSocHematolEducProgram.