O
r
IG
IN
A
l
A
r
T
IC
lE
Prevalence of human papillomavirus (HPV) and HPV-16
genotyping by real-time PCR in patients with several
cervical pathologies
ABSTRACT
Purpose:thisstudywasplannedtoevaluatetheprevalenceofHPV(exceptingtype16)andHPV 16byreal-timePCRincolposcopypatientsandtointerpretetheresultswithage,ageoffirstsexual intercourse(FSI),parityandPapsmearresults.Methods:onehundredandtwocolposcopypatients (50and52ofthepatientswereclassifiedascolposcopypositiveandnegative,respectively)applying toGynecologyclinicwereincluded.HPV(exceptingtype16)andHPV16weredetectedbyreal-timePCRusingtheL1region.Real-timenestedamplificationsofMY09/11productsweredoneby GP5+/GP6+primersandCyanine-5labeledHPVandHPV16DNAspecificprobeafterHPVDNA extractionbyphenolchloroformisoamylalcohol.Results:HPV(exceptingtype16)andHPV16 werepositivein12%and18%ofthecolposcopypositivepatientsrespectively.HPV(exceptingtype 16)andHPV16werepositivein5.7%and3.8%ofthecolposcopynegativepatients,respectively. Conclusion: therewasastatisticallysignificantdifferencebetweencolposcopypositiveandcol-poscopynegativepatientscomparingHPV16withtotalHPVpositivity(p=0.021fortype16and p=0.010fortotalHPV)buttherewasnotastatisticallysignificantdifferencebetweencolposcopy positiveandcolposcopynegativepatientswhenwecomparedHPV(exceptingtype16)positivity (p=0.314).Inconclusion,HPVdetectionandtypingmaybehelpfulforcervicalcancerscreening andprevention.
Keywords:HPVtype16,real-timePCR,colposcopy.
[Braz J Infect Dis 2010;14(1):19-23]©ElsevierEditoraLtda.
Authors
BediaDinc1 SeyyalRota1 AnilOnan2 GulendamBozdayi1 CagatayTaskiran2 AydanBiri2 HaldunGüner2
1DepartmentofMedical Microbiologyand
2DepartmentofObstetrics
andGynecologyofGazi University,Facultyof Medicine,06500,Ankara, Turkey.
Submittedon:07/20/2009 Approvedon:11/05/2009
Correspondence to:
BediaDinc,MD GaziUniversity,Facultyof Medicine,
DepartmentofMedical Microbiology, DekanlikBinasi,2.Kat, Besevler,Ankara,Turkey, 06500.
Tel:+903122024628 Fax:+903122124647 E-mail:
bhdogan@yahoo.com
Wedeclarenoconflict
INTRODUCTION
Cervicalcancerisgloballythesecondmostcom-moncancerinwomenwithapproximately493,000 newcasesannually.1 Alsocervicalcancerisanim- portantpublichealthproblemindevelopingcoun-tries.Thereisastatisticallysignificantrelationship betweencervicalcancerandthenumberofsexual partnersofawoman.Thesedatadirectustosexu-ally transmitted agents, especipartnersofawoman.Thesedatadirectustosexu-ally viruses.2,3 As a result of the epidemiological studies on cervical cancer, there is no doubt about the importance of human papillomavirus (HPV), especially type 16and18,asanaetiologicalagent.2-5 Asaconse- quence,thedetectionandtreatmentofHPVinfec-tion can be an important stage in diagnosis and treatmentofcervicalcancer.ThePapsmeartest, as the most widely used cancer screening test in the world, is cost effective and organised screen-ingprovidedadeclineincervicalcancermortality. However, in developing countries, where screen-ing programmes are uncommon, cervical cancer
ofdeathamongwomen.HPVcanonlybereliably detectedbyDNAbasedtestssincemorphological changesoncytologysuchaskoilocytosisarenot specificforoncogenicHPVs.Theseobservations underscore the need to develop more effective diagnostic methods.6,7 PCR based methods am-plifyingnucleicacidsofHPVarecommonlyused becauseofthelimitedsensitivityofPapsmeartest leadingtosamplepreparationandinterpretation problems, insufficient serological tests and im-possibilityofinvitrocultures.DetectionofHPV DNA,especiallyinlatentinfections,canbehelp-fulindetectingcancerandprecursorlesions.8This studywasdesignedtoevaluatetheeffectofHPV, especiallytype16incolposcopypatients,bothwith andwithoutcervicalpathologiesandtoassociateit withtheclinicalfindingsinourhospital.
MATERIAL AND METHODS
includ-observationofcervicalacetowhiteepithelium,punctuation, atypical vascularisation or mosaic pattern in colposcopic examinationwereclassifiedascolposcopypositivepatients, and patients with normal colposcopic findings were clas-sifiedascolposcopynegative.Allcolposcopypatientswere screenedbyPapsmeartestandrepliedaquestionnairein-cludingquestions,suchasage,ageofFSI,andparity.
Samples: cervicalsmearsampleswerecollectedintubes containing3-5mLsterilephosphatebufferedsaline(PBS)in GynecologyClinicofGaziUniversityMedicalFacultyfrom colposcopypatientsbeforeapplicationofaceticacid.After transportingtomoleculardiagnosislaboratory,allsamples werevortexedandaliquotedin1.5mLeppendorftubesand frozenat-86ºCuntilDNAextraction.
DNA extraction: the cervical samples were digested in a buffercontaining20mg/mLproteinaseK(20mM(NH4)2SO4, 75mMTrisHCl[pH8,8]0,1%Tween20)at55°Cfor3hours; followedby10minutesat95°C.DNAisolationwasperformed by phenol-chloroform extraction and ethanol precipitation. DNAwasthensuspendedinsteriledistilledwaterandstoredat -86°Cuntilamplification.
DNA amplification: nested real-time PCR method was used for the analysis of HPV DNA and HPV 16 positivity. MY09/11primerset(5’-CGTCCMARRGGAWACTGATC-3’), (5’-CMCAGGGWCATAAYAATGG-3’,TibMolbiol,Germany) wasusedforPCRamplificationsfollowingtheextractionofthe DNA. Real-time nested amplifications of MY09/11 products weredonebyGP5+/GP6+primersandCyanine-5labeledHPV 16DNAspecificprobe[PrimerF5’TTTGTTACTGTGGTA- GATACTAC3’,PrimerR5’GAAAAATAAACTGTAAATCAT- ATTC3’,Cy5.0signalprobe5’Cy5-GTTTCTGAAGTAGATAT-GGCAGCACA-biotin3’(TibMolbiol,Germany)].Real-time PCRproductanalysiswasdonebymeltingcurveanalysison LightCyclerSoftwareversion3.5.3(LC2.0RocheDiagnostics, Germany).Meltingpeaksof78-82°Cshowedthedetectionof HPVDNAinthesample.Probemeltingpeaksofpositivesam-pleshavebeenanalyzedinthesamerunandHPV16positive samplesyieldedpeaksaround68°C.
Ethical review of the proposal and the consent
The research proposal was approved by the ethical review boardoftheFacultyofMedicine,GaziUniversity.Informed consentwasobtainedfromallwomenpriortothesample collection.
RESULTS
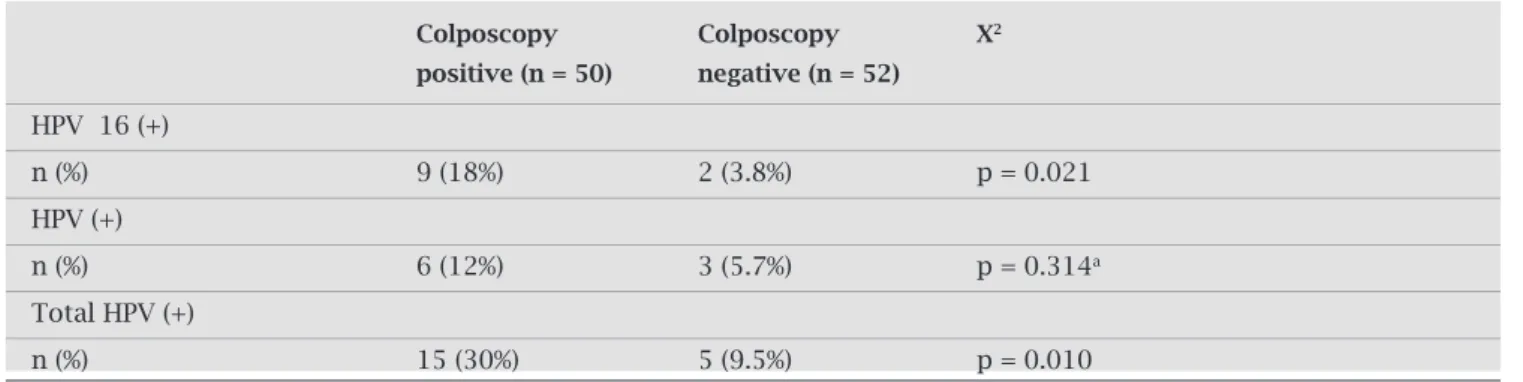
A total of 50 colposcopy positive women (18-63 years old; mean age ± SD: 39 ± 7) and 52 colposcopy negative women(17-65yearsold;meanage±SD:40±9)werein-cluded in the study. There was a statistically significant differencebetweencolposcopypositiveandcolposcopynega- tivepatientswhenwecomparedHPV16andtotalHPVposi-tivitybyPearsonchi-squaretest(p=0.021fortype16and p=0.010fortotalHPV)buttherewasnotastatisticallysig- nificantdifferencebetweencolposcopypositiveandcolposco-pynegativepatientswhenwecomparedHPV(exceptingtype 16)positivitybyFisher’sexacttest(p=0.314)(Table1).
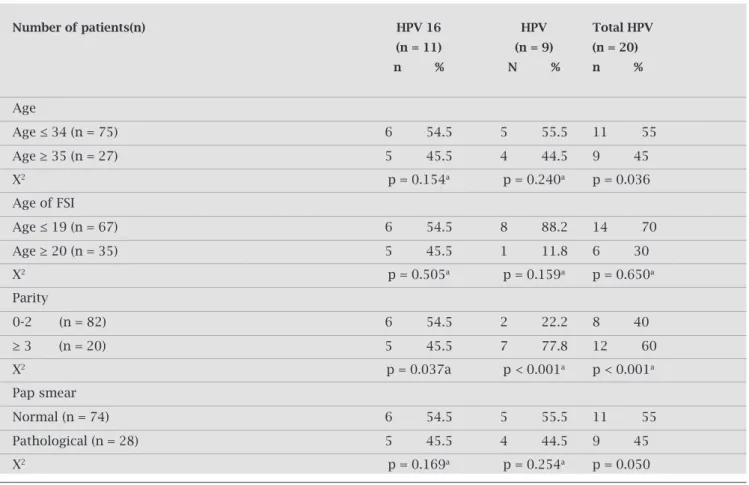
According to age of women included in the study there wasnotastatisticallysignificantdifferencebetweenpatients age≤ 34 and≥ 35 when we compared HPV 16 and HPV (exceptingtype16)positivitybyFisher’sexacttest(p=0.154) fortype16andp=0.240forHPV(exceptingtype16)but therewasastatisticallysignificantdifferencebetweenpatients age≤34and≥35whenwecomparedtotalHPVpositivityby Pearsonchi-squaretest(p=0.036)(Table2).
There was no statistically significant difference between FSIinpatients≤19-yearoldandFSIinpatients≥20-yearold when we compared HPV 16, HPV (excepting type 16) and totalHPVpositivitybyFisher’sexacttest(p=0.505fortype 16,p=0.159forHPV(exceptingtype16)andp=0.650for totalHPV)(Table2).
Parity seems statistically significant between 0-2 parity patientsand≥3paritypatientswhenwecomparedHPV16, HPV(exceptingtype16)andtotalHPVpositivitybyFish-er’sexacttest(p=0.037fortype16andp<0.001forHPV (exceptingtype16)andp<0.001fortotalHPV)(Table2).
Table 1. Distribution of HPV DNA PCR results according to patient groups
Colposcopy Colposcopy X2
positive (n = 50) negative (n = 52)
HPV 16 (+)
n (%) 9 (18%) 2 (3.8%) p = 0.021
HPV (+)
n (%) 6 (12%) 3 (5.7%) p = 0.314a
Total HPV (+)
n (%) 15 (30%) 5 (9.5%) p = 0.010
According to examination of Pap smear results, there wasnotastatisticallysignificantdifferencebetweenthepa-tientswithnormalPapsmearandpathologicalPapsmear whenwecomparedHPV16andHPV(exceptingtype16) positivitybyFisher’sexacttest(p=0.169fortype16and p=0.254forHPV(exceptingtype16)butthepvaluewas 0.050byPearsonchi-squaretestwhichisaborderlinevalue whenwecomparedtotalHPVpositivity(Table2).
DISCUSSION
BecauseHPV,especiallytype16,isreportedasanimportant riskfactorfordevelopmentofcervicaldysplasiaandcancer, and99.7%ofthecervicalcancerscanbeprovedrelatedto HPV,oneofthemaingoalsofpreventingcervicalcanceris screeningforHPV.9
Tunceret al.10detected in their study 12.5%, 19.4%, 46.3%and83.3%ofHPVtype16positivityinCINI,CIN II,CINIIIandinvasivecarcinomaspecimens,respectively, byPCRinTurkishpatients.AlsoOnanet al.11fromTurkey
andCINIIIspecimens,respectively,andHPVpositivityin patients with CIN III was significantly higher than in pa-tientswithCINIandCINII.
In 102 colposcopy patients (both colposcopy positive andnegative),a19.6%ofpositivityinfavouroftotalHPV wasdetectedinourstudy,9.5%positivityand30.0%positiv-ityincolposcopynegativeandpositivegroups,respectively. This result indicates that there is a statistically significant differencebetweentwogroups(p=0.010).
In joint assessment of HPV (excepting type 16) and HPV16wefounda55%ofpositivityinpatientsyoung-erthan34anda45%ofpositivityinpatientsolderthan 35.Ourresultscorrelatewiththestudiesindicatingade-creaseinHPVinfectionastheageincreases,12-15although Koet al.16intheirstudyfoundthatwomenbetweenages 30-69 had lower HPV positivity rate (ranging from 14-34%)comparedtowomenwithyoungerthan30andolder than70(rangingfrom47-52%).
ConsideringtotalHPV,wefound70%positivecasesin thegroupwhoexperiencedtheirFSIattheageof19orbe-Table 2. Distribution of HPV positivity according to variables
Number of patients(n) HPV 16 HPV Total HPV
(n = 11) (n = 9) (n = 20)
n % N % n %
Age
Age ≤ 34 (n = 75) 6 54.5 5 55.5 11 55
Age ≥ 35 (n = 27) 5 45.5 4 44.5 9 45
X2 p = 0.154a p = 0.240a p = 0.036
Age of FSI
Age ≤ 19 (n = 67) 6 54.5 8 88.2 14 70
Age ≥ 20 (n = 35) 5 45.5 1 11.8 6 30
X2 p = 0.505a p = 0.159a p = 0.650a
Parity
0-2 (n = 82) 6 54.5 2 22.2 8 40
≥ 3 (n = 20) 5 45.5 7 77.8 12 60
X2 p = 0.037a p < 0.001a p < 0.001a
Pap smear
Normal (n = 74) 6 54.5 5 55.5 11 55
Pathological (n = 28) 5 45.5 4 44.5 9 45
X2 p = 0.169a p = 0.254a p = 0.050
FSI: first sexual intercourse.
their FSI at the age of 20 or after.Although there is not a statistically significant difference between the two groups and HPV results, our findings confirmed the findings of previousstudies12,17,18thatHPVinfectionismorecommon amongwomenwhoexperiencedtheirFSIintheearlyages. Consequently,Floreset al.19indicateintheirstudythatolder ageatFSIissignificantlyassociatedwithadecreasedriskof highgradeCINorcancerinHPV(+)women.
Therearecontradictoryresultsinthestudiesevaluating HPV-cervicalcancerandparityrelationship,someofwhich suggest that there is no association with parity and HPV-cervicalcancer.20,21ConsideringallHPVtypes,40%positive casesinthegroupwhogavetwoorlessbirthsand60%posi-tivecasesinthegroupwhogavethreeormorebirthswere foundinourstudyandHPVseemsstatisticallysignificant between0-2paritypatientsand≥3paritypatientswhenwe comparedHPV16,HPV(exceptingtype16)andtotalHPV positivity.Shieldset al.22andCastellsagueet al.23suggestthat HPVexposedwomenwithhighparityareatincreasedrisk forcervicalcancer.Ourstudysuggeststhatparityseemsa riskfactorforHPVandconsequentlyforcervicalcancer.On thecontrary,Sellorset al.17inCanadadetectedapositivity of17.1%inwomenwhonevergavebirth,12.7%inwomen who gave one birth, 8.7% in women who gave two births and6.5%inwomenwhogavethreeormorebirths.
We first performed colposcopy and cytology and than detectedHPVDNAbyreal-timePCR.Itissuggestedthat,as comparedtoPaptesting,HPVtestinghasgreatersensitivity forthedetectionofCIN,24andtheadditionofaHPVtest toaPaptesttoscreenwomenintheirmid30’sforcervical cancer reduces the incidence of grade 2 or 3 CIN or can-cer.25Cuzick26emphasizesinareviewthatdetectionofHPV DNA by molecular tests is more sensitive but less specific andthereforetheuseofthesemethodstogethermustbethe goldstandardincervicalscreeningprograms.
Grceet al.27 detected HPV DNA in colposcopy positive patientsinZagreb.Theresultswere64.4%positive.Asthey detectedthesmearscytologically,theycametotheconclusion thatasthegradeofSILincreasedtherewasanincreaseinthe prevalanceofhighriskHPVs(HPVDNAtype16prevalance was8.5%and17.1%inLSILandHSILcases,respectively). The results were found to be statistically significant. Fifeet al.28 found35%HPV16positivityinwomenwithpathologi-calPapsmearresultsintheirstudybyPCR.AllASCUS,LSIL, HSIL,CINI,CINII,CINIIIcasesinourstudywereconsid-eredaspathologicalPapsmearresults.Wefound45.5%HPV 16positivityinwomenwithpathologicalPapsmearresults. While 45% positivity (total HPV) was found in the group withpathologicalPapsmearresults,theratioofHPVpositiv-ity(totalHPV)was55%inthegroupwithnormalPapsmear results.Ourresultsarenotcorrelatedwithliteratureresults. Thereasonforthisdeclinationmaybeduetosampleprepara-tionorsmearresultsinterpretation.
HPVDNA,especiallytype16,followedbytype18,45, 31and33isdiagnosedinmorethan99%ofcervicalcancer biopsies.Althoughtheratioschangeaccordingtothediag- nosismethod,HPVtype16isdetectedin33-50%ofcervi-calcancertissuesandthisisthemainreasontofocuson type16.29Antonishynet al.30foundintheirstudythatthe mostcommonlyidentifiedgenotypeinpatientswithcervi-calintraepithelialneoplasiagrade2orworsewasHPV-16 (46.7%),followedbyHPV-31(14.7%)andHPV-18(3.9%) by real-time polymerase chain reaction. They found that HPV-31iscontributingsignificantlytotheproportionof womenwithcervicalintraepithelialneoplasiaintheirstudy populationandshowsahigherprevalencethanHPV-18in high-gradelesions.
Westudiedsmearsamplesandfounda19.6%positiv-ity in all colposcopy patients. This result shows the high sensitivityofreal-timePCR.Real-timePCR,themostad-vancedandsensitiveofthemolecularmethods,wasused inourstudy.Sincetheexperimentoccurredinacloseden-vironment,therewasminimalcontaminationcomparedto conventionalmethodsandthesensitivityishigherdueto onemorestepinamplification.31,32
WedetectedbothHPVandHPV16positivitybyreal- timePCR.Inordertodetecttypesotherthan16,sequenc-ingcanbeperformed.
Inconclusion,weaimedtodetecttheprevalenceofHPV, especiallytype16incolposcopypatientsasaspecificgroup withcervicalcomplaintsandthisisthefirststudyreflecting theHPVresultsofcolposcopypatientsinourcountry.Since cervicalcancerisanimportantpublichealthproblemamong womenandHPV,especiallytype16issignificantlyrelated with cervical cancer, detection of HPV will be helpful for designingeffectivecervicalcancerpreventionprograms.We thinkthatfurtherstudiesincludinglargenumbersofcolpos-copypatientsmustbeperformedtoevaluatetheprevalance ofHPVinthispatientgroup.
REFERENCES
1. MonkBJ,HerzogTJ.Theneweraofcervicalcancerprevention: HPVvaccination.GynecolOncol2008;109(2Suppl):S1-3. 2. BenedetJL.Progressingynecologiccancerdetectionandtreatment.
InternationalJournalofGynecologyandObstetrics2000;70:135-47.
3. SpinilloA,DebiaggiM,ZaraFet al.HumanImmunodeficiency
VirusType1-RelatedNucleicAcidsandPapillomavirusDNA inCervicovaginalSecretionsofImmunodeficiencyVirus-In-fectedWomen.ObstetricsandGynecology2001;97:999-1004.
4. Matos E, Loria D,Amestoy GMet al. Prevalance of Human
Papillomavirus Infection Among Women in Concordia, Ar- gentina:APopulation-BasedStudy.SexuallyTransmittedDis-eases2003;27:593-9.
5. Moberg M, Gustavsson I, Gyllensten U. Real-Time PCR-Based SystemforSimultaneousQuantificationofHumanPapillomavi-rusTypesAssociatedwithHighRiskofCervicalCancer.Journal ofClinicalMicrobiology2003;41:3221-8.
7. PerronsC,KleterB,JelleyRet al.DetectionandGenotypingof HumanPapillomavirusDNAbySPF10andMY09/11Primers inCervicalCellsTakenFromWomenAttendingaColposcopy Clinic.JournalofMedicalVirology2002;67:246-52.
8. SeversonJ,EvansT,LeePet al
.HumanPapillomavirusInfec-tions: Epidemiology, Pathogenesis, and Therapy. Journal of CutaneousMedicineandSurgery2001;5:43-60.
9. MayeauxEJ.ReducingtheeconomicburdenofHPV-related diseases.JAmOsteopathAssoc2008;108(4Suppl2):52-7. 10. Tuncer S, Ustacelebi S. Detection of Human Papillomavirus
Type16and18bypolymerasechainreactionincervicalbi-opsysamples.Flora1996;1:40-4.
11. OnanMA,TaskiranC,BozdayiGet al.Assessmentofhuman
papillomaviralloadofarchivalcervicalintraepithelialneopla- siabyreal-timepolymerasechainreactioninaTurkishpopu-lation.EurJGynaecolOncol2005;26(6):632-5.
12. OzturkS,KaleliI,KaleliB,BirF.Investigationofhumanpapil-lomavirusDNAincervicalspecimensbyhybridcaptureassay. MikrobiyolBul2004;38(3):223-32.
13. OliveiraLH,RosaML,PereiraCRet al
.Humanpapillomavi-rus status and cervical abnormalities in women from public andprivatehealthcareinRiodeJaneiroState,Brazil.RevInst MedTropSãoPaulo2006;48(5):279-85.
14. Dunne EF, Unger ER, Sternberg Met al. Prevalence of HPV
infection among females in the United States. JAMA 2007; 297(8):813-9.
15. FerreccioC,CorvalanA,MargozziniPet al
.Baselineassess-ment of prevalence and geographical distribution of HPV typesinChileusingself-collectedvaginalsamples.BMCPub-licHealth2008;8:78.
16. KoV, Nanji S, Tambouret RH,Wilbur DC. Testing for HPV asanobjectivemeasureforqualityassuranceingynecologic cytology:positiveratesinequivocalandabnormalspecimens and comparison with theASCUS to SIL ratio. Cancer 2007; 111(2):67-73.
17. Sellors JW, Mahony JB, Kaczorowski Jet al. Prevalence and
predictors of human papillomavirus infection in women in Ontario,Canada.CMAJ2000;163:503-8.
18. KahnJA,RosenthalSL,SuccopPAet al
.Mediatorsoftheasso-ciationbetweenageoffirstsexualintercourseandsubsequent humanpapillomavirusinfection.Pediatrics2002;109(1):E5.
19. FloresYN,BishaiDM,ShahKVet al.Riskfactorsforcervical
canceramongHPVpositivewomeninMexico.SaludPublica Mex.2008;50(1):49-58.
20. CastlePE,WacholderS,LorinczATet al.Aprospectivestudyof
high-gradecervicalneoplasiariskamonghumanpapilloma- virus-infectedwomen.JNatlCancerInst.2002;94(18):1406-14.
21. Vaccarella S, Herrero R, Dai Met al. Reproductive factors,
oralcontraceptiveuse,andhumanpapillomavirusinfection: pooledanalysisoftheIARCHPVprevalencesurveys.Cancer EpidemiolBiomarkersPrev.2006;15(11):2148-53.
22. Shields TS, Brinton LA, Burk RDet al.A case-control study
ofriskfactorsforinvasivecervicalcanceramongU.S.women exposedtooncogenictypesofhumanpapillomavirus.Cancer
EpidemiolBiomarkersPrev.2004;13(10):1574-82.
23. CastellsaguéX,DíazM,deSanjoséSet al.Worldwidehuman
papillomavirus etiology of cervical adenocarcinoma and its cofactors: implications for screening and prevention. J Natl CancerInst.2006;98(5):303-15.
24. Mayrand MH, Duarte-Franco E, Rodrigues Iet al. Human
papillomavirusDNAversusPapanicolaouscreeningtestsfor cervicalcancer.NEnglJMed.2007;357(16):1579-88.
25. Naucler P, Ryd W, Törnberg Set al. Human papillomavirus
andPapanicolaouteststoscreenforcervicalcancer.NEnglJ Med.2007;357(16):1589-97.
26. Cuzick J. Role of HPV testing in clinical practice.Virus Re-search.2002;89:263-9.
27. GrceM,HusnjakK,BozikovJet al
.EvaluationofGenitalHu-manPapillomavirusInfectionsbyPolymeraseChainReaction among Croatian Women. Anticancer Research. 2001; 21:579-84.
28. FifeKH,CramerHM,SchroederJM,BrownDR.Detectionof Multiple Human Papillomavirus Types in the Lower Genital TractCorrelatesWithCervicalDysplasia.JournalofMedical Virology.2001;64:550-9.
29. YangYY, Koh LW, Tsai CHet al. Correlation of viral factors
withcervicalcancerinTaiwan.JMicrobiolImmunolInfect. 2004;37:282-7.
30. AntonishynNA,HorsmanGB,KellnRAet al.Theimpactof
thedistributionofhumanpapillomavirustypesandassociated high-risk lesions in a colposcopy population for monitoring vaccineefficacy.ArchPatholLabMed.2008;132(1):54-60.
31. ShahKV,SolomonL,DanielRet al.ComparisonofPCRand
HybridCaptureMethodsforDetectionofHumanPapilloma-virusinInjectionDrug-UsingWomenatHighRiskofHuman ImmunodeficiencyVirusInfection.JournalofClinicalMicro-biology.1997;35:517-9.