Pleasecitethisarticleinpressas:CalsavaraAJC,etal.Post-sepsiscognitiveimpairmentandassociatedriskfactors:Asystematicreview. AustCritCare(2017),http://dx.doi.org/10.1016/j.aucc.2017.06.001
ARTICLE IN PRESS
G Model
AUCC-376; No.ofPages11
AustralianCriticalCarexxx(2017)xxx–xxx
Contents lists available atScienceDirect
Australian
Critical
Care
j o u r n a l h o m e p a g e :w w w . e l s e v i e r . c o m / l o c a t e / a u c c
Review
Paper
Post-sepsis
cognitive
impairment
and
associated
risk
factors:
A
systematic
review
Allan
J.C.
Calsavara
a,b,∗Vandack
Nobre
bTatiana
Barichello
c,dAntonio
L.
Teixeira
b,daSchoolofMedicine,UniversidadeFederaldeOuroPreto,OuroPreto,MG,Brazil
bPostgraduatePrograminHealthSciences:InfectologyandTropicalMedicine,SchoolofMedicine,UniversidadeFederaldeMinasGerais,BeloHorizonte,
MG,Brazil
cUniversidadedoExtremoSulCatarinense,Criciúma,SC,Brazil
dTranslationalPsychiatryProgram,DepartmentofPsychiatryandBehavioralSciences,MedicalSchool,TheUniversityofTexasHealthScienceCenterat
Houston,Houston,TX,USA
a
r
t
i
c
l
e
i
n
f
o
r
m
a
t
i
o
n
Articlehistory: Received13July2016
Receivedinrevisedform10May2017 Accepted 4 June 2017
Keywords: Cognition
Neuropsychologicaltests Sepsis
Sepsis-associated encephalopathy
a
b
s
t
r
a
c
t
Introduction:Post-sepsiscognitiveimpairmentisoneofthemajorsequelaeobservedinsurvivorsof
sepsis.Thiscognitiveimpairmentcanbeglobalormayaffectspecificdomains.Abetterunderstanding ofthesedeficitsandassociatedriskfactorscouldinfluencethecareofpatientswithsepsis.
Objective:Toperformasystematicreviewtoinvestigatethepresenceofcognitiveimpairmentandits
associatedriskfactorsamongpatientswhosurvivedsepsis.
Methods:ThesearchwasconductedinMEDLINE(1966toMarch2017)andEMBASE(1988toMarch2017).
Weincludedstudieswithindividualswhowere18yearsorolderwithpost-sepsiscognitiveimpairment.
Results:Weanalysed 577articles.Sixteenstudiesmettheinclusioncriteria.Morethan74,000,000
patientswereevaluatedintheselectedstudies.Significantvariationwasobservedinthedefinition ofsepsisandcognitiveimpairment.TwelvestudiesusedACCP/SCCMcriteriaforsepsis,while cogni-tiveimpairmentwasdefinedpertestused.Post-sepsiscognitiveimpairmentwasobservedin12.5to 21%ofsurvivorsofsepsis.Attention,cognitiveflexibility,processingspeed,associativelearning,visual perception,workmemory,verbalmemory,andsemanticmemorywerethespecificdomainsaffected. Depressivesymptoms,centralnervoussysteminfection,lengthofhospitalisationduetoinfection,and temporalproximitytothelastperiodofinfectionwereassociatedwithcognitiveimpairment.
Conclusion:Thestudiesareheterogeneous,andthereisurgentneedforacommonlanguage,
includ-ingdefinitionsandneuropsychologicaltests,fortheinvestigationofpost-sepsiscognitiveimpairment. Despitethis,thereismountingevidencefortheclinicalrelevanceofpost-sepsiscognitiveimpairment.
Systematicreviewregistration:PROSPEROCRD42017054583(www.crd.york.ac.uk/PROSPERO).
©2017AustralianCollegeofCriticalCareNursesLtd.PublishedbyElsevierLtd.Allrightsreserved.
1. Introduction
Sepsis isa systemic inflammatory response of thehostto a pathogenicmicroorganism.Sepsisisamajorhealthproblem,with datademonstratinganincreaseinincidencerate1–4andmortality incertaingroups.5Arecentglobalstudyshowedthatonethirdof patientswithsepsisdiebeforeleavingthehospital.6Furthermore,
∗ Correspondingauthor.Permanentaddress:EscoladeMedicina,Universidade FederaldeOuroPreto,CampusMorrodoCruzeiro,OuroPreto,MGCEP35400-000, Brazil. Fax: +55 3135591001.
E-mailaddress:[email protected](A.J.C. Calsavara).
accordingtotheGlobalSepsisAlliance,atleast20%ofsurvivorsof sepsishavesomeformofsequelae,7suchasphysicalorcognitive impairment,mooddisorders,andpoorqualityoflife.8
Cognitive dysfunctionin patientswho survivesepsis can be characterized by new deficits (or exacerbations of preexisting mild deficits) in global cognitionor executive function.9 Septic associatedencephalopathy(SAE)maybedefinedasacognitive dys-functionassociatedwithsepsis,withoutthepresenceofinfection, inthecentralnervoussystem(CNS)orstructuralbraininjuryafter excludingmetaboliccauses.SAEmaybeacute,subacute,orchronic. SAEmanifestedonlyduringthecourseofthediseasewith improve-mentafteritscontrolcanbeclassifiedasacute.10Symptomsthat
http://dx.doi.org/10.1016/j.aucc.2017.06.001
Pleasecitethisarticleinpressas:CalsavaraAJC,etal.Post-sepsiscognitiveimpairmentandassociatedriskfactors:Asystematicreview. AustCritCare(2017),http://dx.doi.org/10.1016/j.aucc.2017.06.001
ARTICLE IN PRESS
G Model
AUCC-376; No.ofPages11
2 A.J.C.Calsavaraetal./AustralianCriticalCarexxx(2017)xxx–xxx
lastweekstomonthscanbeconsideredsubacute,whilesymptoms thatpersistoverayeararecategorizedaschronic.Subacuteand chronicdeficitselicit substantialinterestas affectedindividuals mayrequirerehabilitationorhomecare.
Recently,therehasbeenaprogressiveincreaseofstudies evalu-atingcognitivechangesassociatedwithsepsis.Duetothepotential negativeimpactofsuchamendments,thereisgreatinterestfrom thescientificcommunity,healthmanagers,andpatients’families onthesubject.Conductingasystematicreviewonthetopiccould summarizethestudiescarriedoutsofar,providethereaderwith thebestavailableevidence,andindicatethegapsthatstillexist.We conductedasystematicreviewtodeterminecognitiveimpairment anditsassociatedriskfactorsamongpatientswhosurvivedsepsis.
2. Methods
2.1. Typesofparticipants
Individuals who were 18 years or older with post-sepsis cognitive impairment were included in this study. Post-sepsis referstoanytime afterthediagnosisofsepsis. Sepsisis a life-threatening organ dysfunction caused by a dysregulated host responsetoinfection.11Patientswithhumanimmunodeficiency virus(HIV)infectionwereexcludedasimmunosuppressionanduse ofantiretroviraldrugscouldhaveinfluencedthecourseofsepsis andcognitionoftheseindividuals.12,13Individualsunder18were alsoexcludedduetoneurodevelopmentalfeaturesthatmustbe takenintoconsiderationwhenstudyingchildrenandadolescents.
2.2. Studyselectioncriteria
Publicationsselectedforreviewwerecase-control,cohort,and clinicaltrialsstudieswritteninEnglish,Spanish,orPortuguese. Tobeincluded,studiesneededtohaveassessedtheassociation betweencognitivedysfunctionandsepsisand/oratleastone poten-tialriskfactorfortheoccurrenceofSAE.Therewasnorestriction regardingdateofpublicationandtimeoffollow-upofeachstudy. Studiesthatreportedsubjectivemeasuresofcognitiveoutcome, suchastheopinionsofthestaffregardingthecognitivestateof patientswereexcluded.Singlecasereports,unpublishedstudies, scientificmeetingabstracts,reviewstudies,commentsandletters totheeditorwereexcluded.
2.3. Outcomemeasures
Theprimaryoutcomemeasureforthisreviewwasthecognitive performanceofpost-sepsisindividualsasmeasuredbycognitive tests,scales,orbatteriessuchas,butnotlimitedto,theWechsler AdultIntelligenceScale,Boston NamingTest,WechslerMemory Scale,TrailMakingTest(TMT),InformantQuestionnaireon Cogni-tiveDeclineintheElderly,andMini-MentalStateExamination.
2.4. Searchstrategy
The search was conducted in the databases MEDLINE (1966–present)andEMBASE(1988–present)(seeAppendixA, Sup-plementarydata). ThesearchwasfirstconductedonMay10th, 2015,andwasupdatedonOctober17th,2015,andonMarch20th, 2017.Theinformationthatcouldnotbeextractedbyreadingthe articleswasrequestedbye-mailfromthecorrespondingauthor.
2.5. Studyselection
Twoinvestigatorsindependentlyreviewed thesearchresults toidentifyrelevantstudies.Disagreementswereresolvedby con-sensus,andifnecessaryathirdinvestigatorwasconsulted.First, studieswereexcludedbasedonthetitle;titlesthatwerenotrelated tothesubjectsepsisorcognitionwereexcluded.Inthenextstage,
studieswereevaluatedbyreadingtheabstracts;studiesthatdid notexaminecognitioninthecontextofsepsiswereexcluded.After readingtheabstracts,selectedstudieswerethoroughlyread;atthis laststage,studiesthatmetalltheeligibilitycriteriadescribedabove wereincludedinthereview.TheNewcastle-Ottawaformassignsa maximumoffourpointsforselection,twopointsforcomparability andthreepointsforexposureoroutcome.Inthecurrentstudy,we consideredastudyawardedsevenormorepointsasahigh-quality study.14
2.6. Dataextractionprocessandliteraturequalityassessment
WedevelopedadataextractiontablebasedontheCochrane template.15 One investigator (A.J.C.C) extracted the data and a second(A.L.T.)verifiedtheextracteddata.Inaddition,two inves-tigators(A.J.C.C.andA.L.T.)independentlycross-checkedtherisk ofbiasusingtheNewcastle-OttawaScaleforobservationalstudies. Anydisagreementbetweeninvestigatorswasresolvedby consen-sus,andifnecessaryathirdinvestigator(V.N.)wasconsulted.
2.7. Dataitems
Thefollowinginformationwastakenfromeachselectedstudy: (1)countrywherethestudywasconducted,(2)typeofstudy,(3) follow-upperiod,(4)samplesize,(5)characteristicsofthestudy population(meanage,sex,eligibilitycriteria,classificationsused, anddatacollectionlocation),(6)primaryandsecondaryoutcome, (7)cognitiveoutcomeevaluated,and(8)mainresultsofthestudy. Otherdatasuchas(1)thedefinitionofsepsisused,(2)definition ofcognitiveimpairmentused,(3)cognitiveimpairmentassociated withsepsisfound,and(4)riskfactorsassociatedwithpost-sepsis cognitiveimpairmentwerealsoextracted.
Ingeneral,studiesthathavenotincorporatedmultivariate tech-niquestoidentifyriskfactorshavelittlevalue.Forthepurposesof thissynthesis,onlyvariablesforwhichatleastonestudyreported eitherariskratio(RR)orodds ratio(OR)(regardlessof statisti-calsignificancereported)orastatisticallysignificantassociation (regardlessoftheOR/RRreported)wereconsidered.
3. Results
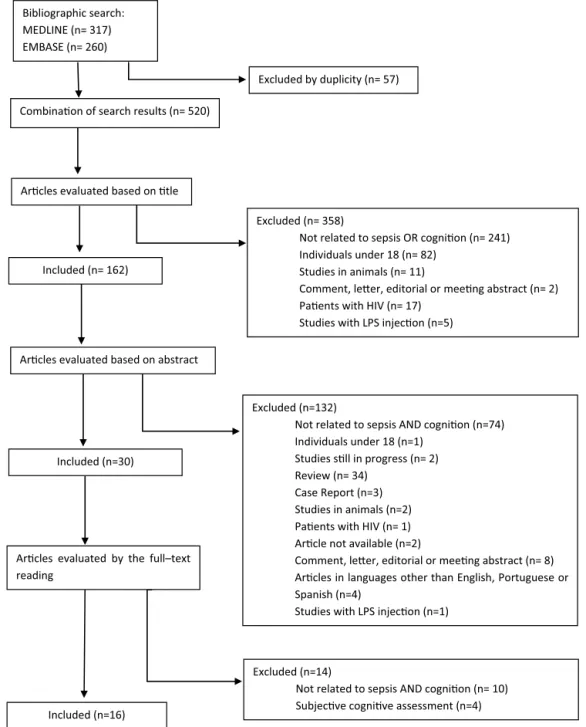
3.1. Descriptionofstudies
ThesearchresultsarepresentedinFig.1.Fivehundred seventy-sevenpotentiallyrelevantarticleswereidentified.Afterexclusion of57duplicatestudies,byjudgingthetitleandabstract,490articles wereexcluded,astheydidnotmeettheinclusioncriteria.Thirty articleswereretainedforin-depthinspection.Sixteenarticleswere retainedfor thissystematic review.Therewas100% agreement betweenreviewersintheselectionofarticlesthatmettheinclusion criteriaofthestudy.
3.2. Characteristicsofincludedstudies
Thesixteenselectedstudiescomprisedsixprospectivecohort withcontrolgroup,8,16–20sixprospectivecohortwithoutcontrol group,21–26threeretrospectivecohortwithoutcontrolgroup,27–29 andonecase-controlstudy.30Noclinicaltrialsmettheinclusion criteria.
Please cite this article in press as: Calsavara AJC, et al. Post-sepsis cognitive impairment and associated risk factors: A systematic review. Aust Crit Care (2017), http://dx.doi.org/10.1016/j.aucc.2017.06.001
AR
TICLE IN PRESS
G Model
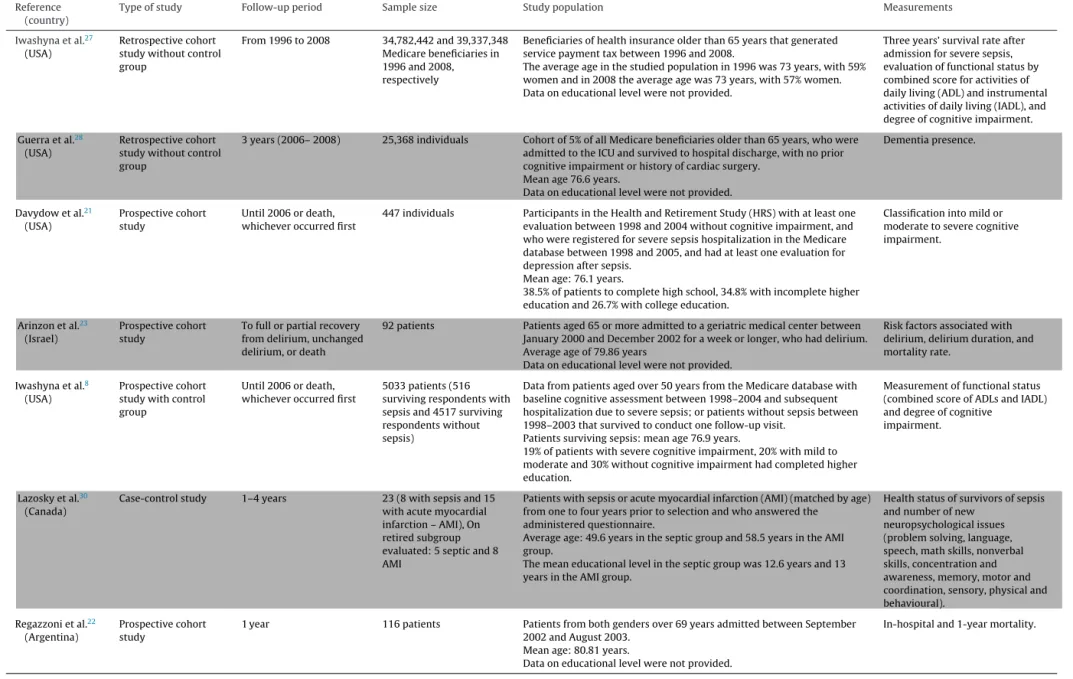
AUCC-376; No. of Pages 11 A.J.C. Calsavara et al. / Australian Critical Care xxx (2017) xxx–xxx 3 Table1Characteristics of studies included.
Reference (country)
Typeofstudy Follow-upperiod Samplesize Studypopulation Measurements
Götzetal.20
(Germany)
Prospectivecohort study with control group
Severesepsisorseptic shock: until 10–15 months after ICU discharge Cirrhosis: not followed up Healthy:until12months afterhospitaldischarge
Severesepsisorseptic Shock: 36
Cirrhosis: 24 Healthy: 23
Patientssurvivedseveresepsisorsepticshock,patientswithcirrhosisand healthy individuals discharged from a University Hospital.
Mean age: severe sepsis or septic shock: 58.9 ±2, cirrhosis: 55.4 ± 6,
healthy: 58 ± 2.
Theauthorsdidnotindicatethemeaneducationallevel.
Visualevokedresponsesusinga set of familiar vs. unfamiliar pictures measured with magnetoencephalography.
Pierrakos et al.26
(Belgium)
Prospective cohort study
1 year 28 patients with sepsis, 14 withcognitivedecline(CD) and14withoutCD
Consecutive patients with sepsis discharged from the ICU between January 2013andJanuary2014.
Meanage:CD:69±15,No-CD:65±16.
Didnotprovidedataoneducationallevel.
Pulsatility index and cerebral blood flowindexmeasurebytranscranial DopplerinpatientswithCDand without-CD.
Götzetal.19
(Germany)
Prospectivecohort studywithcontrol group
Severesepsisorseptic shock:until10–15months afterICUdischarge Healthy:until12months afterhospitaldischarge
Severesepsisorseptic Shock:36
Healthy:30
Patientsthathadsurvivedseveresepsisorsepticshock,andhealthy individualsdischargedfromaUniversityHospital.
Meanage:severesepsisorsepticshock:58.9±2,healthy:50.9±3.
Theauthorsdidnotindicatethemeaneducationallevel.
Peakrestingactivity(frequency andpower)measuredby magnetoencephalography.
Azabouetal.25
(France)
Prospectivecohort study
Untilday28of
hospitalisationoruntilICU discharge
110patientswithsepsis included(45septicshock, 37severesepsisand28 sepsis)
PatientsadmittedtotheICUforsepsisbetweenNovember2011andApril 2014.
Meanagewas63.8years.
The authors did not indicate the mean educational level.
ICUmortalityandrelationship betweenEEGabnormalitiesand occurrenceofdelirium.
Benrosetal.29
(Denmark)
Retrospectivecohort Frombirthtoage19 161,696individuals CognitiveabilitydatafromDanishConscriptionRegistryofmenover18, borninDenmarkbetween1976and1994.
Mean age of patients was 19.4 years. Mean educational level was not provided.
Cognitiveperformanceusingthe DanishConscriptionRegistry database.
Pierrakosetal.24
(Belgium)
Prospectivecohort study
4months 38patients Patientsdiagnosedwithsepsiswithin24-hfromonset.
Mean age of patients with pulsatility index (PI) <1.29 was 62 years and PI >1.3 was 72 years.
Data on educational level was not provided.
PulsatilityIndexmeasuredwith transcranial Doppler in patients with and without SAE.
Lahariya et al.18
(India)
Prospective cohort study with control group
Until occurrence of delirium or ICU discharge
309 patients (81 with delirium and 228 without delirium)
Patients admitted to the cardiology ICU between May and June 2010 who had delirium during hospitalisation.
Mean age in the group with delirium was 61.69 and 77.01 in the group withoutdelirium.
Meaneducationallevelinthegroupwithdeliriumwas7.87yearsandin thegroupwithoutdelirium9.7years.
Incidence and prevalence of delirium in the population studied, risk factors, and mortality associatedwithdelirium.
Merlietal.16(Italy) Prospectivecohort
studywithcontrol group
Threemonthsafter dischargeforcirrhotic patientsandcontrolswith cognitivechangesduring the infectious condition
231(150patientswith cirrhosisand81patients withoutliverdisease)
PatientswithcirrhosishospitalizedbetweenOctober2008andJune2009 (meanage63.4years)andpatientswithoutcirrhosis,betweenJanuary 2010andDecember2010(meanage58years).Eachgroupwasdivided into3subgroups:withoutsepsisorinfection,withinfectionwithout systemic inflammatory response syndrome, and with sepsis. The mean educational level was not indicated by the authors.
Measurementofpsychometric parameters.
Semmleretal.17
(Germany)
Prospectivecohort studyintwocenters withcontrolgroup
From6to24monthsafter ICUdischarge
44patients(25with sepsis/severesepsisand19 withoutsepsis)
PatientsadmittedtotheICUbetweenJanuary2004andAugust2006with sepsisorseveresepsis.
Meanageofpatientswithsepsis:55.64yearsandnon-septic:52.15years. Dataoneducationallevelwerenotprovided.
Please
cite
this
article
in
press
as:
Calsavara
AJC,
et
al.
Post-sepsis
cognitive
impairment
and
associated
risk
factors:
A
systematic
review.
Aust
Crit
Care
(2017),
http://dx.doi.org/10.1016/j.aucc.2017.06.001
AR
TICLE IN PRESS
G Model
AUCC-376;
No.
of
Pages
11
4
A.J.C.
Calsavara
et
al.
/
Australian
Critical
Care
xxx
(2017)
xxx–xxx
Table 1 (Continued)
Reference (country)
Typeofstudy Follow-upperiod Samplesize Studypopulation Measurements
Iwashynaetal.27
(USA)
Retrospectivecohort studywithoutcontrol group
From1996to2008 34,782,442and39,337,348 Medicarebeneficiariesin 1996and2008, respectively
Beneficiariesofhealthinsuranceolderthan65yearsthatgenerated servicepaymenttaxbetween1996and2008.
Theaverageageinthestudiedpopulationin1996was73years,with59% women and in 2008 the average age was 73 years, with 57% women. Data on educational level were not provided.
Threeyears’survivalrateafter admissionforseveresepsis, evaluationoffunctionalstatusby combined score for activities of daily living (ADL) and instrumental activities of daily living (IADL), and degreeofcognitiveimpairment.
Guerra et al.28
(USA)
Retrospective cohort study without control group
3 years (2006– 2008) 25,368 individuals Cohort of 5% of all Medicare beneficiaries older than 65 years, who were admitted to the ICU and survived to hospital discharge, with no prior cognitiveimpairmentorhistoryofcardiacsurgery.
Meanage76.6years.
Dataoneducationallevelwerenotprovided.
Dementia presence.
Davydowetal.21
(USA)
Prospectivecohort study
Until2006ordeath, whicheveroccurredfirst
447individuals ParticipantsintheHealthandRetirementStudy(HRS)withatleastone evaluationbetween1998and2004withoutcognitiveimpairment,and whowereregisteredforseveresepsishospitalizationintheMedicare databasebetween1998and2005,andhadatleastoneevaluationfor depression after sepsis.
Mean age: 76.1 years.
38.5% of patients to complete high school, 34.8% with incomplete higher educationand26.7%withcollegeeducation.
Classificationintomildor moderatetoseverecognitive impairment.
Arinzon et al.23
(Israel)
Prospective cohort study
To full or partial recovery from delirium, unchanged delirium,ordeath
92 patients Patients aged 65 or more admitted to a geriatric medical center between January 2000 and December 2002 for a week or longer, who had delirium. Averageageof79.86years
Dataoneducationallevelwerenotprovided.
Risk factors associated with delirium, delirium duration, and mortalityrate.
Iwashyna et al.8
(USA)
Prospective cohort studywithcontrol group
Until 2006 or death, whicheveroccurredfirst
5033 patients (516 survivingrespondentswith sepsisand4517surviving respondentswithout sepsis)
Data from patients aged over 50 years from the Medicare database with baselinecognitiveassessmentbetween1998–2004andsubsequent hospitalizationduetoseveresepsis;orpatientswithoutsepsisbetween 1998–2003thatsurvivedtoconductonefollow-upvisit.
Patientssurvivingsepsis:meanage76.9years.
19% of patients with severe cognitive impairment, 20% with mild to moderate and 30% without cognitive impairment had completed higher education.
Measurement of functional status (combinedscoreofADLsandIADL) anddegreeofcognitive
impairment.
Lazosky et al.30
(Canada)
Case-control study 1–4 years 23 (8 with sepsis and 15 with acute myocardial infarction – AMI), On retiredsubgroup evaluated:5septicand8 AMI
Patients with sepsis or acute myocardial infarction (AMI) (matched by age) from one to four years prior to selection and who answered the
administered questionnaire.
Averageage:49.6yearsinthesepticgroupand58.5yearsintheAMI group.
Themeaneducationallevelinthesepticgroupwas12.6yearsand13 yearsintheAMIgroup.
Health status of survivors of sepsis and number of new
neuropsychological issues (problemsolving,language, speech,mathskills,nonverbal skills,concentrationand awareness,memory,motorand coordination, sensory, physical and behavioural).
Regazzonietal.22
(Argentina)
Prospectivecohort study
1year 116patients Patientsfrombothgendersover69yearsadmittedbetweenSeptember 2002 and August 2003.
Mean age: 80.81 years.
Data on educational level were not provided.
Pleasecitethisarticleinpressas:CalsavaraAJC,etal.Post-sepsiscognitiveimpairmentandassociatedriskfactors:Asystematicreview. AustCritCare(2017),http://dx.doi.org/10.1016/j.aucc.2017.06.001
ARTICLE IN PRESS
G Model
AUCC-376; No.ofPages11
A.J.C.Calsavaraetal./AustralianCriticalCarexxx(2017)xxx–xxx 5
Bibliographic search: MEDLINE (n= 317) EMBASE (n= 260)
Excluded by duplicity (n= 57)
Arcles evaluated based on tle Combinaon of search results (n= 520)
Excluded (n=358)
Not related to sepsis OR cognion (n= 241) Individuals under 18 (n= 82)
Studies in animals (n= 11)
Comment, leer, editorial or meeng abstract (n= 2) Paents with HIV (n= 17)
Studies with LPS injecon (n=5) Included (n= 162)
Arcles evaluated based on abstract
Excluded (n=132)
Not related to sepsis AND cognion (n=74) Individuals under 18 (n=1)
Studies sll in progress (n= 2) Review (n= 34)
Case Report (n=3) Studies in animals (n=2) Paents with HIV (n= 1) Arcle not available (n=2)
Comment, leer, editorial or meeng abstract (n= 8) Arcles in languages other than English, Portuguese or Spanish (n=4)
Studies with LPS injecon (n=1) Included (n=30)
Arcles evaluated bythe full–text reading
Excluded (n=14)
Not related to sepsis AND cognion (n= 10) Subjecve cognive assessment (n=4) Included (n=16)
Fig.1. Searching results.
Whileconsideringformaleducationdata,onlyfourstudies indi-catedtheaveragelevelofeducationofpatients.8,18,21,30Twostudies indicatedtheaverageschoolingoftheirpatientsas7.918and12.6 years.30 The others referred tothe patients’ educationlevel by categorizingthemintohavinggraduatedornotfromSecondary educationandhavinggraduatedornotfromHighereducation8,21 (Table1).
Thequalityofthestudieswasassessedwiththe Newcastle-OttawaScoreandtheresultsarepresentedinTables2and3.Four studieswereclassifiedashighquality.8,17,21,29Themeanvaluefor thesixteenstudiesassessedwas5.3indicatingalowoverallquality.
3.3. Definitionofsepsis,severesepsis,andsepticshock
There was variation in the definition of sepsis, severe sep-sis,and septicshock among the selected studies.The majority of studies used as reference the criteria of the American
College of Chest Physicians/Society of Critical Care Medicine (ACCP/SCCM).8,16,17,19–22,24,25,27,28,30OthersusedtheInternational StatisticalClassificationofDiseases(ICD-8and/orICD-10).29,31
NoreferencewasmadetothedefinitionofsepsisbyLahariya etal.18,Arinzonetal.23andPierrakosetal.26
3.4. Definitionofcognitiveimpairmentpatients
Pleasecitethisarticleinpressas:CalsavaraAJC,etal.Post-sepsiscognitiveimpairmentandassociatedriskfactors:Asystematicreview. AustCritCare(2017),http://dx.doi.org/10.1016/j.aucc.2017.06.001
ARTICLE IN PRESS
G Model
AUCC-376; No.ofPages11
6 A.J.C.Calsavaraetal./AustralianCriticalCarexxx(2017)xxx–xxx
Table2
Newcastle-Ottawaqualityassessmentscaleforcohortstudiesincludedinthisreview.
Firstauthor Year Selectiona
Compara-bilityb
Outcomea Overall
qualityscorec
Representa-tivenessof theexposed cohort
Selectionof the non-exposed cohort
Ascertainment ofexposure
Outcomeof interestnot presentat start of study
Assessment ofoutcome
Adequacyof durationof follow-up
Adequacyof complete-nessof follow-up
Götz20 2017 ⋆ 0 ⋆ 0 ⋆⋆ ⋆ ⋆ 0 6
Götz19 2016 ⋆ 0 ⋆ 0 ⋆⋆ ⋆ ⋆ 0 6
Pierrakos26 2016 ⋆ ⋆ ⋆ 0 ⋆ ⋆ 0 0 5
Azabou25 2015 ⋆ 0 ⋆ 0 0 ⋆ ⋆ 0 4
Benros29 2015 ⋆ ⋆ ⋆ 0 ⋆ ⋆ ⋆ ⋆ 7
Pierrakos24 2014 ⋆ 0 ⋆ 0 ⋆ ⋆ 0 0 4
Lahariya18 2014 ⋆ ⋆ 0 0 0 ⋆ 0 ⋆ 4
Merli16 2013 ⋆ ⋆ ⋆ 0 0 ⋆ ⋆ 0 5
Semmler17 2013 ⋆ ⋆ ⋆ 0 ⋆⋆ ⋆ ⋆ ⋆ 8
Iwashyna27 2012 ⋆ 0 ⋆ 0 0 ⋆ 0 0 3
Guerra28 2012 ⋆ 0 ⋆ ⋆ 0 ⋆ ⋆ ⋆ 6
Davydow21 2012 ⋆ 0 ⋆ ⋆ ⋆⋆ ⋆ ⋆ 0 7
Arinzon23 2011 ⋆ 0 0 0 0 ⋆ 0 0 2
Iwashyna8 2010 ⋆ ⋆ ⋆ 0 ⋆⋆ ⋆ ⋆ 0 7
Regazzoni22 2008 ⋆ 0 ⋆ ⋆ ⋆ ⋆ ⋆ 0 6
Mean 5.3
aAstudycanbeawardedamaximumofonestarforeachnumbereditemwithintheSelectionandOutcomecategories. b AmaximumoftwostarscanbegivenforComparability.
c Maximumof9points.
Table3
Newcastle-Ottawaqualityassessmentscaleforcasecontrolstudiesincludedinthisreview.
Firstauthor Year Selectiona
Compara-bilityb
Exposurea Overall
quality scorec
Isthecase definition adequate?
Representa-tivenessof the cases
Selectionof controls
Definitionof controls
Ascertainment ofexposure
Samemethod of ascertain-ment for cases and controls
Non-response rate
Lazosky30 2010 ⋆ 0 0 ⋆ ⋆ ⋆ ⋆ 0 5
aAstudycanbeawardedamaximumofonestarforeachnumbereditemwithintheSelectionandExposurecategories. b AmaximumoftwostarscanbegivenforComparability.
c Maximum of 9 points.
AdiagnosisofcognitiveimpairmentwasmadebyMerlietal.16 withtheTrailMakingTestA,BoraDigit-SymbolTestZ-score33 greaterthantwostandarddeviationsfromthemeanofahealthy Italianpopulation,adjustedforageandeducation.34
Semmleretal.17calculatedacompositeZ-scoreforcognition usingtheNeuroCogFX,Trail-makingTestsAand BandMultiple ChoiceWordTest-B.Thescoreconsideredtheresultsofapplied cognitivetestsaftersepsis(NeuroCogFXandTrail-makingTestsA andB),andMultipleChoiceWordTest-BGermanvocabularytest, whichaimistoquantifythepatients’premorbidcognitivestatus. AZ-scorebetween−1.5and−2.0wasconsidereddenotingmild cognitiveimpairment.
Otherstrategiestodefinecognitiveimpairmentweretheuse ofthe“ConfusionAssessmentMethodintheIntensiveCareUnit” (CAM-ICU),18,23–26anMMSEscorelessthan24,26GlasgowComa Score<8despiteresolutionofsepsisafterexclusionofany under-liningpathologythatcouldexplainthis state,26 abnormalClock DrawingTest,26orsearchingpredeterminedInternational Classifi-cationofDiseases(ICD-9-CM)codes35indatabases.28
Althoughmoststudies didnotstate this, alltools described in this section toassess cognitive impairment had been previ-ouslyvalidated.36–45 The neurocognitive outcomes reported by eachauthorwereassessedandarepresentedinTable5.
Somestudiesdidnotexplaintheircategorizationmethodsor cutoffpointsusedtodefinecognitiveimpairment.19,20,22,29,30
3.5. Post-sepsisglobalcognitiveimpairment
Analysingtheselected studiesinthis review, wefoundthat Iwashynaetal.27showedmoderate toseverecognitive impair-mentafter3yearsin16.7%(95%CI:12.3,21.0%)ofpatientswho survivedsepsis.Thesameauthorhadfoundanoddsratio(OR)of 3.34formoderatetoseverecognitiveimpairmentaftersepsisina studypublishedtwoyearsearlier.8InastudybyDavydowetal.,21 cognitiveimpairmentrangedfrom14to20%,with60%of individu-alsbeingclassifiedasmoderatelytoseverelyimpaired.Itwasalso shownbyBenrosetal.29thathospitalcontactduetoanyinfectious conditionworsenedglobalcognitiveperformance,assessedbya DanishIntelligenceTestby76%(95%confidenceinterval,61–92%), sepsiswasassociatedwithameanscoreof1.60-unitslower cogni-tiveability.
3.6. Post-sepsisspecificcognitivedomainsimpairment
Please
cite
this
article
in
press
as:
Calsavara
AJC,
et
al.
Post-sepsis
cognitive
impairment
and
associated
risk
factors:
A
systematic
review.
Aust
Crit
Care
(2017),
http://dx.doi.org/10.1016/j.aucc.2017.06.001
AR
TICLE IN PRESS
G Model
AUCC-376;
No.
of
Pages
11
A.J.C.
Calsavara
et
al.
/
Australian
Critical
Care
xxx
(2017)
xxx–xxx
7
Table4
Definitions,changes,andriskfactorsassociatedwithpost-sepsiscognitiveimpairment.
Reference Definitionofcognitiveimpairment Cognitiveimpairmentassociatedwithsepsis found
Riskfactorsassociatedwithcognitiveimpairment
Götzetal.20 ThecutoffpointsfortheDemTectandClockDrawingTest
(CDT)werenotspecified.
Notassessed ImpairedresponsetoperiodicvisualstimulationwasassociatedwithDemTect andCDTsumscoresinpatientswithsepsis.Non-affectingfactors:APACHEII, CRP,lengthofstayintheICU,andmonthsafterICUdischarge.
Pierrakosetal.26 Persistentcoma(GlasgowComaScore<8),despite
resolutionofsepsisandafterexclusionofanyunderlying pathologythatcouldexplainthisstate,positiveCAM-ICU ondischarge,MMSE<24,abnormalClockDrawingTest.
Notassessed Univariateanalysis:deliriumandpulsatilityindexonfirstdaywereassociated withcognitivedecline(CD).Multivariateanalysis:onlydeliriumwas associatedwithCD.
Götzetal.19 ThecutoffpointsforDemTectandCDTwerenotspecified. Notassessed WithinsepsisgrouppeakrestingfrequencyincreasewithtimeafterICU
dischargeandwithDemTectandCDTsumscoreanddecreasewithage. Non-affectingfactorsAPACHEII,CRP,andlengthofstayintheICU.
Azabouetal.25 DeliriumwasdefinedbytheCAM-ICUandcomawas
definedinnon-sedatedpatientsbyGCS≤8andinsedated
patientsbyRASS≤4.
Notassessed DeliriuminsepticpatientswasassociatedwithSAPS-IIscore,septicshock,and deltafrequencydominant,Synekgrade≥3andYounggrade≥1inearlyEEG.
Benrosetal.29 Nocutoffpointwasgivenforthetestapplied. Infectionwasassociatedwithpooroverall
cognitiveperformanceassessedbytheBørge Prienstest.(OR:−1.76,95%CI:−1.92,−1.61).
Centralnervoussysteminfectionwasassociatedwithgreaterdeclinein cognitiveability.Otherassociatedfactors:numberofvisitstohospitaldueto infection,temporalproximitytothelastinfection,lengthofstay,andfamily historyofinfection.Non-affectingfactors:age,historyofpsychiatricdisorders or substance abuse, history of cancer, and normal weight at birth.
Pierrakosetal.24 CAM-ICUfordelirium. Notassessed NoassociationwasfoundbetweenageorAPACHEIIwithpositiveCAM-ICU.
Lahariyaetal.18 CAM-ICUfordelirium.IQCODEscoregreaterthan
3.31–3.38asanindicationofsuspecteddementia.
Sepsiswasassociatedwiththedevelopmentof deliriumasdiagnosedwiththeCAM-ICU.
PatientswithcognitiveimpairmentmeasuredbytheIQCODEshowed increasedriskofdevelopingdelirium.Otherriskfactorsassociatedwiththe developmentofdelirium:hypokalemia,SOFAscores,using>3drugs, cardiogenicshock,historyofcoronaryarterybypassgrafting,ejectionfraction <30, use of opioids, age >65 years, diabetes, a history of seizures, congestive heart failure, percutaneous transluminal coronary angioplasty, atrial fibrillation, current depression, use of benzodiazepines, warfarin, ranitidine, steroids,orNSAIDs,polypharmacy,increasedcreatinine,anemia,
hypoglycemia,APACHEII,andtheComorbidityIndexofCharlson.
Merli et al.16 Z-score >2 SD of healthy Italian population, adjusted for
ageandeducation.
Patients with cirrhosis and sepsis showed changesintheTrail-MakingTestsAandBand Digit-SymbolTest.Patientswithoutcirrhosis withsepsishadpoorerperformanceonthe TrackTestsandDigitSymbolthanthegroup without infection (39% of septic individuals had subclinical cognitive impairment damage, remaining unchanged).
In the cirrhotic subgroup, the MELD score, albumin, and creatinine were not foundtobeindependentriskfactorsforcognitiveimpairment.Sepsiswasthe onlyriskfactorfound.Riskfactorswerenotevaluatedinthecontrolgroup.
Semmler et al.17 Z-score compared to the historical norm. Composite score
for cognition through the simple average of the
NeuroCogFX and Trail Making Test A and B Z-scores, minus theestimatedpremorbidverbalabilitybytheMultiple ChoicesWordTestZ-score.
Patients with sepsis post-ICU hospitalization had poor performance on the Digit Span test, 2-Back Test, alertness, GoNoGo, verbal memory,andphoneticverbalfluency.Patients withoutsepsispost-ICUhadlowperformance intheDigitSpantest,2-Backtest,interference, andphoneticverbalfluency.
Cognitive deficits were not influenced by the length of stay in the ICU, time after ICU discharge, number of days on mechanical ventilation, APACHE II, and SOFA scores.
Iwashyna et al.27 The cutoff point for m-TICS was not specified. The cutoff
point for IQCODE was 4.59–5.00, denoting moderate to severecognitiveimpairment,accordingtotheauthors.
Hospitalisation for severe sepsis was associated with a 3.34-fold increased risk of developingmoderatetoseverecognitive impairment.
Please
cite
this
article
in
press
as:
Calsavara
AJC,
et
al.
Post-sepsis
cognitive
impairment
and
associated
risk
factors:
A
systematic
review.
Aust
Crit
Care
(2017),
http://dx.doi.org/10.1016/j.aucc.2017.06.001
AR
TICLE IN PRESS
G Model
AUCC-376;
No.
of
Pages
11
8
A.J.C.
Calsavara
et
al.
/
Australian
Critical
Care
xxx
(2017)
xxx–xxx
Table4(Continued)
Reference Definitionofcognitiveimpairment Cognitiveimpairmentassociatedwithsepsis found
Riskfactorsassociatedwithcognitiveimpairment
Guerra et al.28 ICD-9-CM codes: 290.0–290.4, 294.0, 294.1, 294.8, 331.0,
331.1,331.2,331.7,and797.Xrecordedfrom fee-for-serviceclaimsinthesubsequentthreeyearsof follow-up.
Neuropsychological tests were not applied. Risk factors associated with post-ICU dementia: a critical illness with infection,especiallyifseveresepsis,acuteneurologicdysfunctionduringICU stay,anduseofrenalreplacementtherapy(increasedriskonly6monthsafter discharge),patientswithhospitalisationduetoclinicalcausesandpatients withprevioushospitalization.Notassociated:mechanicalventilation,organ dysfunction, length of stay in the ICU and total hospital stay.
Davydowetal.21 Categorisationofcognitivefunctionasmild,andmoderate
tosevereusingthem-TICS.
17%ofpatientswithseveresepsisshowed globalcognitiveimpairmentassessedbythe TICS; most had moderate to severe deficits.
Pre-sepsisdepressivesymptomswereariskfactorforpost-sepsiscognitive impairment.
Arinzonetal.23 CAM-ICUfordiagnosisofdeliriumandDeliriumRating
Scale(ascorehigherthan10–12pointsindicatedpatients with delirium).
Theincreaseinthedurationofdeliriumwas associatedwiththeoccurrenceofsepsis.
Sepsiswasassociatedwiththedurationofdeliriumandincreasedmortalityin patientswithdelirium.
Iwashynaetal.8 Thecutoffpointsforthem-TICSandIQCODEwerenot
specified.
AnORof3.34,95%CI:1.53–7.25wasfoundfor moderatetoseverecognitiveimpairment after-sepsis. No moderate to severe cognitive impairment was found after hospitalisation for reasons other than sepsis.
Notassessed
Lazoskyetal.30 Numberofnewproblemsidentifiedinthe‘Adult
Neuropsychological History form’.
Therewerenocognitivechangesinthe evaluated samples.
None
Regazzonietal.22 Therewasnocategorisationorcutoffpointdefinitionfor
theMMSE.
Globalcognitivestatusevaluatedwiththe MMSEdidnotpredictin-hospitalmortality after sepsis. In a univariate analysis, the MMSE predicted mortality after 1 year; however, in a COXmodelthiseffectwasnotmaintained.
Notassessed(cognitionascovariate)
Table5
Reportedneurocognitiveoutcomesbyauthor.
Reference EEG MEG CAM-ICU GCS MMSE m-TICS IQCODE TMT Digit-Symbol Test
Multiple Choice Word Test-B
Neuro-CogFxa
Auditory Verbal Learning Test
Rey Complex Figure Test
Børge Prien’s Prøveb
Delirium Rating Scale
ICD Adult Neuropsy-chological History form
DemTectc Clock
Drawing Test
Götz et al.20 √ √ √
Pierrakos et al.26 √ √ √ √ √
Götz et al.19 √ √ √
Azabouetal.25 √ √ √
Benrosetal.29 √
Pierrakosetal.24 √
Lahariyaetal.18 √ √
Merli et al.16 √ √ √
Semmler et al.17 √ √ √ √ √
Iwashyna et al.27 √ √
Guerraetal.28 √
Davydowetal.21 √
Arinzonetal.23 √ √ √
Iwashynaetal.8 √ √
Lazosky et al.30 √
Regazzoni et al.22 √ √ √
aNeuroCogFx (Digit Span, 2-back-test, alertness, go-no-go, interference, verbal memory, figural memory, phonetic verbal fluency).
b A Danish intelligence test.
Pleasecitethisarticleinpressas:CalsavaraAJC,etal.Post-sepsiscognitiveimpairmentandassociatedriskfactors:Asystematicreview. AustCritCare(2017),http://dx.doi.org/10.1016/j.aucc.2017.06.001
ARTICLE IN PRESS
G Model
AUCC-376; No.ofPages11
A.J.C.Calsavaraetal./AustralianCriticalCarexxx(2017)xxx–xxx 9
processingspeed,visualperception,andworkingmemoryassessed bytheDigitSymbolTestwasalsolowerinpatientswithsepsis.16
Semmleretal.17foundlongtermmildcognitiveimpairmentsin attention,verbalfluency,executivefunction,andverbalmemoryin survivorsofsepsiswhencomparedtotheirpreviouslyestimated intelligencelevel.
A long-term visual processing impairment in patients with sepsispost-infectionwasobservedbyGötzetal.with magnetoen-cephalographicmeasurements.20
3.7. Associationsbetweensepsisandcognition
Moststudiesassessedtheassociationbetweencognitive impair-mentandsepsis(Table4).8,16,17,21,27,29,30Somestudiesshowedthat sepsisisariskfactorforworseningcognitiveperformanceafterICU discharge28or demonstratedanassociationbetweensepsis and delirium,aclinicalsyndromethatencompassesseveralcognitive changes.18,23–25
Thepresenceofpre-sepsisdepressivesymptomswas consid-eredariskfactorforpost-sepsiscognitiveimpairment.21Infection in the CNS, number of hospital visits due to infection, length of hospitalization due to infection, family history of infection, and temporal proximitytothe latestepisodeof infectionwere identifiedasriskfactorsforcognitiveimpairmentobservedafter infectiousconditions.29Criticalillnessinthepresenceofinfection, especiallyseveresepsis,wasidentifiedasoneoftheriskfactorsfor thesubsequentdiagnosisofdementiawithinthreeyearsofhospital discharge.28
Sepsis was a risk factor for the development18 and longer duration23ofdelirium.Moreover,Arinzonetal.23showedthata diagnosisof sepsisincreasedthemortalityrateinpatientswith delirium.
Severalriskfactorswereevaluatedintheincludedstudies,but manydidnotaffecttheoutcomeevaluated(Table4).Forinstance, itwasfoundthatcognitivedeficitsrelatedtosepsiswerenot influ-encedbythelengthofstayintheICU,ICUdischargetime,number ofdaysonmechanicalventilation,APACHEII,andSOFAscores.17 Thepatient’s age,familyhistory of psychiatricillness,and sub-stanceabusesimilarlydidnotaffectcognitiveperformanceafter infection.29
3.8. Workuponsepsisassociatedencephalopathy
Thediagnosisofsepsis-associated cognitivedysfunctionwas additionallyevaluatedbytheselectedstudies.17,24,25
AnassociationbetweenSAEandelectroencephalography(EEG) parameterswasobserved;astheoccurrenceofcognitive impair-mentinthedaysafterEEGrecordingwasassociatedwithprevious EEGfindings,suchasdeltafrequencydominantEEG,absenceof EEGreactivity,Synekgrade≥3(anEEGclassificationwith prog-nostic valuefor comatose patientthat range from0 to5), and Younggrade≥1(anECGclassificationforsepticencephalopathy thatrangesfrom0to4).25
ThediagnosisofSAEwiththepulsatilityindex,anindicatorof cerebrovascularresistance,measuredbytranscranialDopplerwas evaluated.Apulsatilityindexgreaterthan1.3at24-hpostsepsis diagnosiscorrelatedwithSAE(OR:5.66,95%confidenceinterval: 1.1–29.11).24Althoughnocorrelationbetweencognitive dysfunc-tionaftersepsisandpulsatilityindexwasfound,whenmultivariate analysiswasperformedinanotherstudybythesameauthor.26
Magnetoencephalographic (MEG) techniques were used to studythe long-termbrain electrophysiologychanges after sep-sis.CombiningMEGdatawithneuropsychologicaltests,suggested abnormalthalamocorticaldynamics19andadisruptioninneural networking,especiallycomplexnetworking,inpatientswith long-termpost-sepsis.20
Computerized volumetric techniques of magnetic resonance imagesshowedsignificantdifferencesinleftandtotal hippocam-palvolumesbetweenpatientswithsepsisand healthycontrols. Thehippocampalvolumeinpatientswithoutsepsisadmittedin theICUwasbetweenthatofpatientswithsepsisandhealthy con-trols.Acorrelationanalysisbetweencognitiveperformanceand hippocampalvolumewasnotperformed.17
4. Discussion
Tothebestofourknowledge,thisisthefirstsystematicreview toidentifyand synthesize thebestavailable evidenceon post-sepsis cognitiveimpairment. Theanalysed studies showedthat sepsiscanworsencognitiveperformanceintheshort,medium,and longterms.Theavailablestudiesdidnotproviderobustevidenceto determinetheriskfactorsthatmodulatetheassociationbetween sepsisandcognitiveimpairment.Regardingthegreat heterogene-ityinthedefinitionofcognitiveimpairmentandtheuseofseveral cognitivetests,thecomparisonamongstudieswasimpaired.
Theuseofbatteriesandtestsevaluatingglobalcognitive func-tionwasmostfrequentlyemployed.Thisstrategyisinlinewith the theory that sepsis causes wide brain function alterations. The cognitive test batteries Telephone Interview for Cognitive Status(m-TICS),8,21,27NeuroCogFx,17MMSE,16,22,23,26Clock Draw-ingTest,26AdultNeuropsychologicalHistoryform,30BØrge-Priens test,29IQCODE,8,18,27andDemTect19,20wereusedforglobal cogni-tiveassessment.SometestsastheSymbol-Digittest,16TrailMaking TestAandB,16,17ReyComplexFigureTest17providedmorespecific informationaboutcognitivedomains.Onlyonestudy,by Semm-ler et al.,17 showedchanges in certaincognitivedomains, such asattention,verbalfluency,executivefunction,andverbal mem-ory.Semmler etal.17 proposedthatan asymmetricdistribution ofneurotransmittersinthehumanbrainwouldleadtoincreased vulnerabilityofthelefthemispheretoinflammatoryinsults.
Inacohortstudyperformedwithpatientswithcriticalillness byPandharipandeetal.9thecognitivesequelaerangedbetween24 to40%dependingonthetimingofcognitiveevaluation.Thisstudy usedtheRepeatableBatteryfortheAssessmentof Neuropsycho-logicalStatus(RBANS)thatwasnotusedinthestudiesincludedin thisreview.Asshown,ouranalysissuggeststhatequallyimportant totimingisthechoiceoftestingmeans,forcognitiveevaluation. Tocomparetheincidenceandprevalenceofcognitivedysfunction betweenpatientswithsepsisandpatientswithotherdiseasesit isnecessarytoachieveaconsensusontheappropriatedtesttobe used,beforehand.
Mostoftheevaluatedstudieswereconductedwithpatientsover theageof60years.8,18,21–28Despitesepsisincreasedincidenceand mortalityintheelderlycomparedtoyoungerpatients,46itis impor-tanttoevaluatetheimpactof sepsisonyoungindividualswho arestillpartoftheworkforce.Whereascognitivereservehasbeen identifiedasaprotectivefactorforcognitiveimpairmentinseveral studies,47–49thepossibilityofyoungpatientsexhibitinglesspost sepsisdeficitscouldbeexplainedatleastinpartbytheirhigher cognitivereserve.
Pleasecitethisarticleinpressas:CalsavaraAJC,etal.Post-sepsiscognitiveimpairmentandassociatedriskfactors:Asystematicreview. AustCritCare(2017),http://dx.doi.org/10.1016/j.aucc.2017.06.001
ARTICLE IN PRESS
G Model
AUCC-376; No.ofPages11
10 A.J.C.Calsavaraetal./AustralianCriticalCarexxx(2017)xxx–xxx
developed,and,forthefirsttime,therecommendedprimaryICD-9 andICD-10codesandaframeworkforimplementationfor cod-ingandresearchinanattempttofacilitateresearchandincrease accuracyofcoding.11
Thecriteriausedtoclassifyindividualsaspresentingwith cog-nitiveimpairmentalsodifferedamongstudies,evenwhenusing thesame cognitivetests.The choiceof cut-offsdepends onthe authors’choicebetweenhigherspecificityandhighersensitivity. Whenthegoalis toidentifyindividuals withcognitive impair-ment,testswithgreatersensitivity arepreferred,butwhenthe objectiveistodeterminetheexactnatureofthepatient’sdeficit, specifictestsarepreferable.Amethodtocomparetheperformance ofteststhatmeasuredifferentcognitiveabilitiesisthrough stan-dardizedscores,suchastheZ-score. Onlytwostudiesreported cognitivedatathroughstandardizedscores,16,17 theirscarceuse maybeduetolackofgoodqualitystandardizeddataorevenlack ofsufficientdatainthestudiedpopulation,especiallyamongethnic minorities.50
Wedidnotlocatestudieswithadesignthatrobustlyassessed theriskfactors associatedwiththedevelopmentof post-sepsis cognitiveimpairment.Factorssuchaspre-sepsisdepressive symp-toms,infectionintheCNS,lengthofstay,andtemporalproximity tothelatestepisodeofinfectionseemtobeinvolvedincognitive impairment associated withsepsis. Studieswith patients post-ICUhospitalizationhavesuggestedtheinfluenceofsomefactors oncognition,suchassedativesandanalgesics,51antipsychotics,52 antibiotics,53glycemiccontrol,54corticosteroids,55andcognitive reserve.56Theinfluenceofthesefactorsmustbebetterinvestigated infuturestudies.
InastudypublishedbyLarsonetal.,56cognitivereserve associ-atedwiththemagnitudeofcognitiveimpairmentinpatientswith acuterespiratorydistresssyndrome.Inthissystematicreview,the issuewasnotexplored.InthestudyofSemmleretal.,17although theauthor estimatedcognitivereservewithneuropsychological testing, a correlational analysis between cognitive reserve and post-sepsiscognitiveperformance wasnot performed.Lahariya etal.18usedtheIQCODEscoretoevaluatethepre-morbidity cog-nitivereserveinasample,findingthatpatientswithlowcognitive reserve(i.e. IQCODE>3.3)had an odds ratioof 10.81for delir-ium.
Ourstudyhadanumberoflimitations.Thesearchconducted didnotinvolveacomprehensivelistofsearchterms.The hetero-geneityofthetestsandcutoffpointsusedinthestudiesanalyzed didnotprovidethenecessarydataforperformingameta-analysis. Therewasnoconsensusamongtheauthorsregardingthebesttests toevaluatepost-sepsiscognitivechanges.Thelimitednumberof availablestudies,themarkeddifferencesamongthepopulations studied,andthediversityofoutcomescomplicatedthe general-isabilityof thestudyfindings. Thequalityof thestudiesvaried widely.
Ourreviewrevealedseveralgapsintheaccumulatedknowledge onpost-sepsiscognitiveimpairment.We observedthatthere is needforstudiestoassessthemostappropriateneuropsychological testsandtocompareglobalcognitiveassessmentteststodomain specifictests(e.g.attentionandmemory).Furthermore,the assess-mentofpost-sepsiscognitiveimpairmentinsubgroupsofyoung andelderlypatientscouldprovidecluesabouttheroleofcognitive reserveonpost-sepsiscognitiveimpairment.Studieswith cogni-tiveimpairmentbiomarkers(e.g.neurotrophic,neurodegenerative, inflammatory,andoxidativestressbiomarkers)andneuroimaging havealsobeenperceivedasmissingorsparse.
Inconclusion,thissystematicreviewdemonstratesthatthere isincreasingevidencefor theexistenceofpost-sepsiscognitive impairment.Duetolackofconsistentfindings,thesamecannot bestatedaboutitsassociatedriskfactors.
Funding
Thisresearchdidnotreceiveanyspecificgrantfromfunding agenciesofthepublic,commercial,ornot-for-profitsectors.
AppendixA. Supplementarydata
Supplementarydataassociatedwiththisarticlecanbefound, intheonlineversion,athttp://dx.doi.org/10.1016/j.aucc.2017.06. 001.
References
1.KaukonenKM,BaileyM,SuzukiS,PilcherD,BellomoR.Mortalityrelatedto severesepsisandsepticshockamongcriticallyillpatientsinAustraliaandNew Zealand,2000–2012.JAMA2014;311:1308–16.
2.ShenHN,LuCL,YangHH.EpidemiologictrendofseveresepsisinTaiwanfrom 1997through2006.Chest2010;138:298–304.
3.LaguT,RothbergMB,ShiehMS,PekowPS,SteingrubJS,LindenauerPK. Hospi-talizations,costs,andoutcomesofseveresepsisintheUnitedStates2003to 2007.CritCareMed2011;40:754–61.
4.TaniguchiLU,BierrenbachAL,ToscanoCM,SchettinoGP,AzevedoLC. Sepsis-relateddeathsinBrazil:ananalysisofthenationalmortalityregistryfrom2002 to2010.CritCare2014;18:608.
5. CohenJ,VincentJL,AdhikariNK,MachadoFR,AngusDC,CalandraT,etal.Sepsis: aroadmapforfutureresearch.LancetInfectDis2015;15:581–614.
6. VincentJL,MarshallJC,Namendys-SilvaSA,FrancoisB,Martin-LoechesI,Lipman J,etal.Assessmentoftheworldwideburdenofcriticalillness:theintensivecare overnations(ICON)audit.LancetRespirMed2014;2:380–6.
7. GlobalSepsisAlliance:factsheetsepsis; 2015. http://www.world-sepsis-day. org/CONTENTPIC/2015WSDFactSheetlongEnglish.pdf. [Accessed 18 January 2017].
8.IwashynaTJ,ElyEW,SmithDM,LangaKM.Long-termcognitiveimpairmentand functionaldisabilityamongsurvivorsofseveresepsis.JAMA2010;304:1787–94. 9.PandharipandePP,GirardTD,JacksonJC,MorandiA,ThompsonJL,PunBT, et al. Long-term cognitive impairmentafter critical illness. N EnglJ Med 2013;369:1306–16.
10.GamacheJrFW,DuckerTB.Alterationsinneurologicalfunctioninhead-injured patientsexperiencingmajorepisodesofsepsis.Neurosurgery1982;10:468–72. 11.SingerM,DeutschmanCS,SeymourCW,Shankar-HariM,AnnaneD,BauerM,
etal.TheThirdInternationalConsensusDefinitionsforSepsisandSepticShock (Sepsis-3).JAMA2016;315:801–10.
12.SilvaJM,dosSantosSDS.SepsisinAIDSpatients:clinical,etiologicaland inflam-matorycharacteristics.JIntAIDSSoc2013;16:17344.
13.Watkins CC, Treisman GJ. Cognitive impairment in patients with AIDS—prevalenceandseverity.HIVAIDS(Auckl)2015;7:35–47.
14. YuharaH,SteinmausC,CohenSE,CorleyDA,TeiY,BufflerPA.Isdiabetesmellitus anindependentriskfactorforcoloncancerandrectalcancer?AmJGastroenterol 2011;106:1911–22.
15. CochraneConsumersandCommunication:dataextractiontemplateforincluded studies; 2013. http://cccrg.cochrane.org/sites/cccrg.cochrane.org/files/public/ uploads/det2015revisedfinaljune202016nov29revised.doc. [Accessed 18 January 2017].
16. MerliM,LucidiC,PentassuglioI,GiannelliV,GiustoM,DiGregorioV,etal. Increasedriskofcognitiveimpairmentincirrhoticpatientswithbacterial infec-tions.JHepatol2013;59:243–50.
17.SemmlerA,WidmannCN,OkullaT,UrbachH,KaiserM,WidmanG,etal. Per-sistentcognitiveimpairment,hippocampalatrophyandEEGchangesinsepsis survivors.JNeurolNeurosurgPsychiatry2013;84:62–70.
18.Lahariya S, Grover S, Bagga S,Sharma A. Delirium in patients admitted toacardiacintensivecare unitwith cardiacemergenciesinadeveloping country:incidence,prevalence,riskfactorandoutcome.GenHospPsychiatry 2014;36:156–64.
19.GötzT,BaumbachP,HuonkerR,KrancziochC,WitteOW,DebenerS,etal.Slowed peakrestingfrequencyandMEGoveractivationinsurvivorsofseveresepsisand septicshock.ClinNeurophysiol2016;127:1247–53.
20.GötzT,BaumbachP,ReukenP,HuonkerR,KrancziochC,DebenerS,etal.Theloss ofneuralsynchronyinthepostsepticbrain.ClinNeurophysiol2017;127:2200–7. 21. DavydowDS,HoughCL,LangaKM,IwashynaTJ.Presepsisdepressivesymptoms areassociatedwithincidentcognitiveimpairmentinsurvivorsofseveresepsis: aprospectivecohortstudyofolderAmericans.JAmGeriatrSoc2012;60:2290–6. 22. RegazzoniCJ,ZamoraRJ,PetrucciE,PisarevskyAA,SaadAK,DeMolleinD,etal. Hospitaland1-yearoutcomesofsepticsyndromesinolderpeople:acohort study.JGerontolSerABiolSciMedSci2008;63:210–2.
23. ArinzonZ,PeisakhA,SchrireS,BernerYN.Deliriuminlong-termcaresetting: indicatortoseveremorbidity.ArchGerontolGeriatr2011;52:270–5.
24.PierrakosC,AttouR,DecorteL,KolyvirasA,MalinverniS,GottigniesP,etal. TranscranialDopplertoassesssepsis-associatedencephalopathyincriticallyill patients.BMCAnesthesiol2014;14:1–6.
Pleasecitethisarticleinpressas:CalsavaraAJC,etal.Post-sepsiscognitiveimpairmentandassociatedriskfactors:Asystematicreview. AustCritCare(2017),http://dx.doi.org/10.1016/j.aucc.2017.06.001
ARTICLE IN PRESS
G Model
AUCC-376; No.ofPages11
A.J.C.Calsavaraetal./AustralianCriticalCarexxx(2017)xxx–xxx 11
26.PierrakosC,AttouR,DecorteL,VelissarisD,CudiaA,GottigniesP,etal.Cerebral perfusionalterationsandcognitivedeclineincriticallyillsepsissurvivors.Acta ClinBelg2016;3286:1–6.
27.IwashynaTJ, CookeCR, WunschH, KahnJM.Populationburden of long-termsurvivorshipafterseveresepsisinOlderAmericans.JAmGeriatrSoc 2012;60:1070–7.
28.GuerraC,Linde-ZwirbleWT,WunschH.Riskfactorsfordementiaaftercritical illnessinelderlymedicarebeneficiaries.CritCare2012;16:R233.
29.BenrosME,SorensenHJ,NielsenPR,NordentoftM,MortensenPB,PetersenL.The associationbetweeninfectionsandgeneralcognitiveabilityinyoungmen—a nationwidestudy.PLoSOne2015;10:e0124005.
30. LazoskyA,YoungGB,ZirulS,PhillipsR.Qualityoflifeaftersepticillness.JCrit Care2010;25:406–12.
31. WHO.Internationalstatisticalclassificationofdiseasesandrelatedhealthproblems (InternationalClassificationofDiseases)(ICD)10thRevision—Version:2010.WHO (WorldHealthOrganisation);2010.
32. LangaKM,ChernewME,KabetoMU,HerzogAR,OfstedalMB,WillisRJ,etal. Nationalestimatesofthequantityandcostofinformalcaregivingfortheelderly withdementia.JGenInternMed2001;16:770–8.
33. IversonGL.Zscores.In:KreutzerJS,DeLucaJ,CaplanB,editors.Encyclopediaof clinicalneuropsychology.NewYork,NY:SpringerNewYork;2011.p.2739–40. 34.AmodioP,CampagnaF,OlianasS,IannizziP,MapelliD,PenzoM,etal.Detection
ofminimalhepaticencephalopathy:normalizationandoptimizationofthe Psy-chometricHepaticEncephalopathyScore.Aneuropsychologicalandquantified EEGstudy.JHepatol2016;49:346–53.
35.CentersforDiseaseControlandPrevention,NationalCenterforHealthStatistics. ICD–ICD-9-CM–InternationalClassificationofDiseases,NinthRevision,Clinical Modification.ClassifDisFunctDisabil2013;2008:1–2.
36.FliessbachK,HoppeC,SchlegelU,ElgerCE,HelmstaedterC.NeuroCogFX—Eine computergestützte neuropsychologische testbatterie für verlaufs-untersuchungen bei neurologischen erkrankungen. Fortschritte Neurol Psychiatr2006;74:643–50.
37.Gusmao-FloresD,SalluhJIF,ChalhubRÁ,QuarantiniLC.Theconfusion assess-mentmethodfortheintensivecareunit(CAM-ICU)andintensivecaredelirium screeningchecklist(ICDSC)forthediagnosisofdelirium:asystematicreview andmeta-analysisofclinicalstudies.CritCare2012;16:R115.
38. HerzogAR,WallaceRB.MeasuresofcognitivefunctioningintheAHEADStudy. JGerontolBPsycholSciSocSci1997;52:37–48(SpecNo).
39. JormAF.The InformantQuestionnaireoncognitivedecline intheelderly (IQCODE):areview.IntPsychogeriatr2004;16:275–93.
40. Lehrl S,Triebig G, Fischer B. Multiple choice vocabulary testMWT asa validandshorttest toestimatepremorbidintelligence.Acta NeurolScand 1995;91:335–45.
41.Sanchez-CubilloI,PerianezJA,Adrover-RoigD,Rodriguez-SanchezJM, Rios-LagoM,TirapuJ,etal.ConstructvalidityoftheTrailMakingTest:roleof task-switching,workingmemory,inhibition/interferencecontrol,and visuomo-torabilities.JIntNeuropsycholSoc2009;15:438–50.
42.SchroederRW,Twumasi-AnkrahP,BaadeLE,MarshallPS.ReliableDigitSpan: asystematicreviewandcross-validationstudy.Assessment2012;19:21–30. 43.TombaughTN,McIntyreNJ.Themini-mentalstateexamination:a
comprehen-sivereview.JAmGeriatrSoc1992;40:922–35.
44.WelshKA,BreitnerJCS,Magruder-HabibKM.Detectionofdementiainthe elderlyusingtelephonescreeningofcognitivestatus.Neuropsychiatry Neuropsy-cholBehavNeurol1993;6:103–10.
45.ScheurichA,MüllerMJ,SiessmeierT,BartensteinP,SchmidtLG,Fellgiebel A. ValidatingtheDemTect with 18-fluoro-2-deoxy-glucosepositron emis-siontomographyasasensitiveneuropsychologicalscreeningtestforearly alzheimerdiseaseinpatientsofamemoryclinic.DementGeriatrCognDisord 2005;20:271–7.
46. NasaP,JunejaD,SinghO.Severesepsisandsepticshockintheelderly:an overview.WorldJCritCareMed2012;1:23–30.
47. SternY,GurlandB,TatemichiTK,TangMX,WilderD,MayeuxR.Influence ofeducationandoccupationontheincidenceofAlzheimer’sdisease.JAMA 1994;271:1004–10.
48. CloustonSA,GlymourM,TerreraGM.Educationalinequalitiesinaging-related declinesinfluidcognitionandtheonsetofcognitivepathology.Alzheimers Dement(Amst)2015;1:303–10.
49.HindleJV,HurtCS,BurnDJ,BrownRG,SamuelM,WilsonKC,etal.Theeffects ofcognitivereserveandlifestyleoncognitionanddementiainParkinson’s disease—alongitudinalcohortstudy.IntJGeriatrPsychiatry2016;31:13–23. 50.RiveraMindtM,ByrdD,SaezP,ManlyJ.Increasingculturallycompetent
neu-ropsychologicalservicesforethnicminoritypopulations:acalltoaction.Clin Neuropsychol2010;24:429–53.
51.PandharipandePP,SandersRD,GirardTD,McGraneS,ThompsonJL,Shintani AK,etal.Effectofdexmedetomidineversuslorazepamonoutcomeinpatients withsepsis:anapriori-designedanalysisoftheMENDSrandomizedcontrolled trial.CritCare2010;14:R38.
52.DesamericqG,SchurhoffF,MearyA,SzokeA,Macquin-MavierI,Bachoud-Levi AC,etal.Long-termneurocognitiveeffectsofantipsychoticsinschizophrenia: anetworkmeta-analysis.EurJClinPharmacol2014;70:127–34.
53. KhalifaAE.Antiinfectiveagents affectingcognition:areview.J Chemother 2007;19:620–31.
54. DuningT,vandenHeuvelI,DickmannA,VolkertT,WempeC,ReinholzJ,etal. Hypoglycemiaaggravatescriticalillness-inducedneurocognitivedysfunction. DiabetesCare2010;33:639–44.
55. HauerD,KaufmannI,StreweC,BriegelI,CampolongoP,SchellingG.Theroleof glucocorticoids,catecholaminesandendocannabinoidsinthedevelopmentof traumaticmemoriesandposttraumaticstresssymptomsinsurvivorsofcritical illness.NeurobiolLearnMem2014;112:68–74.