REVISTA
BRASILEIRA
DE
ANESTESIOLOGIA
PublicaçãoOficialdaSociedadeBrasileiradeAnestesiologiawww.sba.com.br
SCIENTIFIC
ARTICLE
Upper
airway
morphology
in
Down
Syndrome
patients
under
dexmedetomidine
sedation
Rajeev
Subramanyam
a,∗,
Robert
Fleck
b,
John
McAuliffe
a,
Rupa
Radhakrishnan
b,
Dorothy
Jung
b,
Mario
Patino
a,
Mohamed
Mahmoud
aaDepartmentofAnesthesia,CincinnatiChildren’sHospitalMedicalCenter,OH,USA bDepartmentofRadiology,CincinnatiChildren’sHospitalMedicalCenter,OH,USA
Received29October2014;accepted26November2014 Availableonline19November2015
KEYWORDS
Airway;
Dexmedetomidine; Imaging;
DownSyndrome; Obstructivesleep apnea;
Sedation
Abstract
Backgroundandobjectives: ChildrenwithDownSyndromearevulnerabletosignificantupper
airwayobstructionduetorelativemacroglossiaanddynamicairwaycollapse.Theobjectiveof
thisstudywastocomparetheupperairwaydimensionsofchildrenwithDownSyndromeand
obstructivesleepapneawithnormalairwayunderdexmedetomidinesedation.
Methods:IRBapprovalwasobtained.Inthisretrospectivestudy,clinicallyindicateddynamic
sagittal midline magnetic resonance images ofthe upper airway were obtained under low
(1mcg/kg/h)andhigh(3mcg/kg/h)dosedexmedetomidine.Airwayanteroposteriordiameters
andsectional areaswere measuredas minimumandmaximumdimensionsby two
indepen-dentobserversatsoftpalate(nasopharyngealairway)andatbaseofthetongue(retroglossal
airway).
Resultsandconclusions: Minimum anteroposteriordiameter and minimumsectional area at
nasopharynxandretroglossalairwayweresignificantlyreducedinDownSyndromecomparedto
normalairwayatbothlowandhighdosedexmedetomidine.However,therewerenosignificant
differencesbetweenlowandhighdosedexmedetomidineinbothDownSyndromeandnormal
airway.ThemeanapneahypopneaindexinDownSyndromewas16±11.Under
dexmedetomi-dinesedation,childrenwithDownSyndromeandobstructivesleepapneawhencomparedto
normalairwaychildrenshowsignificantreductionsinairwaydimensionsmostpronouncedat
thenarrowestpointsinthenasopharyngealandretroglossalairways.
© 2015 Sociedade Brasileira de Anestesiologia. Published by Elsevier Editora Ltda. This
is an open access article under the CC BY-NC-ND license (http://creativecommons.org/
licenses/by-nc-nd/4.0/).
∗Correspondingauthor.
E-mail:[email protected](R.Subramanyam).
http://dx.doi.org/10.1016/j.bjane.2014.11.019
PALAVRAS-CHAVE
Viasaéreas; Dexmedetomidina; Imagem;
SíndromedeDown; Apneiaobstrutivado sono;
Sedac¸ão
MorfologiadasviasaéreassuperioresempacientescomsíndromedeDownsob sedac¸ãocomdexmedetomidina
Resumo
Justificativaeobjetivos: Ascrianc¸ascomsíndromedeDown(SD)sãovulneráveisàobstruc¸ão
significativadasviasaéreassuperioresdevidoàmacroglossiarelativaecolapsodinâmicodas
viasaéreas.Oobjetivodesteestudofoicompararasdimensõesdasviasaéreassuperioresde
crianc¸ascomSDeapnéiaobstrutivadosono(AOS)comviasaéreasnormais(VAN)sobsedac¸ão
comdexmedetomidina(DEX).
Métodos: Aprovac¸ãoIRBfoiobtida.Nesteestudoretrospectivo,imagensclinicamenteindicadas
de ressonânciamagnética dadinâmica dasviasaéreas superioresem plano sagitalnalinha
médiaforamobtidassobdosebaixa(1mcg/kg/h)edosealta(3mcg/kg/h)deDEX.Osdiâmetros
ânteroposterioresdasviasaéreaseasáreasseccionaisforammedidascomodimensõesmínimas
emáximaspordoisobservadoresindependentes,nopalatomole(regiãonasofaríngea)enabase
dalíngua(regiãoretroglossal).
Resultadoseconclusões: Odiâmetromínimoanteroposteriore aáreaseccional mínimadas
regiõesnasofaríngeaeretroglossalestavamsignificativamentereduzidosnaSDemcomparac¸ão
comVAN,tantocomadosebaixaquantocomadosealtadeDEX.Contudo,nãohouvediferenc¸as
significativasentreasdosesbaixaealtadeDEXem SDeVAN.Amédiadoíndice deapneia
e hipopneianaSDfoide 16±11. Sobsedac¸ãocomDEX, ascrianc¸as comSDeAOS quando
comparadascomascrianc¸ascomVANapresentaramreduc¸õessignificativasnasdimensõesdas
viasaéreas,maispronunciadasnospontosmaisestreitosdasregiõesnasofaríngeaeretroglossal.
© 2015 Sociedade Brasileira de Anestesiologia. Publicado por Elsevier Editora Ltda. Este
é um artigo Open Access sob a licença de CC BY-NC-ND (http://creativecommons.org/
licenses/by-nc-nd/4.0/).
Introduction
Down Syndrome (DS) or trisomy 21 is the most common geneticdisorderinhumanswithanestimatedbirthrateof 6000infants/year(1in691livebirths)intheUnitedStates.1 Obstructivesleepapnea(OSA)iscommonandnotedin79% ofchildrenwithDS(95%confidenceinterval,54---94%).2Risk factorsforOSAinthesechildrenincludemidfacehypoplasia, macroglossia,adenoidandtonsillarhypertrophy, laryngotra-chealanomalies,obesity,andmuscularhypotonia.3Evenin theabsenceofOSA,childrenwithDShavereducedairway sizecausedby softtissuecrowdingwithinasmallerfacial skeletalanatomy.4
Children with OSA, with or without DS, are sensitive to respiratory depression by opioids, sedatives, and hyp-notics.Theyareespeciallyvulnerabletothedevelopmentof upperairwayobstructionduring sedationandanesthesia.5 Dexmedetomidine(DEX)isan␣-2receptoragonistcurrently
being used off-label for sedation in pediatric patients at manyinstitutions.Incontrasttoothersedativeagents,DEX has been shown tohave sedative properties that parallel naturalnon-rapideyemovementsleep,withoutsignificant respiratory depression.6,7 These advantages make DEX an attractiveagentforsedatingchildrenwithOSA.8 Wehave previouslyusedmagneticresonanceimaging(MRI)toassess theeffectofincreasingdosesofDEXonairwaydimensionsin childrenwithnormalupperairways(agerange3---10years) andshowedthatincreasingdosesofDEXinthesechildrenis notassociatedwithsignificantincreaseinthedegreeof air-wayobstruction.9 Werecentlyusedasimilarmethodology tocompare the dose---response effectsof DEX and propo-folonairwaymorphology inchildren withOSA (agerange
1---16years).Wefoundthatasthedosageincreased, aver-ageairwaydimensionsweretypicallyunchangedorslightly increased with DEX compared to unchanged or slightly decreasedwithpropofol.10
Ouraim inthe present study wastotest the hypothe-sisthatDSchildrenwithOSAhavesignificantupperairway collapsibilityevenatlowdosesofDEXcomparedtochildren withnormalairway(NA).Wethereforedesigneda retrospec-tivecohortstudycomparingtheupperairwaymorphologies ofchildrenaged3---10yearswithDSandOSAtothosewith NAunderincreasingdosesofDEXsedation.
Materials
and
methods
After institutional review board approval, the data were obtainedinchildrenaged3---10yearswithDSandchildren withNAwhounderwentMRIairwayanalysiswithDEX. Writ-teninformedconsenthadbeenobtainedforsedation.The needforaseparateinformedconsentfortheretrospective reviewwaswaivedbyourIRB.
DownSyndrome(DS)group
induction room with sevoflurane and/or nitrous oxide in oxygen. Atropine 10mcg/kg IV was administered and sevoflurane and/or nitrous oxide were discontinued. DEX wasstartedandMRimagingperformedasdescribedbelow.
Normalairway(NA)group
Themethodologyusedforevaluationofchildrenwith nor-mal airways wasdescribe in a previously study.9 Inbrief, childrenaged3---10yearswhopresentedforanelectiveMRI examination undersedation were included. Children with historyofOSAorsnoring,AmericanSocietyof Anesthesiol-ogyclassification>2,allergytoDEX,presenceofairwayor craniofacialabnormality,obesity,or severedevelopmental delaywereexcluded.
Dexmedetomidineprotocol
BaselineairwayimageswereobtainedduringtheLowDEX infusion (1mcg/kg/h). If the subject moved, a bolus of 0.5mcg/kgover10minwasgivenandtheDEXinfusionrate wasincreasedto1.5mcg/kg/h.Ifthesubjectmoveda sec-ondtime,theresearchstudywasterminatedandadditional anesthesiawasprovidedwithpropofol infusion. Afterthe initialsetofairwayimageswereobtained,abolusdoseof DEX2mcg/kgwasgivenover10minfollowedbyanincrease intheinfusionrateto3mcg/kg/h(highdoseDEX). Univer-sityof Michigan SedationScale (UMSS) wasusedtoassess sedation.11UMSSisasimpletouse,validatedtooltoassess thedepthofsedationinchildren.11Standardmonitoringand spontaneousbreathingwith2L/minofoxygenvianasal can-nulawasused.Levelofsedationwasassessedafterinitial
low dose of DEX before and after imaging. Sedation was not assessed during imaging as this may have necessi-tatedchangingthepatient’sheadpositionandsubsequently biasing airway measurement comparisons. Patients were transferredtothepostanesthesiacareunitfollowing imag-inganddischargedhomeaftermeetingcriteria.
MRimagingprotocol
All patients underwentclinically indicated MRIunder DEX sedation.Onceadequatesedationwasachieved,the cervi-calspinewasmaintainedinaneutralpositionbyplacingthe patient’sheadandneckinavascularcoil.Noartificial air-way(e.g.,oral airwayor nasaltrumpet)orpositioningaid (e.g.,shoulder roll) wasusedduringimaging. Noattempt wasmadetoopenorclosethemouth.ChildrenwithDSwere transferred to the imager after the sevoflurane end-tidal concentrationwasreducedto<0.1%.MRIwasperformedon a1.5Teslaimager(GEHealthcare,Milwaukee,WI,USA)with an8-channelreceiveronlyneurovascularphasedarraycoil (MEDRAD,Inc.,Indianola,PA,USA).Theprimaryimagesfor analysiswererapidlyacquired ina midline,sagittalplane usingfastgradient echoimaging(1 imageevery800ms).9 The scan parameters were: Repetition time/Echo time: 6.98/3.6,Fieldofview:24cm,slicethickness:5mm,matrix 256×128,numberofexcitations:1,flipangle:80◦,receiver
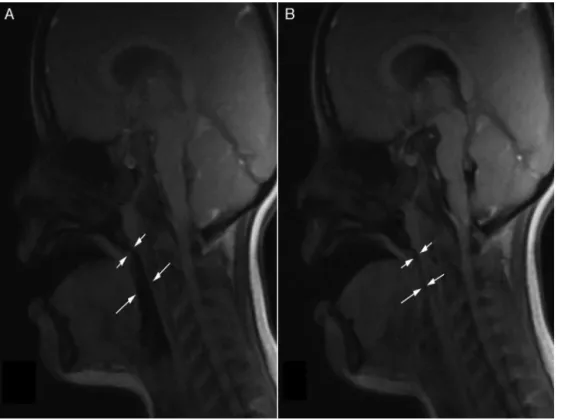
bandwidth 244.1Hz/pixel,baseline resolution: 256,phase resolution: 128. By playing a cine loop of the images, a movieofairwaymotionwascreated(Figs.1and2with asso-ciated video). Upper airwayimageswere obtained during low(1mcg/kg/h)andhigh(3mcg/kg/h)doseDEXsedation. TheimageswerestoredonthePACS(PictureArchivingand
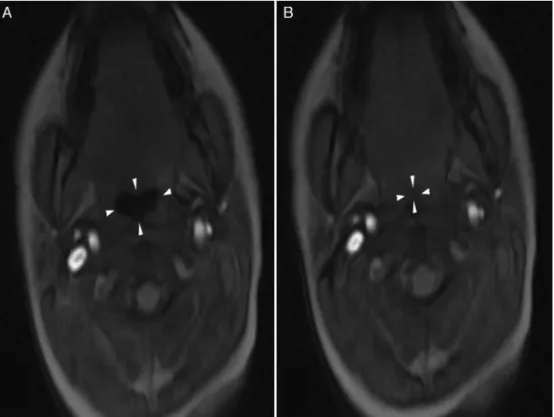
Figure2 Stillimage(A)fromthecineclipshowstheretroglossalairwayincross-sectionwhileopen(arrowheads)andthestill image(B)showstheairwaycompletelycollapsedcentrallycompatiblewithhypopharyngealcollapse.
CommunicationSystem)and reviewedbytwoscorerswho wereblindedtotheDEXdoses.
Theairwaywasmeasuredatthelevelofthesoftpalate (nasopharyngeal airway)and thebase of the tongue (ret-roglossal airway) (Figs. 1 and 2). The sectional area and anterior-posteriordiameterweremeasuredinthe nasopha-ryngeal area (NPA) and the retroglossal area (RGA). The nasopharyngealarea(NPA)wasdefinedanteriorlybya verti-callinetangentialtotheposteriorinferiornasalturbinate, posteriorly by the posterior wall of nasopharynx, superi-orlybythesuperiorwallofnasopharynx,andinferiorlyby superiorandposteriorpartofhardandsoftpalate.The ret-roglossal airwayarea(RGA) wasdefinedanteriorly by the backoftongue,posteriorlybytheposteriorpharyngealwall, superiorlybyahorizontallinedrawnattheinferiormargin ofthesoftpalate,andinferiorlybyahorizontallinedrawn atthebase ofthetongue. Thesectionalareaand antero-posteriordiameterweremeasured intheNPAandRGAon imagesofminimumandmaximumexpansionoftheairway.
Poweranalysis
Analyses performed with R statistical software indicated thatasamplesizeof7patientswouldhavean80%powerto detecta100mm2differenceinthemeansectionalareasof NAandDSairwaysinchildrenunderlowdoseDEX.12A differ-enceof100mm2waschosenbecausechildrenwithDShave baselinenarrowairwaysandwasbasedona95%confidence levelinthemeandifferences.13LowdoseDEXwaschosenin ordertodeterminethemostconservativeestimateof sam-plesizenecessarytoadequatelypowerthestudy.Thiswas doneundertheassumptionthathighdoseDEXwouldcause
moresignificantairwaynarrowingthanlow doseDEXand, subsequently,themeandifferenceinairwaymeasurements wouldbelarger.Aposthocpower analysiswasperformed toverifythisassumption.
Statisticalanalysis
Statistical analysis was performed with R statistical software.12 Normalityof distributionof data waschecked by Shapiro---Wilks test. Descriptive statistics are provided asmean and standard deviation or numbers as appropri-ate.Age, weight, and polysomnography derived variables betweenchildrenwithNAandchildrenwithDSwere com-paredwithWelchtwo-samplet-test.Genderwascompared withtheFischerexacttest.Hemodynamicdatawere com-paredwiththeunpairedt-test.Theminimumandmaximum anteroposteriordiameterandsectionalareasintheNPAand RGAwerecomparedbetweenchildrenwithDSandchildren withNAusingtheunpairedt-test.Thedifferencebetween ‘lowdoseDEX’and‘highdoseDEX’betweenchildrenwithDS andchildrenwithNAwascomparedusingthepairedt-test. Ap-valueof<0.05wasconsideredstatisticallysignificant.
Results
Table1 Demographicandpolysomnographyfindings. Down
Syndrome (n=7)
Normal airway (n=23)
p
Age(years) 5±1 6±2 0.27
Weight(kg) 26±11 22±5 0.42
Male/females 4/3 12/11 1
Sedationscore
Induction 3±1 2±1 0.003
Postanesthesia
careunit
3±0 3±0 0.36
Obstructivesleep apnea(n)
7 0
Polysomnographyfindings
Apneahypopnea
index (events/hour)
17±11
(5.2---37.6)
---Minimaloxygen
saturation(%)
81±5
(72---85)
---All data in mean±standard deviation or absolute numbers.
Rangesarementionedinparenthesis.---,notapplicable.
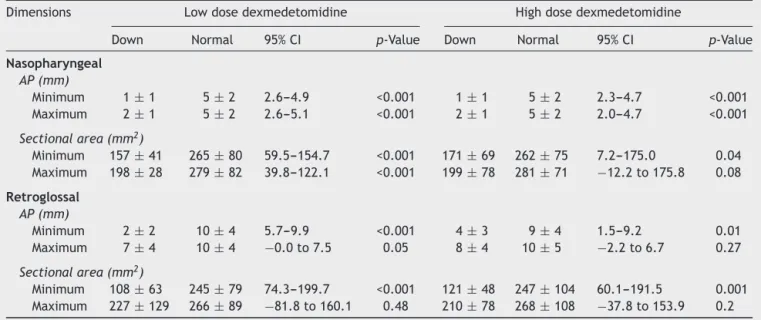
Airwayanteroposteriordiameterandsectionalarea mea-surementsaresummarizedinTable3.
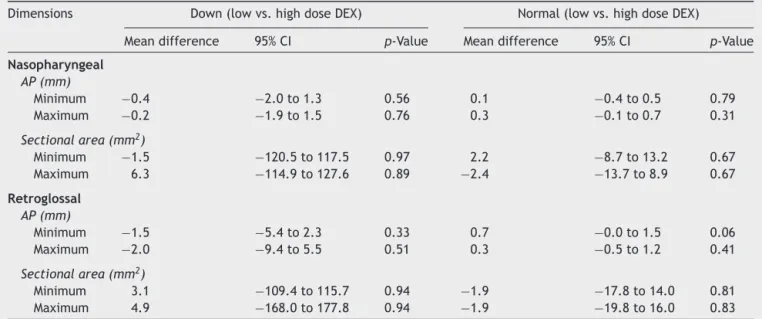
The following three dimensions were reduced signifi-cantlyin childrenwithDSascomparedtoNAat both low andhighdoseDEX:minimumRGAsectionalarea,minimum anteroposteriorNPAdiameter,andminimumanteroposterior RGAdiameter.SedationwithDEXdidnotyieldastatistically significantdose-dependent(lowvs.high)differenceinthe airwaymeasurementsofchildrenwithDSandNA(Table4).
Discussion
OurstudyshowedthatchildrenwithDSandOSAexhibited significantreductionsinanatomicalairwaydimensionswhen comparedtochildrenwithNAunderDEXsedation.Thesafe sedationofchildrenespeciallythosewithahistoryofOSA requiresaclearunderstandingofthepharmacokineticand pharmacodynamiceffectsofthesedativeusedaswellasan appreciationoftheeffectofthechosensedativeonairway collapsibility.Allpractitionersprovidingsedationmusthave anindepthunderstandingoftheinteractionbetweendepth ofsedationandairwaydynamics,rememberingthatdepth of sedation is a continuum fromminimal, moderate, and deepsedationtogeneralanesthesia.
Table2 Hemodynamicdata.
Baseline p Firstscan p HighdoseDEX p
Down Syndrome
Normal children
Down Syndrome
Normal children
Down Syndrome
Normal children
HR(bpm) 101±12 93±14 0.15 97±19 78±15 0.03 84±9 82±19 0.88
SBP(mmHg) 110±10 107±14 0.50 125±14 111±15 0.13 137±14 112±14 0.08
DBP(mmHg) 64±9 56±19 0.16 69±9 59±12 0.15 84±2 66±12 <0.001
HR,heartrate;SBP,non-invasivesystolicbloodpressure;DBP,non-invasivediastolicbloodpressure.
Table3 ComparisonofairwaydimensionsbetweenchildrenwithDownSyndromeandchildrenwithnormalairwayunderboth
lowandhighdosedexmedetomidine.
Dimensions Lowdosedexmedetomidine Highdosedexmedetomidine
Down Normal 95%CI p-Value Down Normal 95%CI p-Value
Nasopharyngeal
AP(mm)
Minimum 1±1 5±2 2.6---4.9 <0.001 1±1 5±2 2.3---4.7 <0.001
Maximum 2±1 5±2 2.6---5.1 <0.001 2±1 5±2 2.0---4.7 <0.001
Sectionalarea(mm2)
Minimum 157±41 265±80 59.5---154.7 <0.001 171±69 262±75 7.2---175.0 0.04
Maximum 198±28 279±82 39.8---122.1 <0.001 199±78 281±71 −12.2to175.8 0.08
Retroglossal
AP(mm)
Minimum 2±2 10±4 5.7---9.9 <0.001 4±3 9±4 1.5---9.2 0.01
Maximum 7±4 10±4 −0.0to7.5 0.05 8±4 10±5 −2.2to6.7 0.27
Sectionalarea(mm2)
Minimum 108±63 245±79 74.3---199.7 <0.001 121±48 247±104 60.1---191.5 0.001
Maximum 227±129 266±89 −81.8to160.1 0.48 210±78 268±108 −37.8to153.9 0.2
Table4 Comparisonofmeandifferencesinairwaydimensionsbetweenlowandhighdosedexmedetomidineinchildrenwith
normalairwayandchildrenwithDownSyndrome.
Dimensions Down(lowvs.highdoseDEX) Normal(lowvs.highdoseDEX)
Meandifference 95%CI p-Value Meandifference 95%CI p-Value
Nasopharyngeal
AP(mm)
Minimum −0.4 −2.0to1.3 0.56 0.1 −0.4to0.5 0.79
Maximum −0.2 −1.9to1.5 0.76 0.3 −0.1to0.7 0.31
Sectionalarea(mm2)
Minimum −1.5 −120.5to117.5 0.97 2.2 −8.7to13.2 0.67
Maximum 6.3 −114.9to127.6 0.89 −2.4 −13.7to8.9 0.67
Retroglossal
AP(mm)
Minimum −1.5 −5.4to2.3 0.33 0.7 −0.0to1.5 0.06
Maximum −2.0 −9.4to5.5 0.51 0.3 −0.5to1.2 0.41
Sectionalarea(mm2)
Minimum 3.1 −109.4to115.7 0.94 −1.9 −17.8to14.0 0.81
Maximum 4.9 −168.0to177.8 0.94 −1.9 −19.8to16.0 0.83
Themeandifferencesinthistableareobtainedasapairedanalysisfromthedatasetanddonotrepresentsimplythedifferenceoftwo
correspondingmeansfromTable3.
Sedatingor anesthetizingachildknowntohave OSAis achallengebecauseanestheticagentsbluntarousal mech-anisms,decreaserespiratorydrive, andreducepharyngeal muscle tone. More than half of all children with DShave OSA, andthese childrenare at higherrisk of adverse air-wayeventsduringproceduralsedation.Ourstudyexamined theanatomicalsagittalsectionalareasanddiameteratthe criticalpartof theairway. Weshow thatairwaysectional areasanddiametersweresignificantlyreducedinchildren withDS compared tothose children withNA at both low andhighdoseDEX.Thechangesinairwaydimensionswere notdose-dependentwithinthe patientgroups. The seem-ingindependence of airwaydimensions andDEXdose can beexplainedbytherelationshipbetweenconsciousnessand upperairwaycollapsibility.Profoundchangesin upper air-waymuscleactivityandcollapsibilityoccurproximatetothe lossof consciousnessandrelatively modestchanges occur withincreasingdepthofanesthesia/sedation.14
Furthermore,wequantitatetheeffectofDEXsedationon airwaymorphologyinchildrenwithDS.Itisourhope that practitioners can utilize this information tobetter assess the depthof sedation of their DSpatients and ultimately avoid theadverse effects of over sedation(e.g. hypoven-tilation,airway obstruction). As pediatric DSpatients can rapidly obstructtheir airways evenat low doses of seda-tion,providersshouldalsoconfirmthatairwaymanagement instrumentsarereadilyavailablebeforesedatingthese chil-dren.
PatientswithDSalsohaveahigherchanceofpersistent OSA followingtonsillectomydue torecurrentenlargement of lingual tonsils and adenoid tissue, reduced muscular tone,hypopharyngealcollapse,andglossoptosis.15,16MRIof the airway in adolescents for evaluation of OSA revealed thatchildrenwithDShavedisproportionatelylargetongues in comparisonto the craniofacialparameters of age- and gender-matched controls.3 The findings from the present
studyshowthatairwaysizewasstablebetweenthedosage levels of DEX studied. This suggests that the airway col-lapsibilityisprobablycausedbyreducedmusculartoneand hypopharyngealcollapse,glossoptosis,midfacehypoplasia, andrelativemacroglossia.3,15,16Pharyngealcollapseismore severeinchildrenwithDScomparedtocontrols, indepen-dentofage,gender,andbodymassindex.17Theaugmented upperairwaydilatoractivitypresentin theawake stateis reduced at sleep onset, and is further attenuated during rapideyemovementsleep,contributingtopharyngeal col-lapse in children withOSA.18,19 Additionalcontributors to airwayobstructionduringsedationwithintravenous pento-barbital inchildren with moderateOSA includelarge soft palate,and,largeadenoidsandtonsils.20
Theairwaysectionalareaandanteroposteriordiameter weremeasuredasminimumandmaximum,whichrepresent dimensions during inhalation and exhalation respectively. Thiswas donetointerpret relative changesin theairway duringtherespiratorycycle.Thepeakimageacquisitionrate of800mswasperformedandallowedforrandomsampling during the respiratory cycle. Segmentation of the airway sizeover thebreathingcycle andtheuseof thepeaksize andminimal sizeobviatedtheneedforsynchronization to therespiratorycycle.Theairwayislargestduringexpiration andsmallestduringinspirationunlesstongue/jawthrusting ispresentandtheairwayismoredynamicinOSA.21
higher fatigability seen in inspiratory muscles in patients withOSA.23
Thereareafewlimitationswithourstudy.Althoughour sample size remains a limitation and may limit our sta-tistical significance particularly withhigh dose DEX group nasopharyngealareameasurements(post hocpower71%), ourpreliminaryfindingsarerelevantindefiningand apply-ing interventionsto improveairway outcomes in children undergoingsedation outside the operating room. Second, all of the patients withDS in our study had moderate to severeOSA. Thefindings maybedifferentinthe minority ofthosepatients withDSwithnoOSA. Whileit wouldbe idealtostudyDSpatientswithnoOSAasathirdgroup,the availabilityofanadequatenumberofDSpatientshavingMRI ofairwayfornon-OSAindicationsisamajorlimitingfactor. Last,themeansedationscoreinchildrenwithDSwas sig-nificantlyhigherduringinduction ascomparedtochildren withNAand mayhaveaffected measurementsduringlow doseDEX.AlthoughUMSScaptureschangesinthedepthof sedation,theinabilityofthescaletodiscriminatemoderate anddeeplevelsofsedationmaylimititsusefulnessinsuch situations.24
Conclusions
Insummary,childrenwithDSwithOSAexhibitedsignificant reductionsinanatomicalairwaydimensionswhencompared to children with NA under DEX sedation, supporting our hypothesis.The relativereductioninairwaydimensions is equalatbothlow doseandhighdoseDEX,whichsuggests thattheobserveddifferencesareuniquetoDSandnotdue todifferences insedation.These changesaremost signif-icant at the narrowest points in the nasopharyngeal and retroglossalairways.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
References
1.ParkerSE,MaiCT,CanfieldMA,etal.UpdatedNationalBirth PrevalenceestimatesforselectedbirthdefectsintheUnited States, 2004---2006. Birth Defects Res A: Clin Mol Teratol. 2010;88:1008---16.
2.Shete MM, Stocks RM, Sebelik ME, et al. Effects of adeno-tonsillectomyonpolysomnographypatternsinDownsyndrome childrenwithobstructivesleepapnea:acomparativestudywith childrenwithoutDownsyndrome.IntJPediatr Otorhinolaryn-gol.2010;74:241---4.
3.GuimaraesCV,DonnellyLF,ShottSR,etal.Relativeratherthan absolutemacroglossiainpatientswithDownsyndrome: implica-tionsfortreatmentofobstructivesleepapnea.PediatrRadiol. 2008;38:1062---7.
4.UongEC,McDonough JM,Tayag-Kier CE,etal. Magnetic res-onance imagingof the upper airway in children with Down syndrome.AmJRespirCritCareMed.2001;163:731---6.
5.BrownKA.Outcome,risk,anderrorandthechildwith obstruc-tivesleepapnea.PaediatrAnaesth.2011;21:771---80.
6.Nelson LE, Lu J, Guo T, et al. The alpha2-adrenoceptor agonist dexmedetomidine converges on an endogenous
sleep-promoting pathway to exert its sedative effects. Anesthesiology.2003;98:428---36.
7.HsuYW, Cortinez LI,RobertsonKM,et al. Dexmedetomidine pharmacodynamics:PartI:Crossovercomparisonofthe respi-ratoryeffectsofdexmedetomidineandremifentanilinhealthy volunteers.Anesthesiology.2004;101:1066---76.
8.Mahmoud M, Gunter J, Donnelly LF, et al. A comparison of dexmedetomidinewithpropofolformagneticresonance imag-ingsleepstudiesinchildren.AnesthAnalg.2009;109:745---53.
9.Mahmoud M, Radhakrishman R, Gunter J, et al. Effect of increasingdepthofdexmedetomidineanesthesiaonupper air-waymorphologyinchildren.PaediatrAnaesth.2010;20:506---15.
10.MahmoudM,JungD,SalisburyS,etal.Effectofincreasingdepth ofdexmedetomidineandpropofolanesthesiaonupperairway morphologyinchildrenandadolescentswithobstructivesleep apnea.JClinAnesth.2013;25:529---41.
11.MalviyaS,Voepel-LewisT,TaitAR,etal.Depthofsedationin childrenundergoingcomputed tomography:validityand reli-abilityoftheUniversityofMichiganSedationScale(UMSS).Br JAnaesth.2002;88:241---5.
12.RCoreTeam.R:alanguageandenvironmentforstatistical com-puting.Vienna,Austria:RFoundationforStatisticalComputing; 2013.
13.Dyken ME, Lin-Dyken DC, Poulton S, et al. Prospective polysomnographicanalysisofobstructivesleepapneainDown syndrome.ArchPediatrAdolescMed.2003;157:655---60.
14.HillmanDR,WalshJH,MaddisonKJ,etal.Evolutionofchanges inupperairwaycollapsibilityduringslowinductionof anesthe-siawithpropofol.Anesthesiology.2009;111:63---71.
15.DonnellyLF,ShottSR,LaRoseCR, etal.Causes ofpersistent obstructive sleep apnea despite previous tonsillectomy and adenoidectomyin childrenwithDown syndrome as depicted on static and dynamic cine MRI. AJR Am J Roentgenol. 2004;183:175---81.
16.Fricke BL, Donnelly LF, Shott SR, et al. Comparison of lin-gualtonsil sizeas depictedonMR imagingbetweenchildren withobstructive sleep apnea despite previous tonsillectomy and adenoidectomy and normal controls. Pediatr Radiol. 2006;36:518---23.
17.Fung E, Witmans M, Ghosh M, et al. Upper airway findings in children with Down syndrome on sleep nasopharyn-goscopy:case---control study. JOtolaryngol Head Neck Surg. 2012;41:138---44.
18.FogelRB,TrinderJ,MalhotraA,etal.Within-breathcontrolof genioglossalmuscleactivationinhumans:effectofsleep-wake state.JPhysiol.2003;550:899---910.
19.MezzanotteWS,TangelDJ,WhiteDP.Wakinggenioglossal elec-tromyogram in sleep apnea patients versus normal controls (a neuromuscular compensatory mechanism). J Clin Invest. 1992;89:1571---9.
20.ArensR, McDonough JM,Costarino AT, et al. Magnetic reso-nanceimagingoftheupperairwaystructureofchildrenwith obstructivesleepapneasyndrome.AmJRespirCritCareMed. 2001;164:698---703.
21.AbbottMB,DonnellyLF,DardzinskiBJ,etal.Obstructivesleep apnea:MRimagingvolumesegmentation analysis.Radiology. 2004;232:889---95.
22.MihaescuM,MurugappanS,GutmarkE,etal.Computational fluid dynamics analysis of upper airway reconstructed from magneticresonance imagingdata. AnnOtolRhinol Laryngol. 2008;117:303---9.
23.ChienMY,WuYT,LeePL,etal.Inspiratorymuscledysfunction inpatientswithsevereobstructivesleepapnoea.EurRespirJ. 2010;35:373---80.