www.jped.com.br
ORIGINAL
ARTICLE
Evaluation
of
a
neonatal
screening
program
for
sickle-cell
disease
夽
,
夽夽
Rodrigo
Eller
a,∗,
Denise
Bousfield
da
Silva
baMedicalSchool,UniversidadeFederaldeSantaCatarina(UFSC),Florianópolis,SC,Brazil
bDepartmentofPediatrics,UniversidadeFederaldeSantaCatarina(UFSC),Florianópolis,SC,Brazil
Received14June2015;accepted21October2015 Availableonline15February2016
KEYWORDS
Sicklecelldisease; Neonatalscreening; SantaCatarinaState
Abstract
Objective: EvaluatetheNeonatalScreeningProgramoftheHealthSecretariatoftheStateof SantaCatarinaforsickle-celldisease,fromJanuary2003toDecember2012,regardingprogram coverageanddiseasefrequency.
Methods: Descriptive,observational,cross-sectionalstudywithretrospectivedatacollection. Thevariablesanalyzedwere:numberoflivebirthsintheStateofSantaCatarina;numberof screenedchildren;numberofchildrendiagnosedwithsickle-celltraitandsickle-celldisease; typeofsickle-celldiseasediagnosed;ageatthetimeofsamplecollection,ethnicity/skincolor, gender, andorigin ofchildren with sickle-cell disease. Descriptivemeasures andfrequency tableswereusedfordataanalysis.
Results: Duringthestudy period,therewere 848,833livebirthsand730,412sampleswere screenedbytheprogram,resultinginacoverageof86.0%.Therewere6173samplespositive forsickle-celltraitand39forsickle-celldisease.Amongchildrenwithsickle-celldisease,the medianageatthetimeofsamplecollectionwas6days.Regardingtheethnicity/skincolor, 25(64.1%)children werewhite,sevenwereblack,andsevenotherswerenotspecified.The MidwestandtheHighland(PlanaltoSerrano)ofSantaCatarinaweretheregionswiththehighest incidenceofsickle-celldisease.
Conclusion: CoveragebytheNeonatalScreeningProgramofSantaCatarinaisgood,butdidnot demonstrateanimprovementtrendovertheyears.Thefrequencyofsickle-celldiseaseislow andlowerthanintheNorth,Northeast,andMidwestregions.Themedianageindaysatthe timeofcollectionisolderthantheagerecommendedbytheMinistryofHealth.
©2016SociedadeBrasileiradePediatria.PublishedbyElsevierEditoraLtda.Thisisanopen accessarticleundertheCCBY-NC-NDlicense(http://creativecommons.org/licenses/by-nc-nd/
4.0/).
夽 Please citethisarticleas:EllerR, SilvaDB. Evaluationof aneonatal screeningprogramfor sickle-celldisease. JPediatr (Rio J).
2016;92:409---13.
夽夽ArticleassociatedtoHospitalInfantilJoanadeGusmão,Florianópolis,SC,Brazil.
∗Correspondingauthor.
E-mail:rodrigoellersrl@gmail.com(R.Eller).
http://dx.doi.org/10.1016/j.jped.2015.10.002
PALAVRAS-CHAVE
Doenc¸afalciforme; Triagemneonatal; EstadodeSanta Catarina
Avaliac¸ãodeumprogramadetriagemneonatalparadoenc¸afalciforme
Resumo
Objetivo: avaliaroProgramadeTriagemNeonataldaSecretariadeSaúdedoEstadodeSanta Catarina(PTN-SES/SC)paradoenc¸afalciformenoperíododejaneirode2003adezembrode 2012,emrelac¸ãoàsuacoberturaeàfrequênciadadoenc¸a.
Métodos: estudodescritivo,observacionaletransversalcomcoletaretrospectivadosdados. Asvariáveisanalisadasforam:númerodenascidos-vivosnoEstadodeSantaCatarina;número decrianc¸astriadas;númerodecrianc¸asdiagnosticadascomtrac¸oedoenc¸afalciforme(DF); tipodeDFdiagnosticada;idadedacoleta,cor/rac¸a,sexoeprocedênciadascrianc¸ascomDF. Foramutilizadasasmedidasdescritivaseastabelasdefrequênciaparaanálisedosdados.
Resultados: noperíodoestudado,houve848.833nascidos-vivose730.412amostrastriadaspelo programa,gerandocoberturade86,0%.Dasamostrastriadas,foramencontradas6.173crianc¸as comtrac¸ofalciformee39comDF.Entreascrianc¸ascomDF,amedianadaidadeemdiasna data dacoleta foide6. Das39 crianc¸asdoentes,25(64,1%)eramdacor/rac¸abranca,7da negrae7deoutracor/rac¸a.Asregiõesdoplanaltoserranoedomeio-oestedeSantaCatarina foramasregiõescommaiorincidênciadeDF.
Conclusões: acoberturadoPTN-SES/SCéboa,contudonãoapresentoutendênciademelhora ao longodos anos.A frequência daDF ébaixa emenor quenas regiõesNorte, Nordestee Centro-oeste.Amedianadaidadeemdiasnomomentodacoletaestáacimadopreconizado peloMinistériodaSaúde.
©2016SociedadeBrasileiradePediatria.PublicadoporElsevierEditoraLtda.Este ´eumartigo OpenAccesssobumalicenc¸aCCBY-NC-ND(http://creativecommons.org/licenses/by-nc-nd/4.
0/).
Introduction
Sickle-celldisease(SCD)representsagroupof autosomal-recessiveinheritedhematologicaldiseases,whichincludes several genotypes, with a prevalence of hemoglobin S (HbS).1---3 The presence of this abnormal hemoglobin is responsiblefor themainclinicalmanifestationsofthe dis-ease,whichoriginatefromvaso-occlusive phenomenaand chronicischemicdisorders.4
The distributionof SCD is a heterogeneous one and is morecommonamongthoseofAfricanascendancy.According to2009datafromtheBrazilianInstituteofGeographyand Statistics(Instituto Brasileiro deGeografia e Estatística ---IBGE),in Brazil, black and brown individuals represented 6.9%and 44.2% of thepopulation, respectively, reflecting theheterogeneityofSCDinthecountry.5
InJune2001,throughMinistryofHealthDecreeNo.822, severaldiseases wereadded tothelistof thosescreened throughtheexistingNational Neonatal Screening Program (NSP)(phenylketonuriaandcongenitalhypothyroidism),to includethedetectionofSCDandotherhemoglobinopathies, as well as cystic fibrosis.6 The diagnosis of SCD is per-formed by a laboratory through the detection of HbS and its association with quantitative fractions of other hemoglobins.
The NSP, together with multidisciplinary care, has allowedasignificantlyreductioninmorbidityandmortality fromthedisease,asshownbystudiesinothercountries.6
The aim of this study wasto evaluate the NSP of the Health Secretariat of the State of Santa Catarina (Pro-grama de Triagem Neonatal da Secretaria da Saúde do EstadodeSanta Catarina[NSP-SES/SC])for SCDandother hemoglobinopathiesin relation totheir coverage and the
incidenceofSCDandsickle-celltraitfromJanuary2003to December2012.
Methods
Thiswasadescriptive,observational,cross-sectionalstudy approvedbytheEthicsCommitteeofHospitalInfantilJoana deGusmãounderopinionNo.029/2013.
The assessed variables were obtained retrospectively fromadatabaseattheCentralLaboratory(Laboratório Cen-tral[LACEN])oftheHealthSecretariatoftheStateofSanta Catarina(SecretariadaSaúdedoEstadodeSantaCatarina [SES/SC])andtheLiveBirthInformationSystem(Sistemade Informac¸õessobreNascidosVivos[SINASC]).The collected datacomprise theperiodfromJanuary2003toDecember 2012.
Thevariablesincluded:numberoflivebirthsintheState ofSantaCatarina;numberofchildrenscreenedbythe NSP-SES/SCfor SCD andother hemoglobinopathies;number of children diagnosed with SCD and sickle-cell trait by the NSP-SES/SC;typeofSCDdiagnosedbytheaforementioned program;finaldiagnosisofchildrenwhosefirstsampleswere inconclusive;ageatcollection,ethnicity/skincolor,gender, and origin of children with SCD according to the macro-regionsofSantaCatarina.7
Regardingthe variableethnicity/skincolor, it usedthe criterionestablishedbytheBrazilianInstituteofGeography andStatistics(InstitutoBrasileirodeGeografiaeEstatística [IBGE]).5Theinformationonthisvariablewasobtainedfrom thefilecompletedbythenursingstaffatthetimeofblood samplecollection.
inthemunicipalitiesofSantaCatarina.The sample,taken fromthechild’sheelanddepositedonfilterpaperdiscs,was latersenttoLACENforanalysis.
The method used to test blood samples was high-performance liquid chromatography associated with the cation-exchangechromatography,usingtheVariantIIdevice (Bio-Rad®,CA,USA).
Samples with results different from the Hb FA pat-tern were submitted to another distinct chromatography test.Theisoelectricfocusingtestwasusuallyusedforthis assessment. When theresults of both testsweredistinct, constitutinganinconclusiveresult,thechildwascalledfor anewsamplecollection.
Children whose samples had an indeterminate result, i.e.,ahemoglobintypedifferentfromthehemoglobinthat canbeidentifiedbystandardscreeningtests,werereferred fordiagnosticclarificationatHospitalInfantilJoanade Gus-mão(HIJG),areferralcenterintheStateofSantaCatarina forneonatalscreeningtests.
Descriptivemeasuresandfrequencytableswereusedfor analysisoftheresults.
Results
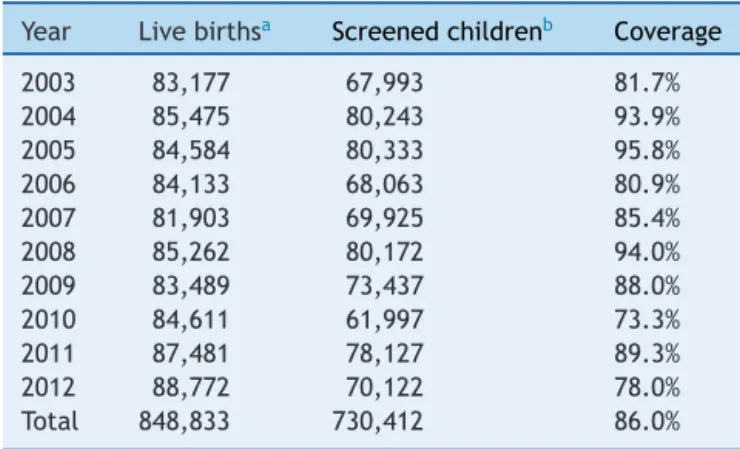
FromJanuary2003toDecember2012,730,412sampleswere submittedtoscreeningtestoftheNSP-SES/SC.Duringthis period, 848,833 children were born in the state, accord-ingtoSINASC,whichindicatesaprogramcoverageof86.0% (Table1).
SCDwasidentifiedin 39children ofthe screened sam-ples.Ofthese,26casesshowedtheFSpattern(inorderof decreasingquantity,presenceofhemoglobinsFandS,inthe absenceofhemoglobinA);12FSC(presenceofhemoglobins F,Sand C,inthe absenceof hemoglobinA);andoneFSA (presenceofhemoglobinsF,SandA;Table2).Eachofthese samples was submitted to two tests --- high-performance liquid chromatography and isoelectric focusing --- with
Table1 Numberoflivebirths,screenedchildren,and per-centageofcoverageoftheNeonatalScreeningProgramof theHealthSecretariatoftheStateofSantaCatarina,from January2003toDecember2012.
Year Livebirthsa Screenedchildrenb Coverage
2003 83,177 67,993 81.7%
2004 85,475 80,243 93.9%
2005 84,584 80,333 95.8%
2006 84,133 68,063 80.9%
2007 81,903 69,925 85.4%
2008 85,262 80,172 94.0%
2009 83,489 73,437 88.0%
2010 84,611 61,997 73.3%
2011 87,481 78,127 89.3%
2012 88,772 70,122 78.0%
Total 848,833 730,412 86.0%
Nojustificationswerefoundforthelowcoveragein2010.
a LiveBirthInformationSystem(SistemadeInformac¸õessobre
NascidosVivos[SINASC]).7
b Central Laboratory (Laboratório Central [LACEN]) of the
HealthSecretariatoftheStateofSantaCatarina.
Table2 Numberofchildrenwithsickle-celltraitand dis-ease diagnosedbythe NeonatalScreeningProgramofthe HealthSecretariatoftheStateofSantaCatarina,from Jan-uary2003toDecember2012.
Year FASpattern FSpattern FSCpattern FSApattern
2003 491 3 2 0
2004 666 5 1 0
2005 655 3 2 1
2006 550 3 2 0
2007 607 0 0 0
2008 692 2 3 0
2009 659 6 1 0
2010 509 1 0 0
2011 683 1 0 0
2012 661 2 1 0
Total 6173 26 12 1
Source:CentralLaboratory(LaboratórioCentral[LACEN])ofthe HealthSecretariatoftheStateofSantaCatarina.
equivalentresults.Whethertherewasfurtherconfirmation oftheresultswithotherdiagnostictestswasnotassessed.
The resultwasindeterminatein 302samples, i.e.,the hemoglobin was different from hemoglobins that can be diagnosedby neonatalscreening methods. These children werereferredtotheHIJGfordiagnosticclarification.
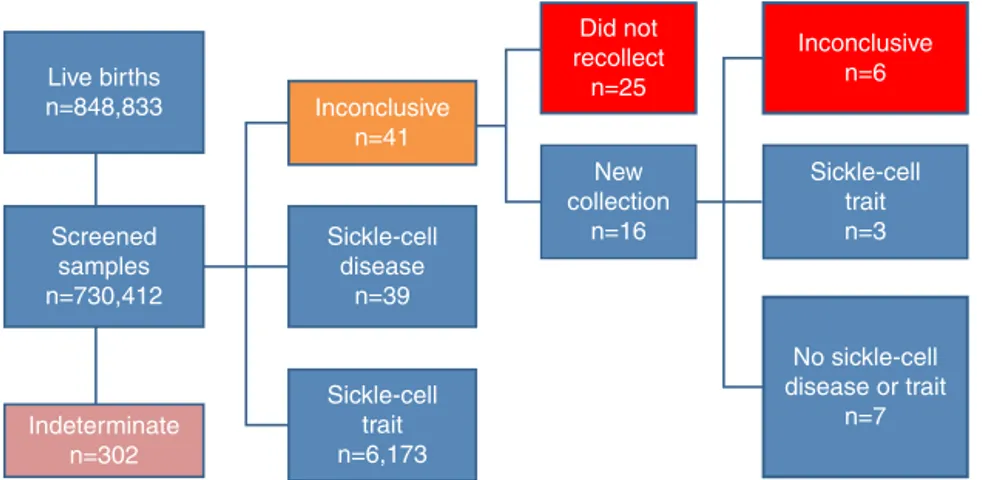
OfthesamplesanalyzedbytheNSP-SES/SC,41showed inconclusiveresults and25childrenwiththeseresultsdid notundergoasecondtest,despitetheprogram’s recommen-dation.Oftheremaining16childrenthathadanewsample collected,sixonceagain showedan inconclusivepattern, sevenshoweda normalpattern, andthreehadsickle-cell traitpattern.Thus,31childrenwithaninconclusivepattern didnothaveadefinitediagnosis(Fig.1).
Regardingtheorigin, theMidwestandHighland macro-regionshadthehighestpercentagesofSCDcases(Table3). Regardingtheskin color/ethnicityofchildren withSCD (patternsFS,FSC,andFSA), 25(64.1%)werewhite;seven wereblack,andsevenwereofethnicity/skincolornot spec-ifiedinthesamplecollectionform.Asforgender, 24were malesand15werefemales. Themeanage onthesample collectiondaywas9.08daysandthemedianwas6days.
Discussion
Themainresultofthisstudywastoconfirmthefrequency ofSCD(1:18,728)andsickle-celltrait(1:118)inthe State ofSantaCatarina,aswellasthecoverageoftheneonatal screeningprograminthestate(86.0%).
TheincidenceofSCDwaslow,possiblyduetothesmall proportionofAfricandescendantsintheStateofSanta Cata-rina. Blacks, according to IBGE,accounted for only 2.2%, andmixed-race,11.7%of thestatepopulation intheyear 2009.5The highestSCDincidenceoccurredintheMidwest andHighlandmacro-regionsofSantaCatarina.
Live births n=848,833
Screened samples n=730,412
Indeterminate n=302
Inconclusive n=41
Inconclusive n=6 Did not
recollect n=25
New collection
n=16 Sickle-cell
disease n=39
Sickle-cell trait n=6,173
Sickle-cell trait n=3
No sickle-cell disease or trait
n=7
Figure1 Numberoflivebirths,screenedsamples,andrespectivediagnosesattheNSP-SES/SC,fromJanuary2003toDecember 2012.
inParanábetween2002 and2004showedan incidenceof 1:20,320.11
The observed coverage may not reflect the actual screening coverage of all live births.Even though all the municipalitiesinthestate havejoinedthisprogram,some childrenmayhavebeenscreenedintheprivatehealthcare network,whichdoesnotgeneraterecordfortheNSP-SES/SC andreflectsafailureofthestate’smonitoringprogram.
In studies conducted in Brazil, it was observed that the program coverage in the Federal District was 83.4% in 200612; in Bahia, 88.9% between the years 2007 and 200913; and in Mato Grosso do Sul, 91.8% between 2006 and 2010.14 The goal of 100% coverage of the neonatal screeningprogramsisstillachallenge,eveninmore devel-oped countries.9 Canada and Belgium, in 2006, achieved coverageof76%15and87%,16 respectively.
Among the children with SCD in the period analyzed inthis study,the meanage onsample collectionday was 8.58daysandthemedianwas6days,showingtheneedto improvethesevariables, astheMinistry ofHealth recom-mendsthatthe collectionbeperformed between3 and5 daysoflife.Althoughthetestcanbeperformedlater,itis
ofutmostimportancetocollectthesamplewithintheideal periodforsomeofthescreeneddiseases,inordertoprovide earlydiagnosisandtreatment,aswellastobenefitfromthe preventionofsequelae.
Ahighernumberofwhitepatientswasobserved(64.1%) amongthe39 SCDcases.The higherincidenceof the dis-ease in whites canbe explainedby the highernumber of theseindividualsamongtheStateofSantaCatarina’s pop-ulation,which,accordingto2009IBGEdata,accountedfor 85.7%ofthetotalpopulation,5inadditiontothewhite eth-nicity being misattributed to the mixed-race population. Thedatalimitationregardingskincolor/ethnicityoriginates fromthefactthatthisinformationisfilledoutbythe per-son whocompletedthe bloodsample formand notbased onadetailedfamilybackgroundquestionnaire,considering theinterpretativedisagreementbetweenskincolorand eth-nicityofaparticularindividual.Itisessential,however,to be awarethat none of theselimitations affect the major aspectsassessedinthisstudy.
Regardingchildrenwithinconclusiveandindeterminate results,itisimportanttocreatemechanismstointensifythe searchfortheseindividuals,aimingtoexpandtheprogram’s
Table3 Distributionof sickle-cell disease per healthmacro-region ofthe StateofSanta Catarina, from January 2003to December2012.
Healthmacro-region Numberofcasesof sicklecelldiseasea
Numberoflivebirths perregion
Incidence perregion
Great-West 0 97,705 0
Midwest 8 86,921 1:10,865
Northhighlands 1 56,624 1:56,624
Northeast 8 122,663 1:15,333
ItajaíValley 1 120,420 1:120,420
Itajaíriverestuary 5 76,502 1:15,300
Highland 4 42,259 1:10,565
GreaterFlorianópolis 10 129,362 1:12,932
South 2 116,427 1:58,213
Total 39 848,883 1:18,728
Source: LiveBirthInformationSystem(SistemadeInformac¸õessobreNascidosVivos[SINASC])7andCentral Laboratory(Laboratório Central[LACEN])oftheHealthSecretariatoftheStateofSantaCatarina.
scope andto identifyand provideearly treatment to any SCDcarriers---whichinthisstudywasofalowfrequency, consideringthenumberofscreenedsamples.
Onelimitationofthisstudyisduetolackofpatient mon-itoring afterdiagnosis through the neonatalscreening, in ordertoassess patient adherence tothe interdisciplinary outpatientfollow-upandhealthmeasuresproposedbythe program.Recently,astudyperformedinMinasGeraisfound thatneonatalscreeninginthatstate,evenifcarriedoutin acomprehensiveandeffectivemanner,wasnotenoughto reduceSCDmortality,whichwas7.4%inthefirst14years, withslightlymorethanhalfofthedeathsoccurringbefore 2yearsofage.17
ThedrasticreductioninmortalityrelatedtoSCDinthe earlyyearsoflife,from26%to1---2%,occurreddueto com-prehensive care provided to this population at pediatric referralcenters.Therefore,earlydiagnosisandtreatment ofSCDhelpstoincreasesurvivalandsignificantlyimproves thequalityoflifeoftheseindividuals.6,18
Thus,itcanbeconcludedthattheNSPhasgoodcoverage inSantaCatarinaandthattheincidenceofSCDinthestate isquitelow.Nevertheless,itisessentialtoassesspatients’ adherencetohealthmeasuresrecommendedbytheNSP,as withoutthisfollow-up,thediagnosisalonebecomes point-less.Withtheinformationprovidedbythisstudy,theauthors intendtoseektheaffectedchildrenandassessadherence tooutpatientmedicalfollow-up,aswellasthehealth sup-portofferedtopatients,inordertoactually evaluatethe program’sefficacy.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
References
1.Lobo CL, Ballas SK, Domingos AC, Moura PG, do Nasci-mentoEM,CardosoGP,etal.Newbornscreeningprogramfor hemoglobinopathiesinRiodeJaneiro,Brazil.PediatrBlood Can-cer.2014;61:34---9.
2.CamposJúnior D,BurnsDA, LopezFA. Tratadode pediatria: Sociedade Brasileira de Pediatria. 3rd ed. Rio de Janeiro: Manole;2014.
3.Kliegman RM. Nelson tratado de pediatria. 14th ed.Rio de Janeiro:Elsevier;2014.
4.GoldmanL,AusielloD.Cecilmedicina.23rded.RiodeJaneiro: Elsevier;2009.
5.Instituto Brasileiro de Geografia e Estatística. Síntese de indicadores sociais: uma análise das condic¸ões de vida da populac¸ãobrasileira 2010. Availablefrom: http://biblioteca. ibge.gov.br/visualizacao/livros/liv45700.pdf[cited01.09.14]. 6.HasselKL.PopulationestimatesofsicklecelldiseaseintheU.S.
AmJPrevMed.2010;38:S512---21.
7.Tabnet. Departamento de Informática do Sistema Único de Saúde (DATASUS). Available from: http://tabnet.datasus. gov.br/cgi/deftohtm.exe?sinasc/cnv/nvsc.def[cited01.09.14]. 8.AlmeidaA,GodinhoTM,TelesMS,RehemAPP,JalilHM,Fukuda TG,etal.Avaliac¸ãodoprogramadetriagemneonatalnaBahia noanode2003.RevBrasSaúdeMaternInfant.2006;6:85---91.
9.BotlerJ,Camacho LA, CruzMM.Phenylketonuria,congenital hypothyroidismandhaemoglobinopathies:publichealthissues foraBraziliannewbornscreeningprogram.CadSaudePublica. 2012;28:1623---31.
10.Sommer CK, Goldbeck AS, Wagner SC, Castro SM. Triagem neonatalparahemoglobinopatias: experiênciade um anona redepúblicadoRioGrandedoSul,Brasil.CadSaudePublica. 2006;22:1709---14.
11.Watanabe AM, Pianovski MA, Zanis Neto J, Lichtvan LC, Chautard-Freire-MaiaEA,DomingosMT, etal.Prevalênciada hemoglobinaSnoestadodoParaná,Brasil,obtidapelatriagem neonatal.CadSaudePublica.2008;24:993---1000.
12.DinizD,GuedesC,BarbosaL,TauilPL,MagalhãesI.Prevalência dotrac¸ofalciformeedaanemiafalciformeemrecém-nascidos do DistritoFederal,Brasil,2004 a 2006. Cad SaudePublica. 2009;25:188---94.
13.Amorim T, Pimentel H, Fontes MIMM, Purificac¸ãoA, LessaP. Avaliac¸ão do Programa de Triagem Neonatal na Bahia entre 2007e2009---Aslic¸õesdadoenc¸afalciforme.GazMedBahia. 2010;80:10---3.
14.IvoML,Araujo OM,BarbieriAR, CorrêaFilhoRA, PontesER, deOliveiraBotelhoCA.Scope andefficiencyofthenewborn screeningprograminidentifyinghemoglobinaS.RevBras Hema-tolHemoter.2014;36:14---8.
15.TherrellBL,AdamsJ.NewbornscreeninginNorthAmerica.J InheritMetabDis.2007;30:447---65.
16.LoeberG.NeonatalscreeninginEurope:thesituationin2004. JInheritMetabDis.2007;30:430---8.
17.Sabarense AP, Lima GO, Silva LM, Viana MB. Characteriza-tionofmortalityinchildrenwithsicklecelldiseasediagnosed through the Newborn Screening Program. J Pediatr (Rio J). 2015;91:242---7.