w w w . r e u m a t o l o g i a . c o m . b r
REVISTA
BRASILEIRA
DE
REUMATOLOGIA
Original
article
Characterization
of
inflammatory
markers
associated
with
systemic
lupus
erythematosus
patients
undergoing
treatment
Rodolfo
Pessato
Timóteo
∗,
Douglas
Cobo
Micheli,
Reginaldo
Botelho
Teodoro,
Marlene
Freire,
Dernival
Bertoncello,
Eddie
Fernando
Candido
Murta,
Beatriz
Martins
Tavares-Murta
UniversidadeFederaldoTriânguloMineiro,Uberaba,MG,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received13March2015 Accepted20December2015 Availableonline11March2016
Keywords:
Systemiclupuserythematosus Cytokines
Phagocytosis Integrin CXCR2
a
b
s
t
r
a
c
t
Objective:Tocharacterizetheinflammatoryprofilesofpatientswithsystemiclupus erythe-matosusreceivingstandardtreatmentcomparedtohealthycontrols.
Patientsandmethods:Peripheralvenousbloodwascollectedfromsystemiclupus erythe-matosus patients (n=14) and controls(n=18) atenrollment. Bloodsampleswere used forquantification,byflowcytometry,ofCD11b(integrin)andChemokinereceptorCXCR2 expressionsurfaceantigeninneutrophilsandlymphocytes,whilecytokineswereassayed inserumsamples.Purifiedneutrophilswereassayedbytheirabilitytophagocytizehuman plasma-opsonizedzymosan.
Results:Patientshadamedian(interquartilerange)diseaseactivityindexof1.0(0–2.0) char-acteristicofpatientsinremission.Interleukin-6andinterleukin-10serumconcentrations weresignificantlyhigherinthepatientgroupcomparedtocontrolsandthephagocyticindex ofcirculatingneutrophilswassignificantlyreducedinpatientscomparedtocontrols.The levelsofinterleukin-2,interleukin-5,interleukin-8andtumornecrosisfactoralphadidnot significantlydifferbetweenpatientsandcontrols.Flowcytometricanalysisrevealedthatthe integrinexpressionlevelswerereducedinlymphocytes(butnotinneutrophils)obtained fromsystemiclupuserythematosuspatients,whilesurfaceexpressionofthechemokine receptor2wassimilarinbothneutrophilsandlymphocytes.
Conclusion: Systemiclupuserythematosuspatientsreceivingstandardtreatmentpresented withelevatedsystemiclevelsofinterleukin-6andinterleukin-10,reducedneutrophil phago-cyticcapacity,andreducedlymphocyteexpressionofintegrinevenwhensymptomswere inremission.Thesealterationstoinnateimmunecomponentsmayputtheseindividuals atagreaterriskforacquiringinfections.
©2016ElsevierEditoraLtda.ThisisanopenaccessarticleundertheCCBY-NC-ND license(http://creativecommons.org/licenses/by-nc-nd/4.0/).
∗ Correspondingauthor.
E-mail:[email protected](R.P.Timóteo). http://dx.doi.org/10.1016/j.rbre.2016.02.009
Caracterizac¸ão
de
marcadores
inflamatórios
associados
a
pacientes
com
lúpus
eritematoso
sistêmico
em
tratamento
Palavras-chave:
Lúpuseritematososistêmico Citocinas
Fagocitose Integrina CXCR2
r
e
s
u
m
o
Objetivo:Caracterizarosperfisinflamatóriosdepacientescomlúpuseritematososistêmico queestavamrecebendotratamentopadrãoemcomparac¸ãocomcontrolessaudáveis.
Pacientesemétodos:Coletou-seosanguevenosoperiféricodepacientescomLES(n=14)e con-troles(n=18)nomomentodaentradanoestudo.Asamostrasdesangueforamusadaspara quantificac¸ão,porcitometriadefluxo,daexpressãodosantígenosdesuperfícieCD11b (inte-grina)edoreceptordequimiocinaCXCR2emneutrófiloselinfócitos,enquantoascitocinas foramavaliadasemamostrasdesoro.Avaliou-seacapacidadedosneutrófilospurificados defagocitarzimosanopsonizadocomplasmahumano.
Resultados: Ospacientesapresentavamumapontuac¸ãomediana(intervalointerquartil)no índicedeatividadedadoenc¸ade1,0(0–2,0),característicadepacientesemremissão.As concentrac¸õesséricasdeinterleucina6einterleucina10foramsignificativamentemaiores nogrupodepacientesemcomparac¸ãocomoscontroles;oíndicedefagocitosede neu-trófiloscirculantesestavasignificativamentereduzidonospacientesemcomparac¸ãocom oscontroles.Osníveisdeinterleucina2,interleucina5,interleucina8efatordenecrose tumoralalfanãodiferiramsignificativamenteentrepacientesecontroles.Aanáliseda cit-ometriadefluxorevelouqueosníveisdeexpressãodaintegrinaestavamreduzidosnos linfócitos(masnãonosneutrófilos)obtidosdepacientescomlúpuseritematososistêmico, enquantoaexpressãodoreceptordesuperfíciedequimiocinaerasemelhanteem neutró-filoselinfócitos.
Conclusão: Ospacientescomlúpuseritematososistêmicorecebendotratamentopadrão apresentaramníveissistêmicoselevadosdeinterleucina6einterleucina10,reduc¸ãona capacidade fagocítica dos neutrófilose expressão reduzida da integrinaem linfócitos, mesmoquandoossintomasestavamemremissão.Essasalterac¸õesnoscomponentesda imunidadeinatapodemcolocaressesindivíduosemmaiorriscodeadquiririnfecc¸ões.
©2016ElsevierEditoraLtda.Este ´eumartigoOpenAccesssobumalicenc¸aCC BY-NC-ND(http://creativecommons.org/licenses/by-nc-nd/4.0/).
Introduction
Systemic lupus erythematosus (SLE) is an autoimmune diseaseinvolving bothT-cellandB-cellabnormalities char-acterized by the loss of tolerance to nuclear self antigens andtheproductionofautoantibodiesthatcause inflamma-tionanddamagetomultipleorgansystems.1Anincreasing
arrayofcytokineabnormalitieshasbeenimplicatedineither thepathogenesisofSLEorassecondarymarkersthatreflect immunedysregulation.2,3
Oneofthecharacteristicsofchronicinflammationis dys-regulationofthechemokinenetwork,includingchangestothe expressionprofilesofCXCR2andCXCR3.CXCR2isaG-protein coupledreceptorbelongingtotheCXCchemokinefamilythat servesasareceptorforIL-8(CXCL8)andCXCL1thatmediates neutrophilrecruitment,cellproliferation,andangiogenesis.4,5
TheCD11b/CD18integrin(Mac-1,complementreceptor3) isasurfacereceptorexpressedbymonocytes,macrophages, neutrophils, dendritic cells, and B cell subsets that binds severalligandsincluding membersoftheICAMfamily and the complement factor iC3b. It is involved in essential immunological processesincluding leukocyte extravasation andphagocytosis.Asinglenucleotidepolymorphism inthe integrin extracellular domain resulting in an amino acid changerepresentsone ofthe strongestgeneticriskfactors associatedwithhumanSLE.6,7
Thevariantformisassociatedwithreducedcelladhesion to ICAM-1, ICAM-2, and iC3b and with impaired phagocy-tosis, suggesting that this polymorphism (that affects the efficient removalof apoptotic cells) isassociatedwith dis-ease pathology. Changes to phagocytosis efficacy are not restricted to CD11b+/CD18+ macrophagesbut also to other
phagocytic cells suchas monocytes and neutrophils.7
Fur-thermore, this polymorphism is associated with impaired regulationofinflammatorycytokinenetworkspossibly asso-ciatedwithSLEdiseaseprogression.6,8
Theimpairedleukocytefunction(i.e.,monocytesand neu-trophils) observed inSLE patients places these individuals at increased risk for acquiring bacterial infections associ-ated with increased rates of morbidity and mortality.9 In
addition todecreasedphagocytosisofmicrobialpathogens, impairedclearanceofapoptoticcellsnegativelyimpactstissue homeostasisbyexposingtheimmunesystemto intracellu-larcomponentsthatarepro-inflammatoryandimmunogenic, therebycontributingtothedevelopmentofchronic inflamma-tionandautoimmunedisorders.10
TreatmentwithprednisolonefailedtomodifytheCD11b expression on peripheral blood neutrophils in patients with ulcerative colitis compared with controls.11 However
dexamethasone inhibited membrane, but not intracellular, expression of CD11b on murine eosinophils, associated to reducedcellchemotaxis.12ConsideringtheCD11b
on functional leukocyte capacity, patients with LES under treatmentmaybesusceptibletofunctionaldeficit in circu-latingimmunecells.
The present study was designed to further character-ize the inflammatory response presenting in SLE patients undergoingtreatment by comparingthe systemiccytokine response,neutrophilphagocyticcapacity,andtheCD11band CXCR2neutrophilandlymphocyteexpressionprofilesinboth patientsandcontrols.
Materials
and
methods
Patients
Women referred by the public health system to the Out-patient Rheumatology Service of the Federal University of TriânguloMineiro(UFTM)wereprospectivelydiagnosedwith SLEaccordingtoguidelinesestablishedbytheAmerican Col-legeofRheumatologyandenrolledinthestudy.Patientshad different times of SLE onset and were being treated with corticoids and/or hydroxychloroquine and/or azathioprine. Diseaseseveritywas establishedusingthe Systemic Lupus ErythematosusDiseaseActivityIndex(SLEDAI)scoresystem. Healthyfemalevolunteersservedascontrols.Thestudy pro-tocolwasapprovedbythe UFTMCommitteeon theUseof HumanSubjects,andwritteninformedconsentwasobtained fromallpatientsandcontrols.
Bloodcollection
Peripheralvenousbloodsampleswerecollectedfrompatients andcontrolsatenrollment.Ateachcollectiontimetwo sam-ples(5mLeach) were collected (withorwithout 100IU/mL heparin).Serawereobtainedbycentrifuging(180×g)theblood for15min.Respectiveserumsampleswerethencollectedand storedat−70◦Cuntiluse.Theassayswereperformedatthe
LaboratoryoftheDisciplineofPharmacologyandatthe Onco-logicalResearchInstitute(IPON)/DisciplineofGynecologyand Obstetrics.
Quantificationofserumcytokinelevels
The serum levels of TNF-␣, IL-2, IL-5, IL-6, IL-8, and IL-10weredetermined byELISA.Briefly,flat-bottomed96-well microtiterplates werecoatedwithantibodies(1–3g/mLin 50L/well) specificforeach cytokine inbindingbuffer and incubated overnight (4◦C). Plates were then washed with
phosphatebufferedsaline-Tween-20(PBST)andnonspecific bindingblockedbyincubatingwellswith100L/wellPBS/1% bovine serum albumin (BSA) for120minat 37◦C. Samples
and standards were added to respective wells (50L/well) andincubatedovernight(4◦C).PlateswerewashedwithPBST
followedbytheadditionoftheappropriatebiotinylated mono-clonalanti-cytokineantibody.After1h,plateswerewashed and avidin peroxidase (diluted 1:5000) added toeach well. After30minthe plates were washed and substrate (100L o-phenylenediaminedihydrochloride[OPD,Sigma,St.Louis, MO])wasadded.Theplatesweresubsequentlyincubatedat roomtemperaturefor15minbeforeH2SO4(50L, 1M)was
addedtoeachwelltostopthereactionandtheopticaldensity measured at490nm using amultiwell plate reader (Multi-skanMCC340MKII,FlowLaboratories).Dataareexpressedin picogramsofcytokine/mLserum.Cytokineconcentrationsin serum sampleswere determined fromthe standard curves generated.
CD11bandCXCR2expressionbyneutrophils andlymphocytes
CD11bandCXCR2expressionlevelswerequantifiedusinga FACSCaliburflowcytometerandCellQuestTMsoftware
(Bec-ton Dickinson, San Jose, CA), with acquisition of 10.000 cellsineachexperiment.Phycoerythrin(PE)-conjugated anti-CD11bantibody,PE/Cy5-conjugatedanti-CXCR2antibody(BD Pharmingen, SanDiego,CA),and controlantibodies(PE- or PECy-5-conjugatedIgG2b;BDPharmingen,SanDiego,CA)were
used.Dataareexpressedasthenumberofabsoluteorthe per-centageofneutrophilsandlymphocytespositive forCD11b and/orCXCR2expression.
Phagocytosisassay
Thephagocytosisindexofneutrophils(2×106/mL)harvested
from eitherSLE patients or healthy controlswas assessed by their ability to phagocytize human plasma-opsonized zymosan(10particles/cell)overa1hperiodat37◦C(5%CO
2).
Thephagocyticindexisexpressedasfollows:%ofphagocytic cells×numberofinteriorizedparticles.
Statisticalanalysis
Statistical analyses were performed using SigmaStat 2.03 software.DistributionwasevaluatedusingtheKolmogorov– Smirnovtestanddifferencesbetweentwounpairedgroups (SLE patients vs. controls) were determined using the Stu-dent’sttestortheMann–Whitneytestaccordingtonormal ornotnormaldistributionofthedata,respectively.Statistical significancewassetatp≤0.05.
Results
Studypopulation
Fourteen womendiagnosedwithSLE(meantime with dis-ease±SDof7.1±4.3y),meanage(±SD)of40.6±9.6y(range, 24–55)and18healthywomen,meanageof35.3±9.8y(range, 26–55)wereenrolledintothestudy(Table1).Agedidnot dif-ferbetweenpatientsandcontrols(p=0.132,Student’sttest). Themedian(andinterquartile)SLEDAIscorewas1.0(0–2.0), indicatingthatSLEwasinclinicalremission.Ineightpatients (57.2%)theSLEDAIscorewaszero,threepatients(21.4%)hada scoreof2,two(14.3%)patientshadascoreof4(moderate dis-easeactivity),andonepatient(7.1%)hadscoreof9(moderate activity)(Table1).
Table1–Systemiclupuserythematosus(SLE)patient characteristicsanddiseaseparameters.
Numberofpatients 14
Averageage±SD,years 40.6±9.6
Agerange 24–55
Sex(female/male),n 14/0
Diseaseduration(mean±SD),years 7.1±4.3
Diseasedurationrange,years 1–15
SLEDAIscore,medians(25–75%) 1.0(0–2.0)
SLEDAIscore,n(%)
Zero 8(57.2%)
2 3(21.4%)
4 2(14.3%)
9 1(7.1%)
Treatment,n(%)
Corticoids 3(21.4%)
Hydroxychloroquine 2(14.3%) Corticoidsplushydroxychloroquine 6(42.9%) Corticoidsorhydroxychloroquineplusazathioprine 3(21.4%)
Table2–SLEandcontrolserumcytokineconcentrations (pg/mL).
Cytokines(pg/mL) Controls(n=18) Patients(n=14)
IL-2 9.6(5.3–19.1) 7.3(6.5–12.8) IL-5 11.6(4.5–25.1) 4.4(2.8–7.7) IL-6 3.9(2.6–5.2) 6.8(5.2–8.5)a
IL-8 13.2(11.5–23.6) 21.3(17.2–31.3)b
IL-10 0.0(0.0–4.5) 13.5(0–17.2)c
TNF-␣ 0.0(0.0–3.5) 1.3(0.2–3.1)
Dataareexpressedasmedians(interquartilerange). a p<0.01.
b p=0.115comparedtothecontrolgroup(Mann–Whitneytest). c p<0.05.
corticoidsandhydroxychloroquine,and3/14(21.4%)received azathioprinepluscorticoidorhydroxychloroquine.
Serumcytokineconcentrations
Serumcytokinelevelswerecharacterizedinbothpatientsand controls.ThemedianserumlevelsofTNF-␣,IL-2,andIL-5did notdiffersignificantlybetweengroups(Table2).IL-8levels wereelevatedinSLEpatientscomparedtocontrolsbutthe lev-elswerenotstatisticallysignificant(p=0.115).Buttheserum concentrationsofIL-6(p<0.01)andIL-10(p<0.05)were sig-nificantlyhigherinSLEpatientsatenrollmentcomparedto controls(Table2).
CD11bandCXCR2expressionbyneutrophils andlymphocytes
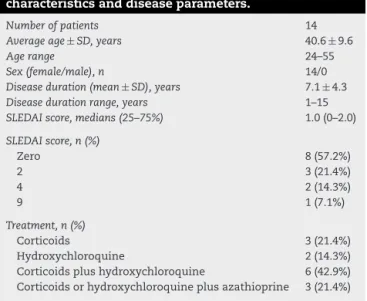
ThesurfaceexpressionofCD11bandCXCR2byneutrophils andlymphocytesharvestedfrompatients(n=14)anda sub-setofrandomlyselectedcontrols(n=16)wasassessedbyflow cytometry.Theabsolute number oflymphocytesharvested fromSLEpatientsexpressingCD11bwaslowercomparedto expressionlevelsobservedincontrols(Fig.1C).Nodifferences inCD11b expressionbyneutrophils were detected(Fig. 1A andB).ThenumberofCXCR2positivecellsdidnotdifferin
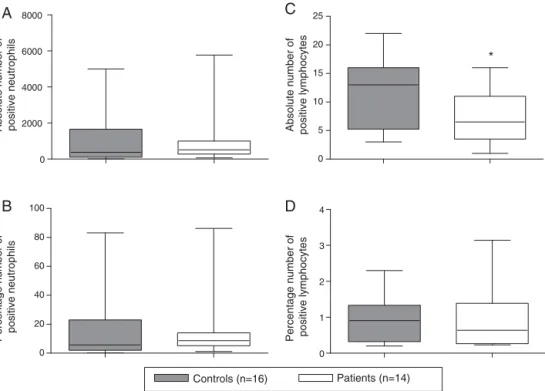
eitherneutrophils(Fig.2AandB)orlymphocytes(Fig.2Cand D)harvestedfrompatientsandcontrols.
Phagocytosisassay
The ability of neutrophils harvested from patients (n=14) andcontrols(n=18)tophagocytizeopsonizedzymosanwas evaluated. Neutrophils from SLEpatients presentedwitha significantly lower median (interquartile range) phagocytic activity index, 178.0 (49.0–304.0) compared to control neu-trophils,577.0(209.0–1131.0)(p=0.017,Mann–Whitney).
Discussion
Thepresentstudydefinedtheinflammatorycytokineprofile ofSLEpatientsundergoingtherapy.Asexpected,allpatients enrolledwerewomensinceSLEisninetimesmorecommonin womenthaninmen.1IncreasedserumlevelsofIL-6andIL-10
weredetectedinSLEpatientscomparedtocontrolsalthough thelevelsofTNF-␣,IL-2,IL-5andIL-8didnotdifferbetween groups.
The nucleosome is a major autoantigen that can be detected as a complex in the serum of SLE patients with the potential ofdirectly activating neutrophils.13 This was
confirmed by incubatingneutrophils from healthy individ-ualswithplasmaobtainedfromSLEpatientsthatresponded byproducingincreasedlevelsofpro-inflammatorycytokines suchasTNF-␣,IL-6,andIL-8.14
IL-6islikelytobeinvolvedinautoantibodyproductionin SLEpatients.3Datapresentedinthisreportdemonstrated
sig-nificantlyelevatedIL-6serumlevelsinSLEpatientscompared tocontrols,accordingtootherstudies.15,16Itwasreportedthat
the levels ofIL-6 correlated withthe IL-12and IL-23levels observedatthetimeofSLEdiagnosisandafterprednisone treatmentwasinitiated.15
Serum IL-8 levels were elevated in lupus patients but notsignificantly.SLEpatientswithactiveorinactivedisease presentedwithelevatedlevelsofchemoattractantproteins, including IL-8,comparedtocontrols,regardlessofthedrug treatmentregimenusedandthedegreeoftissuedamage.17
The authors suggest that long-termtreatment ofSLEwith standardimmunomodulatorydrugregimensfailsto normal-izethelevelsofkeychemoattractantproteinslinkedtoinnate immunityandthatabaselinepro-inflammatorystatepersists inSLEpatients.17
Highsystemicconcentrationsofchemokinessuchas IL-8havethepotentialofinducingreceptordesensitizationand renderingleukocytesunresponsivetosubsequentexposure.18
Our presentdatadidnotidentifydifferencesinthe CXCR2 expressionlevelsoncirculatingneutrophilsorlymphocytes, maybe,inpart,becauseserumIL-8levelsdidnotreachhigher significantlevelsinpatientscomparedtocontrols.Butother studieshavedemonstratedthatneutrophilsfromSLEpatients had decreased CXCR2 gene expressionlevels, comparedto controls, leading to IL-8hypo-responsiveness in vitro.5 The
0 2000 4000 6000 8000
A
Controls (n=16) Patients (n=14)
0 5 10 15 20 25
*
C
0 20 40 60 80 100
B
0 1 2 3 4
D
Percentage number of positive lymphocytes Percentage number of positive neutrophils
Absolute number of positive neutrophils Absolute number of positive lymphocytes
Fig.1–AnalysisofCD11bexpression.Neutrophilsobtainedfromeithercontrols(n=16)orSystemiclupuserythematosus (SLE)patients(n=14)undertreatmentwereanalyzedbyflowcytometry.Theabsolutenumbers(panelsAandC)and percentages(panelsBandD)ofneutrophils(panelsAandB)andlymphocytes(panelsCandD)positiveforCD11bare
shown.The25thand75thpercentilesarerepresentedbyabarcenteredaroundthemedian.Theminimumandmaximum
valuesarerepresentedbyerrorbars.*p<0.05comparedtotherespectivecontrol(Student’sttest).
0 2000 4000 6000 8000 10 000
A
Controls (n=16) Patients (n=14)
0 100 200 300 400 C
Absolute number of positive lymphocytes
0 50 100 150
B
0 5 10 15
D
Percentage number of positive lymphocytes Absolute number of positive neutrophils
Percentage number of positive neutrophils
Fig.2–AnalysisofCXCR2expression.Neutrophilsobtainedfromeithercontrols(n=16)orSLEpatients(n=14)under
treatmentwereanalyzedbyflowcytometry.Theabsolutenumbers(panelsAandC)andpercentages(panelsBandD)of
neutrophils(panelsAandB)andlymphocytes(panelsCandD)positiveforCXCR2areshown.The25thand75thpercentiles
TheroleofIL-10inSLEissomewhatcontroversial.IL-10is generallyconsideredananti-inflammatoryandan immuno-suppressivecytokine.However,it isoverexpressedinlupus patientsandinsomecaseshasbeenreportedtoactasa lupus-promotingmolecule.2Severalstudieshavedemonstratedthat
the serum/plasmaIL-10 levels were markedlyincreased in SLEpatientscomparedtocontrols,19asshowninthepresent
study.
IncreasedIL-10levelshavebeen correlatedwithdisease activity or with the productionof autoantibodiesthat can contributetoSLEpathogenesis19and/ormodulatethe
differ-entiation andfunction ofdendriticcells.Monocyte-derived dendritic cells activated by either SLE serum or following exposuretoexogenousIL-10hadreduced expressionlevels ofhumanleukocyteantigen(HLA)-DRandexhibitedimpaired capacitytostimulateallogenicT-cellproliferation.20
Cells expressing a CD11b polymorphism displayed increased IL-6 production compared to wild type-CD11b-expressingcellsand thisgeneticpolymorphism resultedin an integrin isoform incapable of mediating cell adhesion
viainteractions witheitherICAMsoriC3b.6Data presented
inthisreportshowedthat theCD11bexpressionprofileon neutrophilswassimilarbetweenSLEpatientsandcontrols; however, a reduced absolutenumber oflymphocytes from SLEpatientsexpressedCD11b.Thisdeficiencymayultimately leadtoimmune systemmalfunction and contributetothe developmentofSLE.6
Thepresentstudyalsodetectedthattheneutrophil phago-cytic index was reduced in SLE patients compared with controls,afindingthat,associatedtoreducedexpressionof CD11b,mayputtheseindividualsatagreaterriskfor devel-opinginfections.Continuousexposuretonucleosomesmay partlyexplaintheobservedreductioninactivity associated withpolymorphonuclearneutrophis(PMN)isolatedfromSLE patients,aswellasthereducedPMNcountsandthegeneral stateofinflammationobservedintheseindividuals.14Ithas
beenreportedthatlupuspatientspresentedwithlow-density circulatingneutrophilshaveanalteredphagocytic potential despitetheir proinflammatory phenotype(defined bytheir cytokineprofile).21
A study comparing the function of neutrophils among pediatric-onset SLE patients and healthy subjects demon-stratedthat regardlessofinfection status, medication,and disease severity, SLE patients had an impaired ability to phagocytizeSalmonella-specificlipopolysaccharidescompared tocontrols.9Inaddition,macrophagesisolatedfrompatients
withuntreatedSLEphagocytizedapoptoticneutrophilsless effectivelythanneutrophilsharvestedfromhealthydonors,10
therebycontributingtothedevelopmentofchronic inflamma-tionandautoimmunity.
Ofnote, ourresultsdemonstrate, ata same timepoint, associatedalterationsinparametersoftheinnate immune response,evenwhenSLEpatientsweremainlywithinactive disease,pointingto apotentialmechanism for susceptibil-ity to infections. But a limitation of the present study is thesmallnumberofpatients.Also,althoughmostpatients haddiseaseinremission,thestudydesigndidnotevaluate the individual role ofdisease activity in the inflammatory profileofpatients.Additionalresearchwithalargerpatient population and with patient groups at the same grade of
diseaseactivitywouldfavoramoreaccuratecomparisonof results.
Inconclusion,SLEpatientsreceivingimmunosuppressive treatmentpresentedwithelevatedsystemiclevelsofIL-6and IL-10, reduced neutrophilphagocytic capacity, and reduced lymphocyteexpressionofCD11b.Theseassociatedfindings werepresentevenwhensymptomsweremainlyinremission, suggestingthatthesealterationstoinnateimmune compo-nentsmayputtheseindividualsatagreaterriskfordeveloping infections.
Funding
Thisstudy wasfundedbyFinanciadoradeEstudose Proje-tos(FINEP) (grantnumber 1086/06),Fundac¸ãode Amparoà Pesquisa doEstadode MinasGerais(FAPEMIG)(grant num-ber52-11),ConselhoNacionaldeDesenvolvimentoCientífico e Tecnológico (CNPq) (grant number 507783/2010-7), and Fundac¸ãodeEnsinoePesquisadeUberaba(FUNEPU)(grant number578/2008).
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1.LiuZ,DavidsonA.Taminglupus–anewunderstandingof pathogenesisisleadingtoclinicaladvances.NatMed. 2012;18:871–82.
2.ClarkDN,MarkhamJL,SloanCS,PooleBD.Cytokine inhibitionasastrategyfortreatingsystemiclupus erythematosus.ClinImmunol.2013;148:335–43.
3.DingHJ,GordonC.Newbiologictherapyforsystemiclupus erythematosus.CurrOpinPharmacol.2013;13:405–12. 4.ErikssonC,EneslättK,IvanoffJ,Rantapää-DahlqvistS,
SundqvistKG.Abnormalexpressionofchemokinereceptors onT-cellsfrompatientswithsystemiclupuserythematosus. Lupus.2003;12:766–74.
5.HsiehSC,WuTH,TsaiCY,LiKJ,LuMC,WuCH,etal. AbnormalinvitroCXCR2modulationanddefectivecationic iontransporterexpressiononpolymorphonuclear
neutrophilsresponsibleforhyporesponsivenesstoIL-8 stimulationinpatientswithactivesystemiclupus erythematosus.Rheumatology.2008;47:150–7.
6.MacPhersonM,LekHS,PrescottA,FagerholmSC.Asystemic lupuserythematosus-associatedR77Hsubstitutioninthe CD11bchainoftheMac-1integrincompromisesleukocyte adhesionandphagocytosis.JBiolChem.2011;286:17303–10. 7.Fossati-JimackL,LingGS,CortiniA,SzajnaM,MalikTH,
McDonaldJU,etal.PhagocytosisisthemainCR3-mediated functionaffectedbythelupus-associatedvariantofCD11bin humanmyeloidcells.PLOSONE.2013;8:e57082.
8.FagerholmSC,MacPhersonM,JamesMJ,Sevier-GuyC,Lau CS.TheCD11b-integrin(ITGAM)andsystemiclupus erythematosus.Lupus.2013;22:657–63.
9.WuSA,YehKW,LeeWI,YaoTC,KuoML,HuangB,etal. Impairedphagocytosisandsusceptibilitytoinfectionin pediatric-onsetsystemiclupuserythematosus.Lupus. 2013;22:279–88.
macrophagesofsystemiclupuserythematosuspatients. Lupus.2014;23:133–45.
11.YoshiyamaS,MikiC,OkitaY,ArakiT,UchidaK,KusunokiM. Neutrophil-relatedimmunoinflammatorydisturbancein steroid-overdosedulcerativecolitispatients.JGastroenterol. 2008;43:789–97.
12.LimLH,FlowerRJ,PerrettiM,DasAM.Glucocorticoidreceptor activationreducesCD11bandCD49dlevelsonmurine eosinophils:characterizationandfunctionalrelevance.AmJ RespirCellMolBiol.2000;22:693–701.
13.RönnefarthVM,ErbacherAI,LamkemeyerT,MadlungJ, NordheimA,RammenseeHG,etal.TLR-2/TLR-4-independent neutrophilactivationandrecruitmentuponendocytosisof nucleosomesrevealsanewpathwayofinnateimmunityin systemiclupuserythemathosus.JImmunol.2006;177:7740–9. 14.LindauD,RönnefarthV,ErbacherA,RammenseeHG,Decker
P.Nucleosome-inducedneutrophilactivationoccurs independentlyofTLR-9andendosomalacidification: implicationsforsystemiclupuserythematosus.EurJ Immunol.2011;41:669–81.
15.QiuF,SongL,YangN,LiX.Glucocorticoiddownregulates expressionofIL-12familycytokinesinsystemiclupus erythematosuspatients.Lupus.2013;22:1011–6. 16.Rodríguez-CarrioJ,PradoC,dePazB,LópezP,GómezJ,
Alperi-LópezM,etal.Circulatingendothelialcellsandtheir
progenitorsinsystemiclupuserythematosusandearly rheumatoidarthritispatients.Rheumatology.
2012;51:1775–84.
17.VegaL,BarbadoJ,AlmansaR,González-GallegoR,RicoL, JimenoA,etal.Prolongedstandardtreatmentforsystemic lupuserythematosusfailstonormalizethesecretionof innateimmunity-relatedchemokines.EurCytokineNetw. 2010;21:71–6.
18.RaghuwanshiSK,SuY,SinghV,HaynesK,RichmondA, RichardsonRM.ThechemokinereceptorsCXCR1andCXCR2 coupletodistinctGprotein-coupledreceptorkinasesto mediateandregulateleukocytefunctions.JImmunol. 2012;189:2824–32.
19.PengH,WangW,ZhouM,LiR,PanHF,YeDQ.Roleof interleukin-10andinterleukin-10receptorinsystemiclupus erythematosus.ClinRheumatol.2013;32:1255–66.
20.SunZ,ZhangR,WangH,JiangP,ZhangJ,ZhangM,etal. SerumIL-10fromsystemiclupuserythematosuspatients suppressesthedifferentiationandfunctionof
monocyte-deriveddendriticcells.JBiomedRes. 2012;26:456–66.