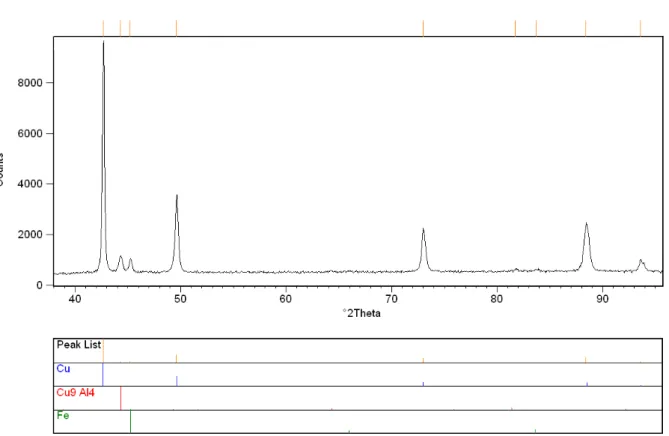
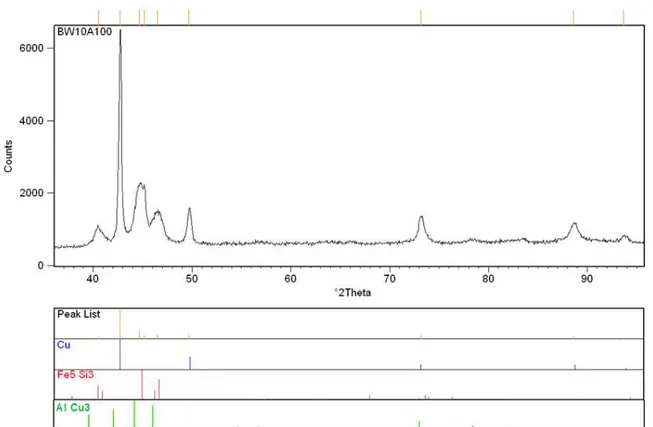
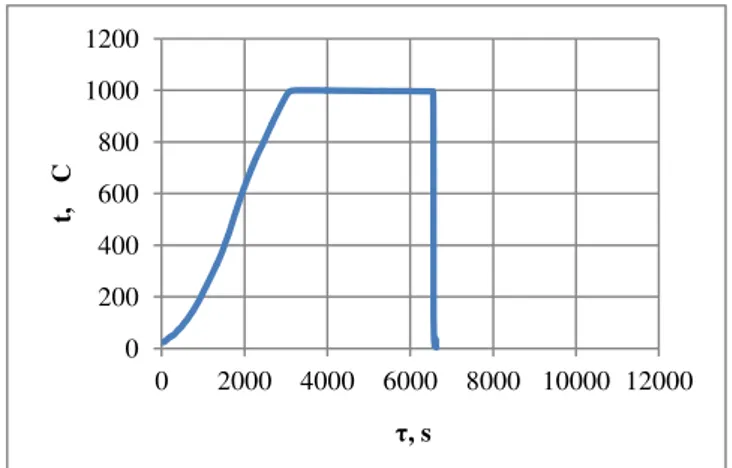
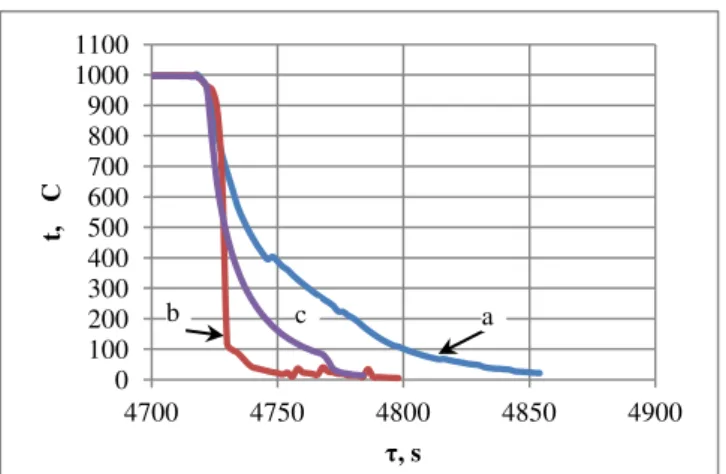
Effect of Annealing Time for Quenching CuAl7Fe5Ni5W2Si2 Bronze on the Microstructure and Mechanical Properties
Texto
Imagem
![Fig. 1. Vertical section through Cu-Al-5%Ni-5%Fe phase diagram [1]](https://thumb-eu.123doks.com/thumbv2/123dok_br/18299076.347472/2.871.544.742.125.423/fig-vertical-section-cu-al-ni-phase-diagram.webp)
![Fig. 2. Thermal conductivity and heat capacity Cp of the elements (a) [13] and copper and its alloys (b) [14]](https://thumb-eu.123doks.com/thumbv2/123dok_br/18299076.347472/3.871.82.789.289.996/fig-thermal-conductivity-heat-capacity-elements-copper-alloys.webp)
Documentos relacionados
Cerca de 2500 amostras de leite foram utilizadas para construção do espectro de referência e 1625 amostras foram utilizadas para validar a identificação dos cinco adulterantes
A infestação da praga foi medida mediante a contagem de castanhas com orificio de saída do adulto, aberto pela larva no final do seu desenvolvimento, na parte distal da castanha,
Na hepatite B, as enzimas hepáticas têm valores menores tanto para quem toma quanto para os que não tomam café comparados ao vírus C, porém os dados foram estatisticamente
Effect of volume fraction of phases and precipitates on the mechanical behavior of UNS S31803 duplex stainless
The probability of attending school four our group of interest in this region increased by 6.5 percentage points after the expansion of the Bolsa Família program in 2007 and
Steaming changes the colorimetric parameters of wood, mainly its luminosity, and proved to be able to lessen the difference between heartwood and sapwood color of Eucalyptus
Para o efeito, apresentam-se nos Quadro 7.2 a Quadro 7.6, para os 5 anos em análise dos relatórios e contas das três SADs objeto do nosso estudo, e para cada uma das