www.bjorl.org
Brazilian
Journal
of
OTORHINOLARYNGOLOGY
ORIGINAL
ARTICLE
Oral
changes
in
individuals
undergoing
hematopoietic
stem
cell
transplantation
夽
Regina
Haddad
Barrach
a,
Mair
Pedro
de
Souza
b,
Daniela
Polo
Camargo
da
Silva
a,∗,
Priscila
Suman
Lopez
a,
Jair
Cortez
Montovani
aaUniversidadeEstadualPaulista(UNESP),SãoPaulo,SP,Brazil bHospitalAmaralCarvalho,Jaú,SP,Brazil
Received5November2013;accepted27April2014 Availableonline22October2014
KEYWORDS
Mucositis;
Hematopoieticstem celltransplantation; Medicaloncology
Abstract
Introduction:Patientsundergoinghematopoieticstemcelltransplantationreceivehighdoses ofchemotherapyandradiotherapy,whichcausesevereimmunosuppression.
Objective: Toreportanoraldiseasemanagementprotocolbeforeandafterhematopoieticstem celltransplantation.
Methods:Aprospectivestudywascarriedoutwith65patientsaged>18years,with hemato-logicaldiseases,whowereallocatedintotwogroups:A(allogeneictransplant,34patients);B (autologoustransplant,31patients).Atotalofthreedentalstatusassessmentswereperformed: inthepre-transplantationperiod(moment1),oneweekafterstemcellinfusion(moment2), and100daysaftertransplantation(moment3).Ineachmoment,oralchangeswereassigned scoresandclassifiedasmild,moderate,andsevererisks.
Results:Themostfrequentpathologicalconditionsweregingivitis,pericoronitisinthethird molar region,and ulcersatthe thirdmomentassessments. However, atmoments 2and3, themostcommondisease wasmucositisassociatedwithtoxicityfromthedrugsusedinthe immunosuppression.
Conclusion: Mucositis accounted for the increased score and potential risk of clinical complications.Gingivitis,ulcers,andpericoronitiswereotherchangesidentifiedaspotential riskfactorsforclinicalcomplications.
© 2014Associac¸ãoBrasileira de Otorrinolaringologiae CirurgiaCérvico-Facial. Publishedby ElsevierEditoraLtda.Allrightsreserved.
夽
Please citethisarticleas:Barrach RH,de SouzaMP, daSilvaDP, Lopez PS,MontovaniJC. Oralchangesinindividuals undergoing hematopoieticstemcelltransplantation.BrazJOtorhinolaryngol.2015;81:141---7.
∗Correspondingauthor.
E-mail:[email protected](D.P.C.daSilva). http://dx.doi.org/10.1016/j.bjorl.2014.04.004
PALAVRAS-CHAVE
Mucosite; Sistema hematopoético; Oncologia
Alterac¸õesoraisemindivíduossubmetidosàtransplantedecélulashematopoiéticas
Resumo
Introduc¸ão:Pacientes submetidos a transplante de células hematopoiéticas recebem altas doses de quimioterapia eradioterapia que podem causarimunossupressão e doenc¸as orais graves.
Objetivo:Apresentarumprotocolodeavaliac¸ãodedoenc¸asoraisanteseapóstransplantede célulashematopoiéticas.
Método: Estudoclínicoprospectivode65pacientescomidadeacimade18anos,comdoenc¸as hematológicassubmetidas atransplantede células hematopoiéticas,divididos em dois gru-pos:A(transplantealogênico,34pacientes)eB(transplanteautólogo).Foramrealizadastrês avaliac¸õesodontológicas:períodoantesdotransplante(momento1),umasemana(momento 2)e100diasapósotransplante(momento3).Em cadamomentoasalterac¸õesoraisforam pontuadaseclassificadascomoleve,moderadaegrave.
Resultados: Asalterac¸ões orais mais frequentes foram: gengivite, pericoronite do terceiro molareúlceras.Entretantonosmomentosdoisetrêsaprincipaldoenc¸afoiamucositeassociada atoxicidadesdasdrogasusadasnaimunossupressão.
Conclusão:Mucositefoiprincipalalterac¸ão,comapontuac¸ãomaisaltaecommaiorriscode complicac¸ões.Gengivites,úlcerasepericoronitesforamoutrasalterac¸õescomriscomenorde complicac¸ões.
©2014Associac¸ãoBrasileira deOtorrinolaringologiaeCirurgiaCérvico-Facial.Publicadopor ElsevierEditoraLtda.Todososdireitosreservados.
Introduction
Hematopoietic stem cell transplantation (HSCT) is the replacement of a diseased or deficient bone marrow by normal cells, with the aim of reconstituting the marrow and providing immune system control. It is classified as autologouswhen bone marrow precursor cells come from thepatient,andasallogeneic, when thecells come from anotherperson(donor),whomayormaynotberelatedto therecipient.1,2
Patientswho will undergoHSCT are submitted tohigh dosesofchemotherapyandradiationtherapytoeradicate theunderlyingdisease,whichinducesanintense immuno-suppressionperiod knownasthe conditioning phase. This periodischaracterizedbypossibletissuedamageand infec-tionsduetoimmunosuppressivedrugtoxicity.3---5
Mucositisis oneofthemostcommonoraltissuelesions describedin theseimmunosuppressed patients,caused by thetoxicityofthesedrugs;itresultsinafragilityoftheoral mucosawithdecreasednumbersofbasalcells,andeventhe onsetofulcerations.6---8Itisconsideredthemostimportant oralcavitycomplicationinpatientsundergoingbonemarrow suppression,andisalsothemostcommon,withanincidence of90%.9,10Kolbinsonetal.11describedearlychangesinthe oral mucosa such as erythema, ulceration, and epithelial pseudomembraneformation thatappearbetween fiveand sevendays after theonset of chemotherapy andimprove afterthreeweeks.
Theselesionsrepresentasignificant riskfactorfor sys-temicinfections,particularlyinpatientswithneutropenia, and20---50% ofcasesofsepticemiaoriginatefromtheoral cavity.12---14 ForPuyal et al.,10 Köstler et al.,15 and Yama-gataetal.,16 theratesofoccurrenceoforallesionsvaried,
dependingonthetypeofunderlyingdisease,thetreatment usedandoralstatusbeforeconditioningfortransplantation, andreflectedmainlypre-existingoralconditions.
Lesions such as root fragments, periodontal pockets, periapicallesions,andremovabledenturesareconsidered reservoirsofopportunisticpathogensthatcantrigger infec-tionsduringimmunosuppression.9,17---19
Thesereports indicate that theprevention of oral dis-ease prior to HSCT is extremely important and that oral diseases shouldbetreatedbeforetransplantationto elim-inate potential risk factors for systemic infections.7,20---22 Dreizen et al.22 reported that 30---50% of patients under-going chemotherapy for oncologictreatment develop oral alterationsorlesions,andthatincidencecanbesignificantly reducedbypriororalintervention.However,wefoundfew studiesintheliterature,exceptthoseformucositis,which attemptedtoquantifyoralalterationsthatoccurinpatients undergoingbonemarrowtransplantation.Whentheauthors, appliedascoretothecondition,theyeliminatedthe subjec-tivenatureofthepotentialofthesealterationsforclinical complications.3,7,8,23---30 Thus,theobjectiveofthisresearch wastodevelopastandardizedmethodforthequantitative evaluationofalterationsorlesionsoftheoralcavity,andto identifythepotentialforclinicalcomplicationsinpatients submittedtoHSCT.
Methods
bothgenders,withorwithouthematologicalmalignancies, whounderwentHSCTandimmunosuppressioninthe above-mentioned period were included. Patients were grouped according to the type of HSCT: group A (allogeneic), 34 patients; and group B (autologous), 31 patients. Initially, allansweredaquestionnaire,whichcontainedinformation onoral health andoral hygienehabits. Subsequently, the patientswereevaluatedaccordingtothestatusoftheoral cavityat20days,onaverage,beforeconditioningandHSCT (moment1).Thesecond evaluationwascarriedoutinthe first week aftertransplantation (moment 2),and the last evaluation100daysafterHSCT(moment3).
Attheevaluationmoments(1,2,and3)scoresweregiven tothedifferentalterationsfoundintheoralcavity, accord-ingto theproposed dentalstandardization (Table 1).The score giventothe oral alterations wasbased onprevious work,whichgavegreaterorlesserdegreeofimportanceto thehistopathologicalalterationsoftheoralcavity.3,7---10,14,24 Theriskclassificationanddegreesoftoxicitywerebasedon theworkofPorak,14 Dreizenetal.,22andParulekanetal.24 Inmoment1,patientswereclassifiedaccordingtotherisk forcomplications:mild(upto15points),moderate(16---30 points),andsevere(31---50points)risk.Inmoments2and3, thedegreesoftoxicitywereclassifiedasfollows:mild (15 points),moderate(16---30points),andsevere(31---50points;
Table1).
Evaluationresultswerecomparedamongthemselvesand between groups. For the variables gender, age, disease, drugsfor conditioning,cell source,andtypeof transplan-tation,thechi-squaredorFisher’sexacttestwereusedfor comparisonofthegroups. Forthevariablesdentalcaries, gingival pockets, tooth mobility, tooth extractions, den-tures, orthodontic devices, oral lesions, bacterial, viral, fungal infections, and mucositis, the Mann---Whitney test was used. Comparison between groups at each evalua-tionwasperformed usingFriedman’s test andcomparison of moments within each group was performed using Fisher’s test. The level of significance was set at 5% (p<0.05).
Results
Aftertheassessmentoforalalterations,theclassificationof risksandtoxicityofpatientswasperformedbysummationof thescores(Tables2---4).Inthefirstassessment,before con-ditioning,most patients receivedamild risk classification inbothgroups.Theonlypatientclassifiedashaving moder-ateriskhadgingivalpockets>6mmandtoothmobility;that patient’sscorewas>16(Table5).
Inthesecondevaluation,ofthe34assessedpatientsin groupA,18hadmildtoxicity,15moderate,andonepatient hadseveretoxicity.IngroupB,ofthe31patientsassessed, 19 had mild toxicity, 11 moderate, and one patient had severe toxicity (Table 6). As it can be observed, the risk and toxicityclassification in both groups were performed according to the presence of mucositis and its degree of severity.
Inthethirdevaluation,asaresultofdeaths,28patients wereassessedingroupAand30patientsingroupB.Allbut onepatient hadmild toxicityin thisassessment, and one patientpersistedwithseveretoxicity(Table7).
Table1 Datastandardizationformwithscoresrelatedto changesintheoralcavity.
Caries:
Nocaries:()
Presenceof1---5decayedteeth:1point() Presenceof5---10decayedteeth:2points() Presenceof10---15decayedteeth:3points() Presenceofmorethan15decayedteeth:4points()
Gingivalpockets:pointspertooth,maximumof15points:
Normal(absenceofbleedingatperiodontalprobing):() Gingivalpocketsof3---5mm:1point()
Gingivalpocketsof5---8mm:2points() Gingivalpockets>8mm:3points()
Toothmobility:Classificationofperiodontaldiseasesand conditions,Armitage,1999:pointspertooth,maximum of15points:
Notoothmobility:()
Mild(1---2mmofclinicalattachmentloss):1point() Moderate(3---4mmofclinicalattachmentloss):2points() Severe(>5mm):3points()
Exodontics:pointspertooth,maximumof10points: Absenceofteethwithextractionindication:() Radicularremains:1point()
Semi-impacted/impactedthirdmolar,withhistoryof pericoronitis:1point()
Prostheses: Noprostheses:() Fulldentures:1point()
Removablepartialdentures:1point() Permanentdentures:1point() Denturesonimplants:1point()
Orthodonticappliances: Noorthodonticappliances:()
Presenceoforthodonticappliances:1point()
Orallesions:pointsperorallesion,maximumof10points. Noorallesions:()
Presenceoforallesions:1point()
Mucositis:WHOclassification: Nomucositis:()
MucositisgradeI:5points() MucositisgradeII:10points() MucositisgradeIII:15points() MucositisgradeIV:20points()
Infections: Noinfections:()
Presenceofbacterialinfection:10points() Presenceofviralinfection:10points() Presenceoffungalinfection:10points()
Table2 DistributionoforalabnormalitiesobservedinsubjectsfromgroupA. Oralalterations Moment1
n=34
Moment2
n=34
Moment3
n=28 Toothcaries n=19(59.4%) n=19(59.4%) n=18(66.7%) Gingivalpockets n=8(25.0%) n=8(25.8%) n=7(25.9%) Mobility n=5(15.6%) n=6(18.8%) n=5(18.5%) Toothloss n=10(31.2%) n=9(28.1) n=8(29.6%) Prostheses n=14(41.2%) n=14(41.2%) n=10(35.7%)
Orthodontics 0 0 0
Orallesions n=8(23.5%) n=16(47.1%) n=7(53.6%) Infections --- --- n=2(7.2%) Mucositis 0 n=34(100%) 0
Table3 DistributionoforalabnormalitiesobservedinsubjectsfromgroupB. Oralalterations Moment1
n=31
Moment2
n=31
Moment3
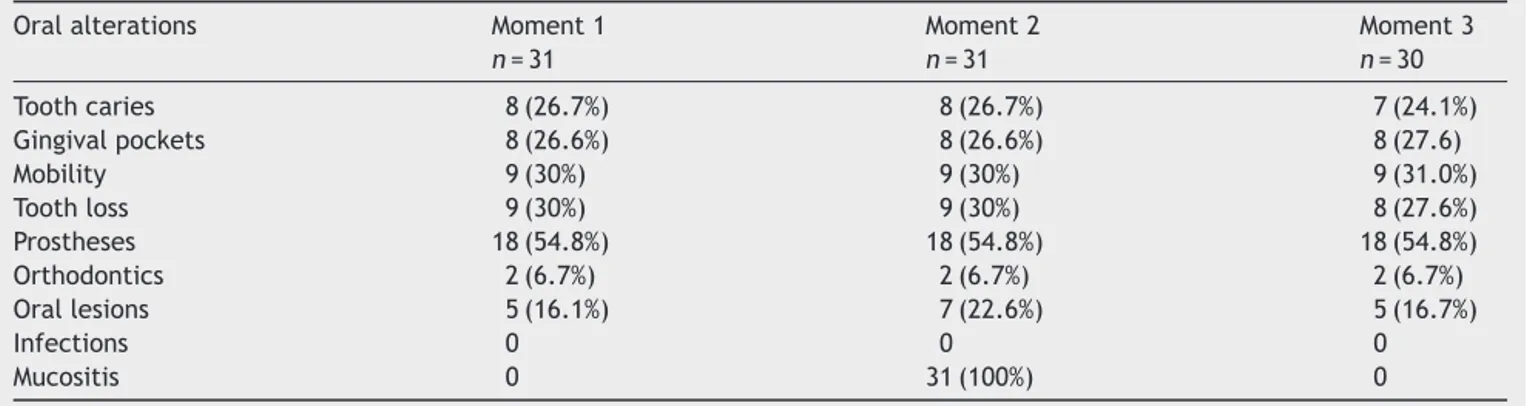
n=30 Toothcaries 8(26.7%) 8(26.7%) 7(24.1%) Gingivalpockets 8(26.6%) 8(26.6%) 8(27.6) Mobility 9(30%) 9(30%) 9(31.0%) Toothloss 9(30%) 9(30%) 8(27.6%) Prostheses 18(54.8%) 18(54.8%) 18(54.8%) Orthodontics 2(6.7%) 2(6.7%) 2(6.7%) Orallesions 5(16.1%) 7(22.6%) 5(16.7%)
Infections 0 0 0
Mucositis 0 31(100%) 0
Table 4 Patient distribution by group and grade of mucositis.
Gradeofmucositis Group
A B
n % n %
I 7 20.6 8 25.8 II 9 26.5 10 32.3 III 10 29.4 9 29.0 IV 8 23.5 4 12.9 Total 34 100.0 31 100.0
Table5 Distributionofpatientsbytransplantationgroup andriskatmoment1,p=0.34.
Riskatmoment1
Mild Moderate Total Group n % n % n % A 33 97.1 1 2.9 34 100.0 B 31 100.0 --- 0.0 31 100.0 Total 64 98.5 1 1.5 65 100.0
Table6 Distributionofpatientsbytransplantationgroup andtoxicityatmoment2.
Gradesoftoxicityatmoment2
Group Mild Moderate Severe
n % n % n %
A 18 52.9 15 44.2 1 2.9 B 19 61.3 11 35.5 1 3.2
2=0.50;p=0.78.
Table 7 Distributionofpatients according togroup and toxicityatmoment3.
Gradesoftoxicityatmoment3
Group Mild Moderate Severe
n % n % n %
A 27 96.4 --- 0.0 1 3.6 B 30 100.0 --- 0.0 --- 0.0
over time within group A (p=0.039). However, a statisti-cally significant difference wasobserved when comparing moment2ofgroupsAversusB,asmucositiswasobserved in 100% of individuals in both groups at these moments (Tables2and3).
Discussion
HSCTisawidelyusedtherapeutictechnique,aimingtocure oncologic patients and to provide disease control.1 How-ever, conditioning with chemotherapy drugs can resultin severaloralandsystemicalterationsinimmunosuppressed patients.3---6Thesemanifestationscanincreasethelengthof hospitalstay,increasetreatmentcosts,anddirectlyaffect thequalityandquantityoflifeofthesepatients.25
IntheautologousHSCT,comorbiditiesoccurmostoften due to the underlying disease activity, whereas in allo-geneicHSCT,theyareduetosystemiccomplicationsandthe graftversushostreactionitself.1,2,17,30Theintense immuno-suppression predisposes transplanted patients to severe infectionsthatcanoccuratanytimeoftransplantation,can becausedbydifferentetiologicalagentssuchasbacteria, fungi,viruses,orparasites.13,21
Establishing risk factors by assessing the status of the oral cavity and systemic complications of HSCT, and by assigningscorestothealterationsatdifferenttimesofthe clinicalfollow-up ofpatientssubmittedto immunoexpres-sion might facilitate the identification of individuals who would potentially have more clinical complications.8,16 A quantitative analysis using scores eliminates the subjec-tivenatureoftheobservations.24Inthepresent study,the developmentand utilization of scores for oral alterations were based on previous studies that sought toassociate, through scores or degrees of intensity, what changes or oraldiseasesassociatedwithtransplantationwould,in the-ory, have greater potential to be associated withclinical complications.14,16,19,22,24
Thecreationoftablesanddentalrecordswasmadebased onthefollowingvariables:dentalcaries, gingivalpockets, toothmobility,rootfragments,toothextractionindication, useofdentures,orthodonticdevices,andorallesions.
Alterations also included the specific complications of HSCT,suchastheoccurrenceofbacterial,fungalandviral infections,aswellasthedegreeofmucositis.8,14,22
Withrespecttodentalcaries,thepresentdataandthose intheliteratureshowednosignificantincreaseinnewdental caries in patients in the post-transplantation period and, astheyarenotrelevanttoHSCTcomplications,theyhave minimalscore.
The gingivalstatus andtoothmobility havea potential riskfactorforcomplications,especiallyperiodontalpockets greater than 6mm.26,27 Gingival pockets ofthis size favor greater accumulationof bacteriaand necrotictissue,and increase the risks for dental and oral disease; therefore, theyreceivedscoresofupto15,withintermediatevalues betweendentalcariesandmucositis.
Inbothgroupsofthepresentstudy,onlyonepatienthad largergingivalpockets,rangingfrom5mmto8mm,andwas classifiedatmoment1asmoderateriskandatmoments2 and3asseveretoxicity,duetothepresenceofdeepgingival pockets.
Anothercommon observation, in approximately80% of thepatients,wasgingivalhyperemia,probablyduetotoxic reactionstodrugsusedduringconditioning.Anotherlikely causeisoralmucosasensitizationduetoantiseptic mouth-wash(chlorhexidine)usedinoralhygiene. Patientsdonot brushtheirteethforfearthattraumacausedbythebrush cancausebleeding,duetothrombocytopenia.
However,the lackofbrushingcanresultinan increase inbacterialplaque,causinggingivitis, suchasthatseenin individualsatmoment2inbothgroups,withaconsequently greaterrisk ofbleeding. When thereisgingival inflamma-tion,plaquecan form morerapidly in thosesites than in non-inflamedonesandthus,mouthwashwouldbeless effec-tivefororalhygiene.However,inthepresentsample,these alterationswerebarelyobserved,whichwasattributedto moreeffectiveactionsbeforetransplantationbythe treat-inginstitution.1,2,10,16,17
Findings such as tooth extractions and semi-impacted thirdmolarswithahistoryofpericoronitisarementionedin theliteratureashavinglittlepotentialassourceof oppor-tunisticpathogensand,astheywereseldomobserved,they werenotscored.21---23
Semi-impacted third molars were observed in eight patientsfromgroupA andninefromgroupB.At moment 2,togetherwithmucositislesions,therewasformationof plaqueandgingivaoverlyingthetooth.Forthistypeof find-ing,andforprosthesesandorthodonticdevices,1pointwas scored.Shulmanetal.28 reportedthatorthodonticdevices anddenturesareriskfactorsforstomatitis.
Evenwiththeknowledgeofthisreportofpossiblehealth risks for individuals wearing prostheses, it was not pos-sible to establish statistically significant data correlating them withoral alterations.29 But since we knew that the oralcavity isthe gatewayof infectionsin patients under-goingimmunosuppressionforbonemarrowtransplantation andthatthe useof orthodonticdevices anddenturescan potentiatethem,weaskedourpatientsnottowearthem, ifpossible.21Theserecommendationsweremadebeforethe first evaluation, and were the reasons for the paucity of alterationsatmoment1.Althoughthatwasnotthepurpose ofthisstudy,wedemonstratedtheimportanceofprevention andtreatmentoforaldiseasestolessentheriskofclinical complicationsinimmunosuppressedindividuals.8,21Thiswas alsotheopinionofSonisandKuns,23whoshowedadecrease inthesecomplicationswhensuchcarewasperformed.
Some non-infectious oral lesions, such as oral leuko-plakia, gingival hyperplasia, and others that are seldom describedbyotherauthors,alsoinfrequentlywereobserved bythepresentauthors.Orallesionssimilartohemorrhagic lesionsthatweredifficulttocharacterize,butdidnotevoke clinicalcomplaintswereobservedin17patientsfromgroup AandeightpatientsfromgroupBatmoment2,caused per-hapsbythrombocytopenia;thesewereresolvedinmoment 3.
Inthepresentstudy,theincidenceofmucositiswas100% inthe second evaluation, at moment 2,for both groupA andgroupBpatients.Theriskassociationofmoment2and mucositiswassignificant.Bothgroupsshowedanassociation of the risk classificationand/or toxicity(mild, moderate, severe, andvery severe) with the onsetof mucositis and degreesofintensity.Mostpatientswereclassifiedashaving mildrisk,butthosewhodevelopedmucositiswereclassified ashavingmoderateorsevererisk.
Thesepatientsalsohadoral lesions,moreoftenonthe tongueandlabialmucosa,characterizedaserythemaofthe oral mucosa, lichen planus, generalized erosions, ulcera-tions,andxerostomia.30
Someauthorsreportedthatthisisduetoloworalfood intake,because of pain caused by these lesions.18,21,25 In the present study, this observation wasmore evident, as 15patientsfromgroupBrequiredtheintroductionof par-enteral nutrition. In this group of patients, the authors observed dysphagia,anorexia, and rapid weightloss soon aftertransplantation,asearlyasmoment2ofthe evalua-tion.
Wedidnotobserveotherinfections,suchasthosecaused byanaerobicorganismsinourstudyprobablydueto preven-tive measures withoral fluconazoleand nystatin, aswell astheuse ofantisepticmouthwash,which areastandard procedureinallpatientsundergoingHSCT.10,14,15
Undoubtedly, the most feared complication of bone marrow transplantation is death, that occurs in 15---20% of allogeneic transplantations and in 5% of autologous transplantations.Is thisrelated tooral alterations? Death occurredinsevenofthepresentpatients,betweenmoments 2and3aftertransplantation. Oneof thedeathsoccurred ingroupB,frommultiple-organfailureandveno-occlusive disease.IngroupA, sixpatientsdied. Ofthese,fourdied duetodiseaserecurrence,one patientdieddue to pneu-moniaandrespiratoryfailure,andonepatientduetoacute host-versus-graftdiseaseandsepsis.Innoneofthemwere thecausesassociatedwithoraldisease.Althoughmucositis isthemain alterationresponsiblefor theincreasein clin-icalcomplications,especiallyinfectiousonesassociatedto conditioning toxicity,such lesions werenot seen in these patients at the time of death, perhaps due to the dra-matic moment experienced by patients and the medical staff.
Conclusion
We affirm that mucositis is the oral cavity alteration of greatest concern as a potential risk for complications in immunosuppressedpatients.Otherlesions,suchasgingivitis andthird-molarpericoronitis,consideredaslow-risk indica-torsin thisstudy were alsoless relevant.This alterations becamemoreevidentwhenthemild,moderateandsevere risk and toxicity scores were used to assess the clinical complicationsinimmunossupressedindividuals.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
References
1. DulleyF,Saboya R.Transplante demedula óssea.In: Trans-plantedeórgãosetecidos.2nded.SãoPaulo:Ed.Segmento Farma;2006.p.849---50.
2.SchubertMM. Oralcomplications. In:Thomas ED,BlumeKG, FormanSJ,editors.Hematopoieticcelltransplantation. Black-wellScience;1999.p.751---65.
3.McGuireDB,AltomontV,PetersonDE.Patternsofmucositisand paininpatientsreceivingpreparativechemotherapyandbone marrowtransplantation.OncolNursForum.1993;20:1493---502. 4.DahllöfG,BagessundM,RingdénO.Impactofconditioning regi-mens on salivary function, caries-associated microorganisms and dental caries in children after bone marrow transplan-tation.A4-yearslongitudinalstudy.Bone MarrowTransplant. 1997;20:479---83.
5.Blijlevens N, Schwenkglenks M, Bacon P, D’addio A, Ein-sele H, MaertensJ, et al. Prospective oral mucositis audit: oral mucositis in patients receiving high-dose melphalan or BEAMconditioningchemotherapy---Europeanbloodand mar-row transplantation mucositis advisory group. J Clin Oncol. 2008;26:1519---25.
6.Sonis ST. Mucositisas a biological processa newhypothesis forthedevelopmentofchemotherapy-inducedstomatotoxicity. OralOncol.1998;34:39---43.
7.SonisST, Castello KA. A data base for mucositis induced by cancerchemotherapy.OralOncol.1995;315:258---60.
8.López-Casta˜no F, O˜nate-Sanchez RE, Roldán-Chicano R, Cabrerizo-MerinoMC.Measurementofsecondarymucositisto oncohematologictreatmentbymeansofdifferentscale.Med OralCirBucal.2005;10:412---21.
9.Epstein JB, Schubert MM. Oral mucositis in myelossupres-sive CA therapy. Oral Surg Oral Med Pathol Radiol Endod. 1999;88:273---6.
10.PuyalM,JimenézC,ChimenosE,LópesJ,JuliáA.Protocolode estudioytratamientodelamucositisbucalinlospacientescom hematopatiasmalignas.MedOral.2003;8:10---8.
11.KolbinsonDA,ShubertMM,FlournoyN,TrueloveEL.Earlyoral changesfollowingbonemarrowtransplantation.OralSurgOral MedPathol.1988;66:130---8.
12.SonisST,O´DonnellKE,PopatR,BragdonC,PhelanS,CocksD, etal.Therelationshipbetweenmucosalcyclooxigenase-2 (COX-2)expression and experimental radiation induced mucositis. OralOncol.2004;40:170---6.
13.NucciM,SpectorN,BuenoAP,SolzacC,PerecmanisT,BachaPC, etal.Riskfactors andattributablemortalityassociatedwith superinfectionsinneutropenicpatientswithcancer.ClinInfect Dis.1995;24:575---9.
14.PorakD.Factorsinfluencingtheseverityofradiationskinand oralmucosal reactions:developmentofaconceptual frame-work.EurJCancerCare(Engl).2002;1:33---43.
15.KöstlerWJ, HejnaM,Wenzel C,Zielinski CC.Oralmucositis complicationschemotherapyandorradiotherapy:optionsfor preventionandtreatment.CancerJClin.2001;51:290---315. 16.YamagataK, OnizawaK, YanagawaT, HasegawaY,Kogina H,
NakasawaT,etal.Aprospectivestudytoevaluateanewdental management protocolbefore hematopoietic stemcell trans-plantation.BoneMarrowTransplant.2006;38:237---42. 17.NevilleBW,DammDD,AllenCM,BouquotJE.Doenc¸as
imuno-logicamentemediadasesuas avaliac¸ões.In:Patologia oral& maxilofacial.2nd ed.RiodeJaneiro:Ed.Guanabara-Koogan; 2004.p.658---66.
18.BarkerGJ.Currentpractices intheoralmanagement ofthe patientsundergoingchemotherapyorbonemarrow transplan-tation.SupportCareCancer.1999;7:17---20.
20.Pico JL, Ávila-Garavito A. Mucositis: its occurrence, conse-quencesand treatment in the oncology setting. Oncologist. 1998;3:446---51.
21.HeimdahlA, Mattisson T,DahllofG, LonnquistB,RingdenO. The oral cavity as a entry for early infections in patients treated with bone marrow transplantation. Oral Surg Med Pathol.1989;68:711---6.
22.DreizenS,McCredieKB,BodeyGP,KeatingMJ.Quantitative analysisoftheoralcomplicationsofanti-leukemia chemother-apy.OralSurgOralMedOralPathol.1986;62:650---3.
23.SonisST,Kuns A. Impactofimproved dental serviceson the frequencyoforalcomplications ofcancertherapy.OralSurg OralMedOralPathol.1986;62:650---3.
24.ParulekanW,MackenzieR,BjarnasonG,JordanRG.Scoringoral mucositis.OralOncol.1998;34:63---71.
25.Dodd MJ, Dibble S, Miaskowski C, Paul S, Cho M, Mcphael L, etal. A comparison ofthe affectivestate and quality of life of chemotherapy patients who do and do not develop
chemotherapy-induced oral mucositis. J Pain Sym Manag. 2001;21:498---505.
26.RambrgP,LindheJ,DahenG,VolpeAR.Theinfluenceofgingiva inflammationonanewplaqueformation.JClinPeriodontol. 1994;21:51---6.
27.DalyCG,HighfieldJE.Effectoflocalizedexperimentalgingivitis onearlysupragingivalplaqueaccumulation.JClinPeriodontol. 1996;23:160---4.
28.ShulmanJD,River-HidalgoF,BeachMM.Risksfactorsassociated withdenturestomatitisintheUnitedStates.JOralPatholMed. 2005;34:340---6.
29.DeLima DC,Nakata GC,BalducciI, Almeida JD. Oral mani-festationsofdiabetesmellitusincompletedenturewearers. JProstetDent.2008;99:60---5.