Phosphites for the control of anthracnose in common bean
Stélio Jorge Castro Gadaga(1), Mário Sobral de Abreu(1), Mário Lúcio Vilela de Resende(1) and Pedro Martins Ribeiro Júnior(2)
(1)Universidade Federal de Lavras, Departamento de Fitopatologia, Caixa Postal 3037, CEP 37200-000 Lavras, MG, Brazil. E-mail: [email protected], [email protected], [email protected](2)Embrapa Semiárido, Rodovia BR-428, Km 152, Zona Rural, Caixa Postal 23, CEP 56302-970 Petrolina, PE, Brazil. E-mail: [email protected]
Abstract – The objective of this work was to evaluate the effect of phosphites in the protection of common bean (Phaseolus vulgaris) against anthracnose. Different phosphite formulations were evaluated by quantifying peroxidase and polyphenol oxidase activity, total phenols, and lignin content. The treatments consisted of sprayings, in the V4, R5, and R7 stages, with: the K, Zn, Mn, K+Mn, K+salicyclic acid, and Cu phosphites; salicylic acid; acibenzolar-S-methyl; the fungicide azoxystrobin; besides a control, without sprayings. The area under the disease progress curve was lower in plants that received applications of the K and Mn phosphites, whose values ranged between 74 and 81%, compared with the control. The K, Zn, and K+salicyilic acid phosphites were effective in controlling the disease. In addition, disease severity was lower with the application of the K, Zn, and Mn phosphites than with the control. Enzyme (peroxidases and polyphenol oxidases) activity and levels of soluble phenols were higher in common bean plants treated with the K and Mn phosphites, although no change was detected in the levels of soluble lignin in the same tissue. Phosphite application reduces the severity of the disease, can enhance enzymatic activity, and is an effective alternative for the control of anthracnose in common bean.
Index terms: Colletotrichum limdemuthianum, Phaseolus vulgaris, alternative control, enzymatic activity, phenolic compounds.
Fosfitos para o controle da antracnose em feijoeiro comum
Resumo – O objetivo deste trabalho foi avaliar o efeito de fosfitos na proteção do feijoeiro comum (Phaseolus vulgaris) contra antracnose. Diferentes formulações de fosfitos foram avaliadas por meio da quantificação da
atividade de peroxidases e polifenoloxidases, de fenóis totais e do teor de lignina. Os tratamentos consistiram de pulverizações, nos estádios V4, R5 e R7, com: fosfitos de K, Zn, Mn, K+Mn, K+ácido salicílico e Cu; ácido salicílico; acibenzolar-S-metil; o fungicida azoxistrobina; além de um controle, sem aplicações. A área abaixo da curva de progresso da doença foi menor em plantas que receberam aplicações dos fosfitos de K e Mn, cujos valores variaram entre 74 e 81%, em comparação ao controle. Os fosfitos de K, Zn e K+ácido salicílico foram eficazes no controle da doença. Além disso, severidade da doença foi menor com a aplicação dos fosfitos de K, Zn e Mn quando comparado ao controle. A atividade de enzimas (peroxidases e polifenoloxidases) e os níveis de fenóis solúveis foram mais elevados em plantas de feijão comum tratadas com os fosfitos de K e Mn, embora não tenha sido detectada mudança nos níveis de lignina solúvel no mesmo tecido. A aplicação de fosfitos reduz a severidade da doença, pode potencializar a atividade enzimática e é uma alternativa eficaz para o controle da antracnose no feijoeiro comum.
Termos para indexação: Colletotrichum limdemuthianum, Phaseolus vulgaris, controle alternativo, atividade enzimática, compostos fenólicos.
Introduction
Anthracnose, caused by Colletotrichum
lindemuthianum (Sacc. & Magn.) Scrib, is one of the
major diseases of common bean (Phaseolus vulgaris
L.) around the world and in Brazil, which is one of the largest bean producers (Carbonell et al., 2010; Chiorato et al., 2010). The disease affects bean plants at all growth stages, causing lesions on leaves, stems, branches, pods, and seeds, which may cause losses of
up to 100% in production when susceptible varieties are used and the conditions are favorable for the pathogen (Rava & Sartorato, 1994).
for plant protection, such as compounds that can induce plant resistance, exploring the endogenous mechanisms of plant defense.
An alternative method for controlling diseases caused by fungi is the use of essential oils, which increases the resistance levels of the treated crop (Garcia et al., 2012). In this case, the induction of resistance involves the activation of latent defense mechanisms in the plants in response to treatments with biotic (extracts, compounds, and molecules extracted from living organisms such as plants and
microorganisms) or abiotic (acibenzolar-S-methyland
analogs) agents (Bonaldo et al., 2005).
Also useful in managing plant diseases are the phosphites, originating from the neutralization of
phosphorous acid (H3PO3) with a base, which may be
sodium hydroxide, potassium hydroxide, or ammonium hydroxide, among others. Potassium hydroxide is the most commonly used to form potassium phosphite, which that can act directly on fungi or activate the defense mechanism of plants (Reuveni, 1997). Tavares et al. (2009) observed that potassium phosphite induced resistance in papaya (Carica papaya L.) plants against
Phytophthora palmivora. When applied before and after the inoculation of Phakopsora euvitis, phosphites were reported to reduce the colonization by the pathogen on vine (Vitis labrusca L.) leaves, reducing the disease in about 50% (Buffara et al., 2013). Guest & Grant (1991) pointed out that potassium phosphite inhibits the growth of fungal hyphae, acting directly as a toxin on the pathogen, and may also be effective in controlling various other pathogens. Phosphites may also indirectly control pathogens by stimulating the formation of phytoalexin, a natural self-defense substance of the plant (Dercks & Creasy, 1989).
When plants have developed specific receptor
proteins (R-proteins), they can recognize the effector proteins produced by the pathogen, directly or indirectly leading to effector triggered immunity (Dalio et al., 2014).
The objective of this work was to evaluate the effect of phosphites in the protection of common bean (Phaseolus vulgaris) against anthracnose.
Materials and Methods
Greenhouse and field experiments were conducted
from July to October 2009 in the experimental areas of
Universidade Federal de Lavras, in the municipality of Lavras, in the state of Minas Gerais, Brazil (21º13'35"S, 44º58'31"W and 21°12"18"S, 44°58'43"W).
The greenhouse experiment was carried out in a randomized complete block design with three replicates. The experimental plot consisted of six plants, with two pots containing three plants each. Five common bean seeds from the carioca group, cultivar BRS Majestoso, were grown in the pots, which contained 3 L of substrate with soil: sand: manure (2:1:1), and were fertilized with 5 g N-P-K (4-14-8), under greenhouse conditions with an average temperature of 25±3°C and relative humidity of 70%±5°C. Thinning was performed 15 days after emergence, leaving the three more vigorous and homogeneous seedlings.
The experiment in the field was also carried out
in a randomized complete block design with three replicates, and the experimental plot consisted of
five 3-m rows containing 30 plants each, spaced at
0.5 m. The three internal rows that formed the useful plot were selected and marked, and nine plants were evaluated; the two outer rows formed the boundary of the experiment.
Data from both experiments were subjected to the analysis of variance using the Sisvar software, version 5.1, Build 72 (Universidade Federal de Lavras, Lavras, MG, Brazil), and mean separation was performed by
Tukey’s test when the F-test was significant at 5%
probability.
In the experiment under greenhouse conditions, a
pure colony of C. lindemuthianum race 65 LV 81 in
M3 medium, from the collection of the laboratory of plant resistance of the Biology Department of Universidade Federal de Lavras, was multiplied in test tubes containing young autoclaved pods (twice for 1 hour at 120°C) of common bean plants; the test tubes containing agar-water were maintained in a growth chamber at 22±1°C for ten days. Inoculation of the pathogen was performed on the third day in the pods using a 3-mm mycelial disc. After sporulation, to obtain a conidial suspension, 15 mL of distilled water were added to the test tubes and stirred, and the obtained
suspension was filtered through a double cheesecloth. Conidia were quantified with the aid of a Neubauer
counting chamber and adjusted to a concentration of
1x106 conidia mL-1 for inoculation in common bean
seedlings. Disease severity was assessed five times,
inoculation in the greenhouse experiment, using the diagrammatic scale developed by Godoy et al. (1997). The six plants that made up the experimental plot were evaluated at 7, 14, 21, 28, and 35 days after inoculation, in order to calculate the area under the disease progress curve (AUDPC) (Shaner & Finney, 1977).
To control anthracnose in the greenhouse, phosphites were applied to leaves to the point of runoff, using manual sprayers model 78605/050 (Tramontina, Carlos Barbosa, RS, Brazil), at the third trifoliate (V4),
pre-flowering (R5), and pod formation (R7) stages. The
treatments applied were: 5 mL L-1 potassium phosphite
1 (K1), containing 340 g L-1 P
2O5 and 240 g L-1 K2O
Reforce (Agrichem do Brasil Ltda., Ribeirão Preto, SP,
Brazil); 5 mL L-1 Zn phosphite, with 340 g L-1 P
2O5 and
100 g L-1 Zn, Reforce Zn (Agrichem do Brasil Ltda.,
Ribeirão Preto, SP, Brazil); 5 mL L-1 Mn phosphite,
with 510 g L-1 P
2O5 and 97 g L-1 Mn, Reforce Mn
(Agrichem do Brasil Ltda., Ribeirão Preto, SP, Brazil);
5 mL L-1 Mn+K1 phosphite, with 510 g L-1 P
2O5, 97 g L-1
Mn, and 240 g L-1 K
2O, Reforce Mn+K1 (Agrichem
do Brasil Ltda., Ribeirão Preto, SP, Brazil); 5 mL L-1
K1+salicylic acid (SA) phosphite, with 34 g L-1 P 2O5,
24 g L-1 K
2O, and 5% SA, Reforce+SA (Agrichem do
Brasil Ltda., Ribeirão Preto, SP, Brazil); 1–5 mL L-1
potassium phosphite 2 (K2), with 434.28 g L-1 P
2O5 and
403.26 g L-1 K
2O, Nutri Phite (Iharabras S.A., Cajuru
de Sorocaba, SP, Brazil); 5 mL L-1 potassium phosphite
3 (K3), with 420 g L-1 P
2O5 and 280 g L-1 K2O, Pepfós
(Pepita Fertilizantes Ltda., Poços de Caldas, MG,
Brazil); 5 mL L-1 Cu phosphite, with 268 g L-1 P
2O5 and
53.6 g L-1 Cu, Fulland (Sudoeste Fertilizantes Ltda.,
Araxá, MG, Brazil); 0.25 g L-1 a.i. SA (Merck Brazil,
Jacarepaguá, RJ, Brazil); 0.0625 g L-1 a.i.
acibenzolar-S-methyl (ASM) (Syngenta Proteção de Cultivos
Ltda., Santo Amaro, SP, Brazil); 3 mL L-1 Agro-Mos,
with 35 g L-1 Cu, 27.5 g L-1 S, and 25 g L-1 Zn, Alltech
(Crop Science Brasil, Araucária, PR, Brazil); 0.3 g L-1
of the fungicide azoxystrobin (Syngenta Proteção
de Cultivos Ltda., Santo Amaro, SP, Brazil); and a control, without sprayings. Inoculation was carried out at 45 days after sowing in the R6 stage by spraying the inoculum suspension to the point of runoff. After inoculation, the pots with plants were covered with transparent plastic bags during 14 hours to provide a moist chamber, in order to promote penetration and colonization of the fungus. Disease severity was
evaluated five times after the treatments, from July 2
to 30, 2009, at seven-day intervals.
For the management of anthracnose in the field,
phosphites, SA, ASM, and azoxystrobin were also applied on the leaves three times, using a 20-L capacity portable sprayer, at the V4, R5, and R7 stages.
The treatments were: 5 mL L-1 K1 phosphite, with
340 g L-1 P
2O5 and 240 g L-1 K2O, Reforce (Agrichem
do Brasil Ltda., Ribeirão Preto, SP, Brazil); 5 mL L-1
Zn phosphite, with 340 g L-1 P
2O5 and 100 g L-1 Zn,
Reforce Zn (Agrichem do Brasil Ltda., Ribeirão Preto,
SP, Brazil); 5 mL L-1 Mn phosphite, with 510 g L-1
P2O5 and 97 g L-1 Mn, Reforce Mn (Agrichem do
Brasil Ltda., Ribeirão Preto, SP, Brazil); 5 mL L-1
K1+SA phosphite, with 34 g L-1 P
2O5, 24 g L-1 K2O,
and 5% SA, Reforce+SA (Agrichem do Brasil Ltda.,
Ribeirão Preto, SP, Brazil); 0.25 g L-1 a.i. SA (Merck
Brazil, Jacarepaguá, RJ, Brazil); 0.0625 g L-1 a.i. ASM
(Syngenta Proteção de Cultivos Ltda., Santo Amaro,
SP, Brazil); 0.3 g L-1 of the fungicide azoxystrobin
(Syngenta Proteção de Cultivos Ltda., Santo Amaro,
SP, Brazil); and a control, without sprayings.
Inoculation was not carried out in the field since this
happened naturally; the presence of the pathogen was
confirmed in the previous crop planted with common
bean, forming the initial source of the inoculum. The severity of the disease was also evaluated seven times after the application, from July 2 to 30, 2009, at seven-day intervals.
Following the experiments in the greenhouse
and in the field, the biochemical mechanisms of
common bean plants were characterized, in order to understand more about the protective responses against C. lindemuthianum with phosphite application. For this, the plants were treated with the two products that performed better in controlling anthracnose in
both experiments: 5 mL L-1 K1 phosphite, Reforce
(Agrichem do Brasil Ltda., Ribeirão Preto, SP, Brazil),
also used as a standard product; and 5 mL L-1 Mn
phosphite, Reforce Mn (Agrichem do Brasil Ltda.,
Ribeirão Preto, SP, Brazil). These products were
inoculum to the point of runoff using manual sprayers, model 78605/050 (Tramontina, Carlos Barbosa, RS, Brazil) with a capacity of 500 mL. After inoculation, the plants in the pots were covered with transparent plastic bags during 14 hours, to provide a moist chamber, in order to promote fungal penetration and colonization. To determine the activity of peroxidases and polyphenol oxidases, as well as the contents of total soluble phenols, tissues were collected from the oldest to the youngest leafs at 0, 0.5, 1, 3, 6, 7.5, 8, 10, 13, and 17 days after spraying with the inductor products. To obtain the lignin content, leaf tissues were collected at 10, 13, and 17 days after spraying.
The samples used to determine enzyme activities
were packed in properly-identified aluminum foil,
frozen in liquid nitrogen, and stored at -80°C in a freezer. To prepare the extracts, each frozen sample was weighed and macerated individually with the aid of a pestle in a porcelain mortar cooled on ice, and
homogenized in 50 mmol L-1 sodium acetate buffer,
pH 5.2, at a ratio of 1 g fresh sample material to 8 mL buffer. The homogenized extract was centrifuged in
a 5804 R centrifuge (Eppendorf do Brasil, São Paulo,
SP, Brazil) at 24,148.8 g for 10 min, and the supernatant was collected in plastic microtubes, stored at -80°C for posterior analysis.
Total protein concentration was obtained by the Bradford method (1976), adjusting a standard curve previously determined with the use of solutions with known concentrations of bovine serum albumin
(0–100 μg mL-1), correlating the protein concentration
with absorbance readings in an 83057-30 spectrophotometer (BioTek U.S., Winooski, VT, USA), at 595 nm.
Peroxidase (EC 1.11.1.6 catalase) activity was determined using the methodology described by Urbanek et al. (1991). Guaiacol was used as the electron donor and hydrogen peroxide as the receiver. The reaction, catalyzed by the peroxidases, produces a colored 3,3-dimethoxy-4,4-biphenylquinone compound. An aliquot of 30 µL crude extract was
added to 170 µL of a solution containing 0.02 mol L-1
guaiacol, 0.06 mol L-1 hydrogen peroxide, and
50 mmol L-1 sodium acetate buffer pH 5.2. The mixture
was incubated at 30°C for 10 min and its absorbance was measured at 480 nm. The variation of 1.0 unit of absorbance per minute was assumed to be 1.0 unit of peroxidase activity per milligram of protein per minute
[Δ480 nm (mg protein per minute)]. The enzymatic
activity was performed in triplicate.
The polyphenol oxidase (EC 1.14.81.1 purified
polyphenol oxidase) activity was determined by adding 50 µL of the enzyme extract to 150 µL of a solution containing potassium phosphate buffer pH 6.5 and 20 mmol L-1 catechol (Gauillard et al., 1993).
After incubation at 30°C, for 10 min, the increase in
absorbance was measured at 410 nm [Δ410 nm (mg protein per minute)]. Analyses were also performed in
triplicate.
For the determination of lignin content and total soluble phenols, the plant leaf tissues were ground in liquid nitrogen using a mortar and a pestle until
obtaining a fine powder. Subsequently, the samples
were lyophilized in a free dryer, FreeZone Liter Benchtop (Labconco, Kansas City, MO, USA) for 12 hours. An aliquot of 30 mg of the lyophilized material was transferred to a 2-mL microtube, homogenized with 1.5 mL of 80% methanol, and was stirred for 15 hours on a rotary shaker protected from light, at room temperature. The suspension was centrifuged in a 5804 R centrifuge (Eppendorf do
Brasil, São Paulo, SP, Brazil) at 24,148.8 g for 5 min. The supernatant (methanol extract) was transferred to a new microtube, which was conducted to obtain total soluble phenols, whereas the solid residue was used to determine lignin. For this, 1.5 mL of water was added to the solid residue, which was homogenized and centrifuged at 24,148.8 g for 5 min, at 4°C, in the same centrifuge. The supernatant was discarded and the residue was dried at 65°C for 15 hours. Subsequently, 1.5 mL mercaptoacetic acid: 2 mol L-1 HCl (1:10) were
added. Then, the microtubes were gently stirred to hydrate the residue and were placed in a boiling water
bath, model 102R (Fanem, São Paulo, SP, Brazil) for
4 hours. Afterwards, the microtubes were centrifuged at 24,148.8 g for 10 min, at 4°C; the supernatant was discarded; and the precipitate was washed with 1.5 mL of ultrapure water and centrifuged again, also at 24,148.8 g for 10 min, at 4°C. Following this procedure, the supernatant was discarded and the precipitate was resuspended in 1.5 mL of 0.5 mol L-1 NaOH, being kept
on a Stickmax rotary shaker (IKA Brasil, Campinas, SP, Brazil) for 15 hours, at room temperature. The
mixture was centrifuged at 24,148.8 g for 10 min, at
4°C, and the supernatant was transferred to a new
were added. The obtained suspension was kept in a cold room, at 4°C, for 4 hours, in order to allow the precipitation of lignin linked to thioglycolic acid.
Then, the mixture was centrifuged at 24,148.8 g for
10 min, at 4°C; the supernatant was discarded; and
the precipitate was resuspended in 2 mL of 0.5 mol L-1
NaOH. The absorbance values of this solution were determined in a 83057-30 spectrophotometer (BioTek U.S., Winooski, VT, USA), at 280 nm, being calculated based on the lignin curve and expressed in µg of lignin per milligram of dry weight according to
Doster & Bostock (1988), with modifications. For the determination of total soluble phenols, 150 μL aliquots of the methanol extract were mixed with 150 μL
of the 0.25 N Folin-Ciocalteu reagent, for 5 min,
homogenized with 150 µL of 1 mol L-1 Na
2CO3 for
10 min, and diluted with 1 mL of ultrapure water, at room temperature, for 1 hour. The absorbance values of this reaction were also determined in a 83057-30 spectrophotometer (BioTek U.S., Winooski, VT, USA), at 725 nm, being calculated based on the catechol curve. The phenolic compounds were expressed in
equivalent μL of catechol per milligram of dry weight
(Spanos & Wrolstad, 1990).
The results were evaluated by the analysis of variance, and means were compared by Tukey’s test, at 5% probability, using the Sisvar software, version 5.1 (Build 72) (Universidade Federal de Lavras, Lavras, MG, Brazil).
Results and Discussion
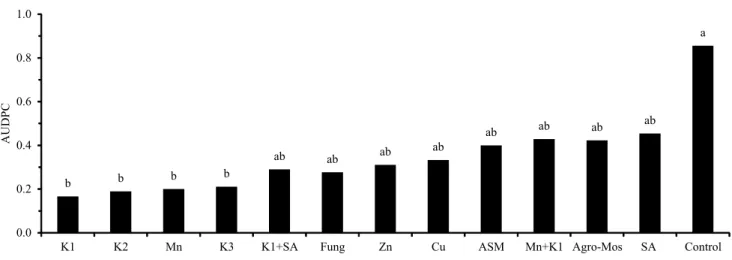
The evaluation of the disease progress curve (AUDPC) showed that the K1, K2, Mn, and K3 phosphites caused the greatest reductions of anthracnose, of 81, 80, 77, and 74%, respectively, being statistically similar to the other treatments but different from the control in the greenhouse experiment (Figure 1). Moreover, K1 phosphite+SA, fungicide, Zn phosphite, Cu phosphite, ASM, K+Mn phosphite, Agro-Mos, and SA were statistically identical to each other, although intermediate when compared with the best treatments and the control.
Negri et al. (2011) reported a positive effect of K
phosphite in reducing the infection by Monilinia
fruticola in 49–73% in flowers and in 57–84% in fruits
of peach [Prunus persica (L.) Batsch]. Araújo et al.
(2010) observed, in vitro and in vivo, that phosphites
interfered in the development of Colletotrichum
gloeosporioides, causing a decrease of 94% in the mycelial growth of the colonies and reducing in
62% the severity of leaf spot caused by Glomerella
cingulata when applied 48 hours after inoculation. Fuzitani et al. (2013) also found positive effects of K
phosphites in controlling Phytophthora palmivora
in Bactris gasipaes Kunth var. gasipaes Henderson.
These results show that there is scientific evidence of the beneficial effects of phosphites in controlling plant
diseases in several pathosystems.
All treatments in the field experiment were
statistically similar and reduced the AUDPC in 74 to 90%, in comparison with the control treatment (Figure 2). The best results were with K phosphite, Zn phosphite, Mn phosphite, and ASM, which allowed controlling the disease similarly to the recommended fungicide. Silva et al. (2014) reported that the K and Ca
phosphites reduced the AUDPC in pepper (Capsicum
annuum L.) against Pectobacterium carotovorum
subsp. carotovorum (Pcc). In the present study, the
phosphites were effective in controlling anthracnose in
common bean in the field trial. Similar results have also been verified by other authors such as Lins et al. (2011),
who observed a decrease in AUDPC when phosphite
was applied in mangoes (Mangifera indica L.) to
control Lasiodiplodia theobromae. Silva et al. (2013), while evaluating soybean (Glycine max L.), found that
K phosphite and Mn significantly reduced the AUDPC
of mildew and had no effect on powdery mildew and Asian soybean rust.
The activities of peroxidases in common bean plants increased in the treatments with phosphites from the third day after spraying the product, compared with the control (Figure 3 A). A slight decrease was observed in the peroxidase levels of plants treated with K phosphite, as well as in the control inoculated 7.5 days
Figure 2. Effect of phosphites, acibenzolar-S-methyl (ASM), fungicide (Fung), and salicylic acid (SA) on the area under the disease progress curve (AUDPC) of anthracnose in 'BRS Majestoso' common bean (Phaseolus vulgaris). K1, potassium phosphite 1; Zn, zinc phosphite; and Mn, manganese phosphite. Means followed by equal letters do not differ by Tukey’s test, at 5% probability.
Figure 3. Activity of guaiacol peroxidase in 'BRS Majestoso' common bean (Phaseolus vulgaris) leaves after treatment with: A, acibenzolar-S-methyl (ASM); B, potassium phosphite (K phosph); and C, manganese phosphite (Mn phosph), compared with the control. The inoculation with
after the treatment; however, in the uninoculated control, these levels were always higher (Figure 3 B). The plants treated with Mn phosphite showed the greatest differences in the levels of peroxidase over the experimental period (Figure 3 C). A clear increase was
verified in plants treated with Mn phosphite inoculated
or not from the sixth day after treatment, reaching a peak at 8 days and continuing until day 17 (Figure 3 C).
The polyphenol oxidase activity was similar when the plants were treated with ASM (Figure 4 A), but varied in function of the treatments K and Mn phosphites (Figure 4 B and C). Uninoculated plants treated with K phosphite showed peak activity of polyphenol oxidase at six days after treatment, performing well above the uninoculated control. However, when inoculation occurred, there was a decrease in these levels with phosphite application and in the control (Figure 4 B). The polyphenol oxidase levels of plants treated with Mn phosphite did not change until the sixth day after treatment; however, after 7.5 days there was a increase in the uninoculated plants treated with Mn phosphite (Figure 4 C).
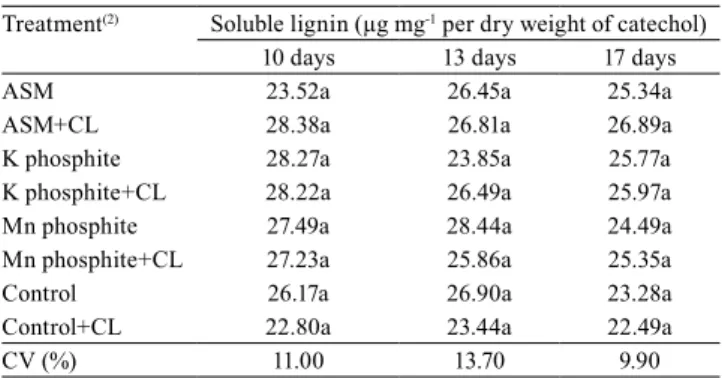
The content of total phenolic compounds varied only at ten days after spraying, in the treatments applied with K phosphite, Mn phosphite, and ASM, in comparison with the control; however, in the other two samplings no differences were found between treatments (Table 1). As for lignin, no difference was observed in any of the treatments (Table 2).
The obtained results show that phosphites increase peroxidase activity, as reported by Ribeiro Júnior et al. (2006), who studied phosphite application in inducing
the resistance of the cocoa (Theobroma cacao L.)
pathosystem to Verticillium dahliae. The authors found a small increase in the peroxidase activity at 13 days
after the application of K phosphite (1.25 mL L-1).
Several functions are attributed to peroxidases in
cellular defense, specifically their involvement in cell wall lignification, suberization, and metabolism, being classified as pathogenesis-related proteins (Loon
& Strien, 1999). Borsato et al. (2010) observed an increase of 65% in the peroxidase activity 48 hours
after inoculation of Uromyces appendiculatus on
the susceptible cultivar IPR Juriti of common bean, when compared with the resistant cultivar BR IPA 11
(Brígida). Felipini & Di Piero (2013) verified that ASM
induced resistance by promoting the accumulation of
peroxidases and β-1,3 glucanases in beet (Beta vulgaris
Figure 4. Activity of polyphenol oxidase in 'BRS Majestoso' common bean (Phaseolus vulgaris) leaves after treatment with: A, acibenzolar-S-methyl (ASM); B, potassium phosphite (K phosph); and C, manganese phosphite (Mn phosph), compared with the control. The inoculation with
L.) leaves. Regarding lignin content, no differences
were verified between the treatments, in alignment
with the results of Daniel & Guest (2006). These
authors inoculated Arabidopsis thaliana (L.) Heynh.
with Phytophthora palmivora in a medium containing
K phosphite and reported phenolic accumulation at the site of infection 6 hours after inoculation and that these compounds were concentrated around the infected cells after 48 hours.
Conclusions
1. Phosphites can control and reduce anthracnose in
common bean (Phaseolus vulgaris) and can be used as
an alternative control method.
2. Phosphite applications on common bean leaves increase the activity of peroxidases and polyphenol oxidases, as well as total soluble proteins, and are effective in the control of anthracnose.
Acknowledgments
To Ministério da Ciência e Tecnologia de
Moçambique, for financial support to the first author.
References
ARAÚJO, L.; VALDEBENITO-SANHUEZA, R.M.; STADNIK,
M.J. Avaliação de formulações de fosfito de potássio sobre
Colletotrichum gloeosporioides in vitro e no controle pós-infeccional da mancha foliar de Glomerella em macieira.
Tropical Plant Pathology, v.35, p.54-59, 2010. DOI: 10.1590/ S1982-56762010000100010.
BONALDO, M.B.; PASCHOLATI, S.F.; ROMEIRO, R.S. Indução de resistência: noções básicas e perspectivas. In: CAVALCANTI,
L.S.; DI PIERO, R.M.; PASCHOLATI, I.S.F.; RESENDE, M.L.V. de; ROMEIRO, R. da S. (Ed.). Indução de resistência em plantas a patógenos e insetos. Piracicaba: Fealq, 2005. p.11-28.
BORSATO, L.C.; DI PIERO, R.M.; STADNIK, M.J. Mecanismos de defesa eliciados por ulvana contra Uromycesappendiculatus em três cultivares de feijoeiro. Tropical Plant Pathology, v.35, p.318-322, 2010. DOI: 10.1590/S1982-56762010000500008.
BRADFORD, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Analytical Biochemistry, v.72, p.248-254, 1976.
BUFFARA, C.R.S.; ANGELOTTI, F.; TESSMANN, D.J.;
SOUZA, C.D. de; VIDA, J.B. Atividade de fosfito de potássio na pré e pós-infecção de Phakopsora euvitis em folhas de videira.
Semina: Ciências Agrárias, v.34, p.3333-3340, 2013. Suplemento 1. DOI: 10.5433/1679-0359.2013v34n6Supl1p3333.
CARBONELL, S.A.M.; CHIORATO, A.F.; CARVALHO, C.R.L.; RAMOS JUNIOR, E.U.; ITO, M.A.; BORGES, W.L.B.; TICELLI, M.; SANTOS, N.C.B. dos; BOLLER GALLO, P. IAC Formoso: new carioca common bean cultivar. Crop Breeding and Applied Biotechnology, v.10, p.374-376, 2010. DOI: 10.1590/ S1984-70332010000400014.
CHIORATO, A.F.; CARBONELL, S.A.M; VENCOVSKY, R.; FONSECA JÚNIOR, N. da S.; PINHEIRO, J.B. Genetic gain in the breeding program of common beans at IAC from 1989 to 2007.
Crop Breeding and Applied Biotechnology, v.10, p.329-336, 2010. DOI: 10.1590/S1984-70332010000400007.
Table 1. Content of total soluble phenols (TSP) in leaves of 'BRS Majestoso' common bean (Phaseolus vulgaris) plants, after 10, 13, and 17 days application of treatment with acibenzolar-S-methyl (ASM), K phosphite, and Mn phosphite(1).
Treatment(2) TSP (µg mg
-1 per dry weight of catechol)
10 days 13 days 17 days
ASM 1.49b 1.57a 1.56A
ASM+CL 1.77a 1.72a 1.65A
K phosphite 1.65a 1.63a 1.60A
K phosphite+CL 1.54b 1.68a 1.63A
Mn phosphite 1.77a 1.63a 1.52A
Mn phosphite+CL 1.87a 1.73a 1.56A
Control 1.32b 1.53a 1.54A
Control+CL 1.42b 1.43a 1.50A
CV (%) 11.50 12.29 10.10
(1)Means followed by equal letters, in the columns, do not differ by
Scott--Knott’s test, at 5% probability. (2)Inoculation of Colletotrichum
lindemu-thianum (CL) occurred seven days after spraying with treatments. CV,
coefficient of variation.
Table 2. Content of soluble lignin in leaves of 'BRS Majestoso' common bean (Phaseolus vulgaris) plants, after 10, 13, and 17 days application of treatment with acibenzolar-S-methyl (ASM), K phosphite, and Mn phosphite(1).
Treatment(2) Soluble lignin (µg mg-1 per dry weight of catechol)
10 days 13 days 17 days
ASM 23.52a 26.45a 25.34a
ASM+CL 28.38a 26.81a 26.89a
K phosphite 28.27a 23.85a 25.77a
K phosphite+CL 28.22a 26.49a 25.97a
Mn phosphite 27.49a 28.44a 24.49a
Mn phosphite+CL 27.23a 25.86a 25.35a
Control 26.17a 26.90a 23.28a
Control+CL 22.80a 23.44a 22.49a
CV (%) 11.00 13.70 9.90
(1)Means followed by equal letters, in the columns, do not differ by
Scott--Knott’s test, at 5% probability. (2)Inoculation of Colletotrichum
lindemu-thianum (CL) occurred seven days after spraying with treatments. CV,
DALIO, R.J.D.; FLEISCHMANN, F.; HUMEZ, M.; OSSWALD, W. Phosphite protects Fagus sylvatica seedlings towards Phytophthora plurivora via local toxicity, priming and facilitation of pathogen recognition. PLoS One, v.9, e87860, 2014. DOI: 10.1371/journal.pone.0087860.
DANIEL, R.; GUEST, D. Defence responses induced by potassium phosphonate in Phytophthora palmivora-challenged Arabidopsis thaliana. Physiological and Molecular Plant Pathology, v.67, p.194-201, 2006. DOI: 10.1016/j.pmpp.2006.01.003.
DERCKS, W.; CREASY, L.L. Influence of fosetyl-Al on
phytoalexin accumulation in the Plasmopara viticola-grapevine interaction. Physiological and Molecular Plant Pathology, v.34, p.203-213, 1989. DOI: 10.1016/0885-5765(89)90044-1.
DOSTER, M.A.; BOSTOCK, R.M. Quantification of lignin
formation in almond bark in response to wounding and infection by Phytophthora species. Phytopathology, v.78, p.473-477, 1988.
FELIPINI, R.B.; DI PIERO, R.M. PR-protein activities in table beet against Cercospora beticola after spraying chitosan or acibenzolar-S-methyl. Tropical Plant Pathology, v.38, p.534-538, 2013. DOI: 10.1590/S1982-56762013000600009.
FUZITANI, E.J.; SANTOS, A.F. dos; MORAES, W. da S.;
DAMATTO JUNIOR, E.R.; NOMURA, E.S. Eficiência de fosfitos no controle da podridão da base do estipe em mudas de
pupunheira. Revista Brasileira de Fruticultura, v.35, p.1000-1006, 2013.
GARCIA, R.A.; JULIATTI, F.C.; BARBOSA, K.A.G.; CASSEMIRO, T.A. Atividade antifúngica de óleo e extratos vegetais sobre Sclerotinia sclerotiorum. Bioscience Journal, v.28, p.48-57, 2012.
GAUILLARD, F.; RICHARDFORGET, F.; NICOLAS, J. New ectrophotometric assay for polyphenol oxidase activity.
Analytical Biochemistry, v.215, p.59-65, 1993.
GODOY, C.V.; CARNEIRO, S.M.T.P.G.; IAMAUTI, M.T.; DALLA PRIA, M.; AMORIM, L.; BERGER, R.D.; BERGAMIN FILHO, A. Diagrammatic scales for bean diseases: development and validation. Zeitschrift für Pflanzenkrankheiten und
Pflanzenschutz, v.104, p.336-345, 1997.
GUEST, D.; GRANT, B. The complex action of phosphonates as antifungal agents. Biological Reviews, v.66, p.159-187, 1991.
LINS, S.R. de O.; OLIVEIRA, S.M.A. de; ALEXANDRE, E.R.; SANTOS, A.M.G. OLIVEIRA, T.A.S. de. Controle alternativo
da podridão peduncular em manga. Summa Phytopathologica, v.373, p.121-126, 2011. DOI: 10.1590/S0100-54052011000300007.
LOON, L.C. van; STRIEN, E.A. van. The families of pathogenesis-related proteins, their activities, and comparative analysis of PR-1
type proteins. Physiological and Molecular Plant Pathology, v.55, p.85-97, 1999.
NEGRI, G.; BIASI, L.A.; WORDELL FILHO, J.W.; MAY-DE
MIO, L.L. Manejo da queima das flores e da podridão-parda do
pessegueiro cultivado em sistema orgânico. Revista Brasileira de Fruticultura, v. especial, E.415-423, 2011.
RAVA, C.A.; SARTORATO, A. Antracnose. In: SARTORATO, A.; RAVA, C.A. (Ed.). Principais doenças do feijoeiro comum e seu controle. Brasília: EMBRAPA-SPI, 1994. p.17-39. (EMBRAPA-CNPAF. Documentos, 50).
REUVENI, M. Post-infection applications of K3PO3, phosphorous acid and dimethomorph inhibit development of downy mildew caused by Plasmopara viticola on grapes. Journal of Small Fruit and Viticulture, v.5, p.27-38, 1997. DOI: 10.1300/J065v05n02_03.
RIBEIRO JÚNIOR, P.M.; RESENDE, M.L.V. de; PEREIRA, R.B.; CAVALCANTI, F.R.; AMARAL, D.R.; PÁDUA, M.A.
de. Fosfito de potássio na indução de resistência a Verticillium dahliae Kleb., em mudas de cacaueiro (Theobroma cacao L.).
Ciência e Agrotecnologia, v.30, p.629-636, 2006. DOI: 10.1590/ S1413-70542006000400006.
SHANER, G.; FINNEY, R.E. The effect of nitrogen fertilization on the expression of slow-mildewing resistance in Knox wheat.
Phytopathology, v.67, p.1051-1056, 1977.
SILVA, M.S. e; CARVALHO, F.C.Q.; SILVA, J.R. da; LINS, S.R. de O.; OLIVEIRA, S.M.A. de. Uso de antagonistas e produtos
alternativos no manejo pós-colheita de podridão mole em pimentão. Revista Ciência Agronômica, v.45, p.718-725, 2014. DOI: 10.1590/S1806-66902014000400009.
SILVA, O.C.; SANTOS, H.A.A.; DESCHAMPS, C.; DALLA
PRIA, M.; MAY DE MIO, L.L. Fontes de fosfito e acibenzolar-S-metílico associados a fungicidas para o controle de doenças
foliares na cultura da soja. Tropical Plant Pathology, v.38, p.72-77, 2013. DOI: 10.1590/S1982-56762013000100012.
SPANOS, G.A.; WROLSTAD, R.E. Influence of processing
and storage on the phenolic composition of Thompson Seedless grape juice. Journal of Agricultural and Food Chemistry, v.38, p.1565-1571, 1990. DOI: 10.1021/jf00097a030.
TAVARES, G.M.; LARANJEIRA, D.; LUZ, E.D.M.N.; SILVA, T.R.; PIROVANI, C.P.; RESENDE, M.L.V. de;
RIBEIRO JÚNIOR, P.M. Indução de resistência do mamoeiro à podridão radicular por indutores bióticos e abióticos. Pesquisa Agropecuária Brasileira, v.44, p.1416-1423, 2009. DOI: 10.1590/ S0100-204X2009001100007.
URBANEK, H.; KUZNIAK-GEBAROWSKA, E.; HERKA, H. Elicitation of defence responses in bean leaves by Botrytis cinerea polygalacturonase. Acta Physiologiae Plantarum, v.13, p.43-50, 1991.