r e v b r a s r e u m a t o l . 2017;57(6):637–640
w w w . r e u m a t o l o g i a . c o m . b r
REVISTA
BRASILEIRA
DE
REUMATOLOGIA
Brief
communication
Hepcidin-25
gives
an
indication
of
the
therapeutic
effectiveness
of
tocilizumab
in
rheumatoid
arthritis
–
Relationship
between
disease
activity
of
rheumatoid
arthritis
and
anemia
A
hepcidina-25
dá
uma
indicac¸ão
da
eficácia
terapêutica
do
tocilizumab
na
artrite
reumatoide
–
Relac¸ão
entre
a
atividade
da
doenc¸a
na
artrite
reumatoide
e
a
anemia
Satoshi
Suzuki
a,b,∗,
Souichiro
Nakano
a,b,
Seiichiro
Ando
a,
Ran
Matsudaira
a,
Yoshinori
Kanai
a,
Kenjiro
Yamanaka
a,
Yoshinari
Takasaki
baKyoundoHospital,SasakiInstitute,DepartmentofRheumatologyandInternalMedicine,Tokyo,Japan
bJuntendoUniversitySchoolofMedicine,DepartmentofRheumatologyandInternalMedicine,Tokyo,Japan
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received16June2015 Accepted24July2016
Availableonline18October2016
Introduction
Therapy for RA has improved rapidly since the advent of biologics. However, although biologics have a greater effectcomparedtothatwithconventionaldiseasemodifying antirheumaticdrugs(DMARDs),thecostofthesenewer ther-apiesremainsextremely high.1 Patientsinwhom biologics
areeffectivehaveincreasedemploymentopportunitiesdueto reduceddiseaseactivity,meaningthatthecost-effectiveness ofthetreatmentisvalid.However,incaseswherethe admin-istrationofbiologicsisnotassociatedwitharesponse,disease activity during the period of treatment and the progres-sionofjoint damageplace anincalculable burdenon both
∗ Correspondingauthor.
E-mail:[email protected](S.Suzuki).
patients and the entire medical system. For this reason, the identification of factors that can predict the effective-nessofbiologicsineachpatient,beforeadministration,isa critical issue; tailor-made therapiesusing singlenucleotide polymorphisms (SNPs) are currently being investigated. In Japan,biologicscurrentlyavailableforuseinRAincludethose that target tumornecrosisfactor (TNF)-␣ (infliximab,
etan-ercept, adalimumab, golimumab, and certolizumab pegol), tocilizumab(TCZ),whichtargetsIL-6,and abatacept,which targetsCD80/86.Wehavepreviouslyreportedonthe relation-shipbetweenTNF-␣therapyandautoantibodies(anti-dsDNA
antibodiesandanti-SS-Aantibodies)inpatientswith insuffi-cientresponses.2Inthepresentstudy,weexaminedpredictive
factorsforthetherapeuticeffectofbiologics,whilefocusing
http://dx.doi.org/10.1016/j.rbre.2016.09.004
638
rev bras reumatol.2017;57(6):637–640Table1–Patientcharacteristics.
Patients Sex Age (y.o)
Duration (years)
Hb(g/dL) CRP (mg/dL)
MMP3 (ng/mL)
CDAI(CDAIat24 weekslater)
MTX (mg/w)
PSL (mg/day)
Previous biologics
1 F 78 4 13.0 0.8 96 8.0(6.2) 8 4 ETN
2 M 58 2 13.6 2.3 258 34.2(24.9) 7.5 10 –
3 F 73 3 11.4 5.1 243 31.6(29.7) – – –
4 F 51 4 11.1 2.7 170 26.0(1.0) 10 10 INF
5 F 61 7 12.5 0.6 284 16.3(4.0) 8 7.5 –
6 F 46 7 13.3 1.8 176 14.3(10.9) 6 8 INF
7 F 49 13 13.6 0.2 – 19.5(1.1) – – ETN
8 F 73 21 10.0 0.1 150 16.0(24.0) 10 7.5 –
9 F 60 20 12.4 3.2 714 24.5(40.0) 6 4 INF
10 F 39 6 10.1 4.2 403 29.2(8.0) 8 4 INF
Hb,hemoglobin;CRP,C-reactiveprotein;MMP3,matrix metalloproteinase3;CDAIclinicaldiseaseactivityindex;MTX,methotrexate;PSL, prednisolone;INF,infliximab;ETN,etanercept.
on the effect of TCZ in ameliorating anemia of chronic
inflammation,viadirectinhibitionofIL-6receptor-mediated
signaling.3 Hepcidin-25,amajorcauseofanemiaofchronic
inflammation,4wasmeasured inthe serumofRApatients.
Wecomparedserumhepcidin-25levelswithclinicaldataand cytokinelevels,toexaminewhetherhepcidin-25couldactas apredictivefactorfortheeffectivenessofTCZ.
Materials
and
methods
Patients
The10patientsselectedforinclusioninthestudyhad previ-ouslybeenadmittedtotheSasakiInstituteKyoundoHospital Division of Internal Medicine and Rheumatology between September 2008 and September 2010. Patients had com-mencedTCZtherapyduringthisperiod,andwereeithernaive to biologics or had changed from other biologics to TCZ. AllRApatientsmettheAmerican CollegeofRheumatology (ACR)1987revisedcriteriafortheclassificationofrheumatoid arthritis.5BasedontheDeclarationofHelsinkiandthe
guide-linesofthe institutionalreviewboardofKyoundoHospital, writtenconsentwasobtainedfromallpatientspriortothe startofthestudy.Thecharacteristicsofthe10patientsare showninTable1.
Methods
Hepcidin-25andIL-6levelsweremeasuredbefore introduc-tion of TCZ and 3 months after introduction. The serum sampleswerestoredat−80◦C.Serumhepcidin-25,whichis
thoughttobetheformofhepcidinwiththegreatest bioactiv-ity,wasmeasured usinganenzyme-linked immunosorbent assay (ELISA) (Hepcidin-25 EIA Kit; BACHEM, Bubendorf, Switzerland). Serum IL-6 was also measured using ELISA (HumanIL-6QuantikineELISAKit;R&DSystems, Minneapo-lis,MN,USA).Bothhepcidin-25andIL-6weremeasuredusing theSandwichELISAprotocol.Testresultsfromroutinetests undertakenduringgeneralexaminationatKyoundo Hospi-tal, including serum hemoglobin, C-reactive protein (CRP), anderythrocytesedimentationrate(ESR),werealsousedin ouranalysis.RAdiseaseactivitywasassessedusingclinical
diseaseactivityindex(CDAI).Student’st-testwasusedfor sta-tisticalanalysis.Ap-value<0.05wasconsideredstatistically significant.
Results
TherapeuticeffectivenessofTCZinRApatients
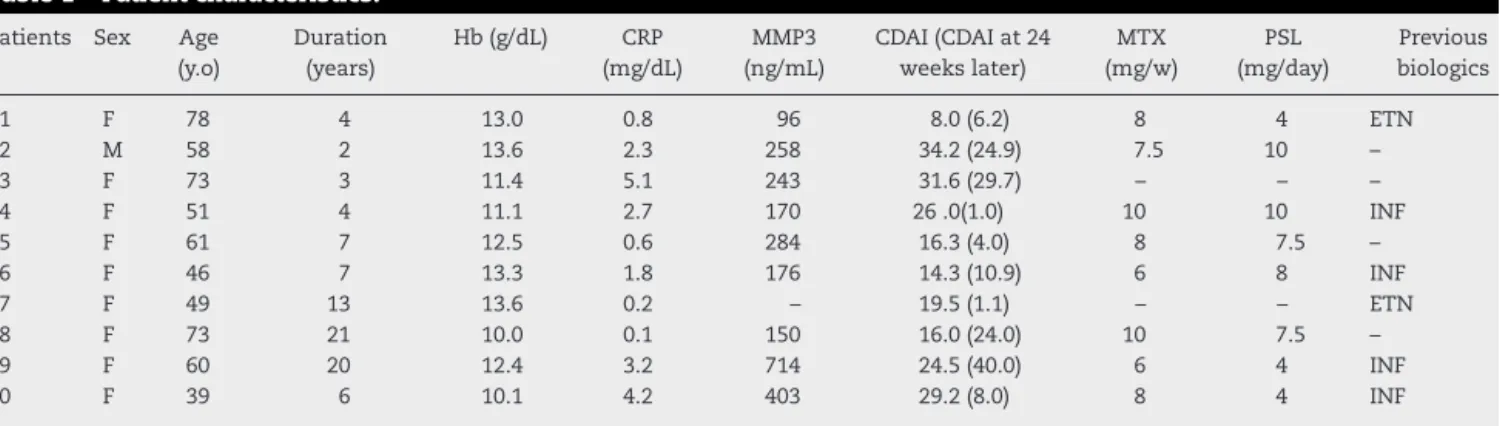
CDAIforpatientspriortoandaftertreatmentwithTCZare showninFig.1A.
SixmonthsafterinitiationoftreatmentwithTCZ, lower CDAIwerenotedin8of10patients.
Comparisonofhepcidin-25levelsinRApatientsand healthysubjects
Inacomparisonofserumhepcidin-25levelsinRApatients beforereceivingtreatmentwithTCZandinhealthysubjects, hepcidin-25levelsweresignificantlyelevatedinRApatients (Fig.1B).
Changesinhepcidin-25andIL-6levelsinRApatients
Serum hepcidin-25 and IL-6 levels in RA patients before administrationofTCZand12weeks(3months)after admin-istrationareshowninFig.1C.
Hepcidin-25
Serumhepcidin-25levelswerehighin8of10patientsbefore administrationofTCZ;inthese8patients,levelsdecreased aftertreatmentwithTCZ.However,inthe2patientsthathad comparatively low levels before administration, treatment with TCZ was associated with an increase inhepcidin-25. TheseresultsreflectedtheeffectivenessofTCZtherapy.Inthe 8patientswhoexhibitedadecreaseinhepcidin-25,TCZ ther-apywaseffective,whileinthelatter2patients,TCZtherapy wasdeterminedtobeineffective.
IL-6
ItisknownthatbecauseTCZisanIL-6receptorantagonist, serumIL-6levelstemporarilyincreaseafteradministration.6
rev bras reumatol.2017;57(6):637–640
639
A
C
D
45
40
35
30
CD
AI 25
20
15
10
5
0
180
p<0.01
n.s 160
140
120
100
80
60
40
20
0 0W
0W
24W AR
(n=10)
Cont. (n=5)
12W 0W 12W 0W 12W
180
Serum level of
hepcidin-25(ng/mL)
Serum level of
hepcidin-25(ng/mL
)
Serum level of IL-6(pg/mL
)
Serum level of Hb(g/dL)
160
140
120
100
80
60
40
20
0
600
500
400
300
200
100
0
15
14.5
14
13.5
13
12.5
12
11.5
11
10.5
10
B
Fig.1–(A)SixmonthsafterinitiationoftreatmentwithTCZ,CDAIwaslowerin8of10patients,andwastherefore consideredtobeeffective.TCZwasfoundtobeineffectivein2of10patients;(B)inacomparisonofserumhepcidin-25 levelsinRApatientsandhealthysubjects,hepcidin-25levelswerefoundtobesignificantlyelevatedinRApatients(p<0.01);
(C)highlevelsofserumhepcidin-25wereobservedpriortoadministrationofTCZ,anddecreased3monthsafterinitiation oftreatment.However,2of10patientshadpre-administrationlevelsthatwerecomparativelylow;theselevelsincreased aftertreatmentwithTCZ.These2caseswereconsistentwiththe2patientsinwhomTCZwasconsideredtobeineffective6 monthsafterinitiationoftreatment.Thegraylineindicatesthesamepatientsasthegraylinein(A);(D)weexamined hemoglobinlevels3monthsafteradministrationofTCZ.Elevatedhemoglobinlevelswerenotedin5of10patients.Elevated levelswereobservedin3of4patientsinwhomanemiawaspresentbeforereceivingtreatment(Hb<12g/dL,dottedline).
patientsafteradministrationofTCZ;however,adecreasewas notedin3patients.
Relationshipbetweenhepcidin-25andanemia
Weobservedchangesinserumhemoglobinlevels3months after the start of TCZ treatment, compared with the pre-TCZhemoglobinlevel(Fig.1D).Hemoglobinlevels,ageneral indicatorofanemia,increasedin5of10patientsafter admin-istrationofTCZ.Whenanalysiswaslimitedtothosepatients inwhomanemiawaspresentpriortoreceivingtreatment(Hb <12g/dL), mostpatientsshowed anincreaseinhemoglobin levels.
Discussion
Hepcidinisapeptidehormonethatisproducedintheliver. There are several known isoforms of hepcidin, including hepcidin-20 and hepcidin-22; hepcidin-25 has the highest bioactivity and is considered tohave astrong relationship withironmetabolism.7Irontakenorallyisabsorbedby
intesti-nalepithelialcells.Inthepresenceofirondeficiency,ironis
releasedtotheperipheralblood.Whennodeficiencyofiron ispresent,ironisexcretedfecallyastheepithelialcellsare sloughedoffintotheintestinallumen.Mostironabsorbedin theintestinaltractisstoredintheliver. Asuitableamount ofironissuppliedfromthelivertotheperipheralblood,for useinerythrocyteproductioninthebonemarrow.Old eryth-rocytesaretrappedanddestroyedinthereticuloendothelial system,andtheironistakenupbythesystem,andreleased againintotheperipheralblood.Inthecaseofchronic inflam-matory diseases suchas RA,thereis anoverproductionof IL-6.When IL-6bindstoitsreceptorsintheliver, hepcidin-25 isproduced.Hepcidin-25bindstoferroportin,expressed inhepatocytesandreticuloendothelialmacrophages,leading to inhibition of intestinal absorption of iron, and inhibi-tionofironreleasefromthereticuloendothelialsystemand hepatocytes; together these factors result in the develop-ment of anemia.8,9 Hepcidinexpression isup-regulated by
ironand inflammationanddown-regulatedbyanemia and hypoxia.10
640
rev bras reumatol.2017;57(6):637–640anticipatedtobeparticularlyeffectiveinthesepatients. How-ever,inthepresentstudyTCZwasnoteffectivein2cases, despitethefactthatserumIL-6levelsbeforetreatmentwere abnormallyhigh;in1ofthesecasesserumhepcidin-25 lev-els werelow (Fig.1C). Ithaspreviouslybeen reportedthat althoughanemiaofchronicinflammationmaybepresentin RApatients, alowlevelofhepcidinthatisstrongly indica-tive of iron deficiency, was also noted.12 In the present
study, marked iron deficiency was noted in patients who hadabnormallyhighserumIL-6levelswithconcomitantlow hepcidin-25levels.
Weconcludedthatbecausehepcidin-25 levelsdecreased afterTCZadministrationandimprovementintheassociated anemia was noted, TCZcan ameliorate anemia of chronic inflammation.
Patientsthathadhighhepcidin-25levelsbefore adminis-trationofTCZandlowerlevels3monthsaftertreatment,were stillevaluatedashavinghadagoodresponse6monthsafter treatment.Basedonthis,weconsiderthathepcidin-25can actasapredictivefactorfortheeffectivenessofTCZ treat-ment.However,cautionisneededindeterminingcasesofiron deficiencyanemia.Measurementofhepcidincanbeeasily per-formed,andtheapplicationofhepcidinasaclinicalindicator inRApatientscanbeexpectedinthefuture.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1. vanderVeldeG,PhamB,MachadoM,IeraciL,WittemanW, BombardierC,etal.Cost-effectivenessofbiologicresponse modifierscomparedtodisease-modifyingantirheumatic drugsforrheumatoidarthritis:asystematicreview.Arthritis CareRes(Hoboken).2011;63:65–78.
2.MatsudairaR,TamuraN,SekiyaF,OgasawaraM,Yamanaka K,TakasakiY.Anti-Ro/SSAantibodiesareanindependent factorassociatedwithaninsufficientresponsetotumor necrosisfactorinhibitorsinpatientswithrheumatoid arthritis.JRheumatol.2011;38:2346–54.
3.IsaacsJD,HarariO,KoboldU,LeeJS,BernasconiC.Effectof tocilizumabonhaematologicalmarkersimplicates interleukin-6signallingintheanaemiaofrheumatoid arthritis.ArthritisResTher.2013;15:R204.
4.MassonC.Rheumatoidanemia.JtBoneSpine.2011;78:131–7.
5.ArnettFC,EdworthySM,BlochDA,McShaneDJ,FriesJF, CooperNS,etal.TheAmericanRheumatismAssociation 1987revisedcriteriafortheclassificationofrheumatoid arthritis.ArthritisRheum.1988;31:727–35.
6.NishimotoN,HashimotoJ,MiyasakaN,YamamotoK,Kawai S,TakeuchiT,etal.Studyofactivecontrolledmonotherapy usedforrheumatoidarthritis,anIL-6inhibitor(SAMURAI): evidenceofclinicalandradiographicbenefitfromanXray reader-blindedrandomisedcontrolledtrialoftocilizumab. AnnRheumDis.2007;66:1162–7.
7.ParkCH,VaroleEV,WaringEJ,GanzT.Hepcidin,aurinary antimicrobialpeptidesynthesizedintheliver.JBiolChem. 2011;276:7806–10.
8.YoshizakiK.Anewtherapeuticstrategyforautoimmuneand chronicinflammatorydiseasebasedonclinicalresultsusing IL-6blockingtherapywithahumanizedanti-IL-6receptor antibody.YakugakuZasshi.2009;129:667–74.
9.HashizumeM,MiharaM.Therolesofinterleukin-6inthe pathogenesisofrheumatoidarthritis.Arthritis.2011.ID 765624.8p.
10.NicolasG,ChauvetC,ViatteL,DananJL,BigardX,DevauxI, etal.Thegeneencodingtheironregulatorypeptidehepcidin isregulatedbyanemia,hypoxia,andinflammation.JClin Invest.2002;110:1037–44.
11.ManalAlyAK,AmalMohamadEB,SalwaAME,AbeerSG. Serumhepcidin:adirectlinkbetweenanemiaof
inflammationandcoronaryarteryatherosclerosisinpatients withrheumatoidarthritis.JRheumatol.2011;38:2153–9.