Braz. j. . vol.82 número6
Texto
Imagem

Documentos relacionados
The diagnosis of LAR is corroborated by demonstrating the presence of the nasal specific IgE through nasal provoca- tion test with allergens or the local synthesis of IgE, without
Results: The treatment involving chemotherapy with cisplatin caused 41.9% and 47.3% hearing loss in the right and left ear, respectively, with a 11.7-fold higher risk of hearing loss
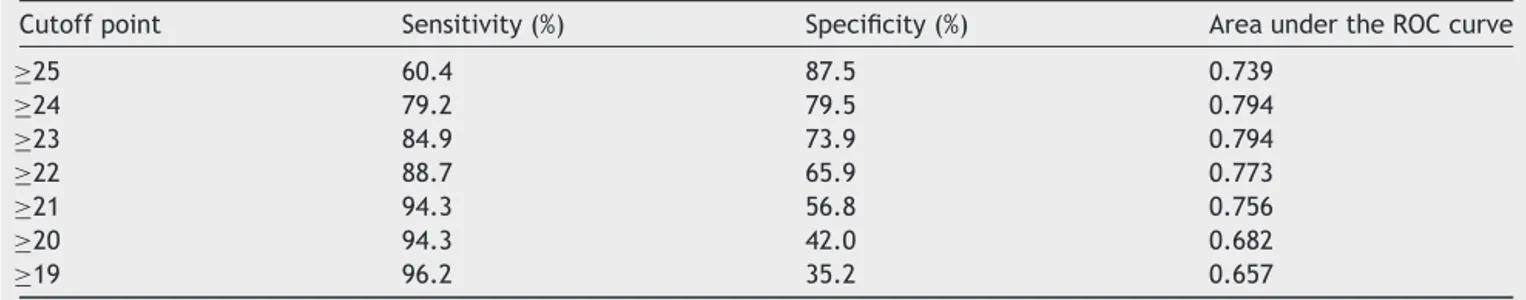
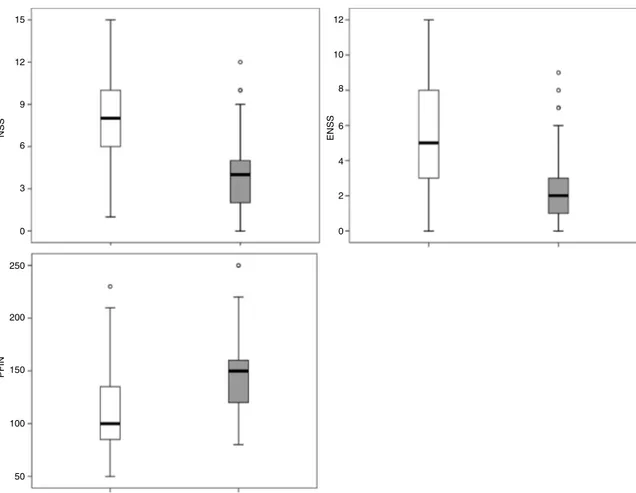
Objective: To measure the distance from the posterior wall of the maxillary sinus to the skull base, according to socio-demographic characteristics, and to detail an
The need for a disease-specific quality of life question- naire to assess the impact of sinonasal symptoms in children has been met by the development and validation of the
In the present study, 29.3% of the otological disorders were affected with tinnitus, of which 30.3% were affected with nasal or nasopharyngeal factors especially tonsillopharyn-
Objective: To evaluate the association of vestibular dysfunction and sensorineural hearing loss (SNHL) in adult patients.. Methods: Prospective, double-blinded, controlled
The current study aimed to evaluate the association of intranasal splinting intervals with bacterial colonization, early and late complications (tissue necrosis, mucosal crusting,
We analyzed the white blood cell count, neutrophil count, lymphocyte count, platelet count, C-reactive protein, mean platelet volume and neutrophil-to-lymphocyte ratio values