Rev. Nutr. vol.27 número5
Texto
Imagem

Documentos relacionados
Takeuchi, Role of nitric oxide in regulation of gastric acid secretion in rats: effects of NO donors and NO synthase inhibitor, Br. Kim, Effects of a nitric oxide donor and nitric
Different diet quality assessment indices or scores have been developed based on admittedly healthy dietary patterns or food guides for the general population, or aiming to
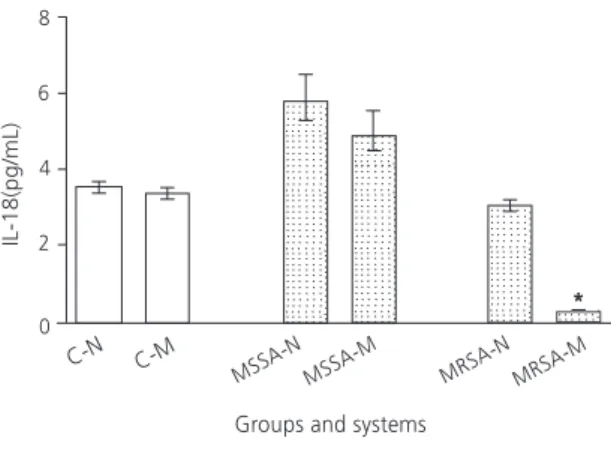
In this report we evaluated the kinetics of cell migration in dermal inflammatory infiltrate, circulating leukocytes and the presence of nitric oxide (NO)/induced nitric oxide
Nitric oxide mediates leptin-induced luteinizing hormone-releas- ing hormone (LHRH) and LHRH and leptin- induced LH release from the pituitary
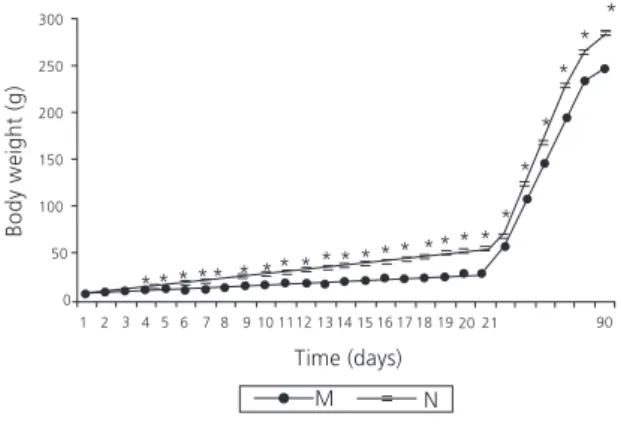
shown in Table 3, when comparing SD propa- gation in the groups malnourished for 1 week, the least affected groups were those mal- nourished during the second week of lacta- tion
Alter- natively, the up-regulation of CB1 could have some neuro- protective role as shown in other studies, which have dem- onstrated increased levels of endocannabinoids and
Pretende ser “uma atividade pensante de espírito ampla, generalizadora, sistematizadora e unificadora, ordenada para a construção de um conhecimento maximamente
“A diferença de juízo entre Sócrates e Górgias sobre o poder da retórica mostra como, do ponto de vista da construção das personagens, elas possuem valores absolutamente