Control of cancer growth using single input autonomous fuzzy
Nano-particles
Fahimeh Razmi1*, Reihaneh Kardehi Moghaddam1, Alireza Rowhanimanesh2
(1)Young Researchers and Elite Club, Mashhad Branch, Islamic Azad University, Mashhad, Iran. (2)Department of Electrical Engineering, Mashhad Branch, Ferdowsi University, Mashhad, Iran.
Copyright 2015 © Fahimeh Razmi, Reihaneh Kardehi Moghaddam and Alireza Rowhanimanesh. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Abstract
In this paper a single input fuzzy controller is applied on autonomous drug-encapsulated nanoparticles (ADENPs) to restrict the cancer growth. The proposed ADENPs, swarmly release the drug in local cancerous tissue and effectively decreases the destruction of normal tissue. The amount of released drug is defined considering to feed backed values of tumor growth rate and the used drug. Some significant characteristics of Nano particles compared to Nano-robots is their ability to recognize the cancerous tissue from the normal one and their simple structure. Nano particles became an attractive topic in Nano science and many efforts have been done to manufacture these particles. Simulation results show that the proposed controlling method not only decreases the cancerous tissue effectively but also reduces the side effects of drug impressively.
Keyword: Nano medicine, Autonomous nanoparticles, Cancer, Single input fuzzy control, Proportional control.
1 Introduction
One of the most common causes of mortality in humans is cancer. Uncontrolled growths of body cells and abnormal modified cells have an important role in the initiation and development of cancer. Cancer cells can separate from the main tissues to which they belong and enter the blood and lymph tissue and go to the other tissues and there they are reproduced and continue to the defective growth cycle. In recent years, considerable progress has been made in the field of nanotechnology which enables us to produce pharmaceuticals and encapsulated nanoparticles for selective drug delivery to an organ-specific or tissue. Local drug delivery by AFNP (Autonomous Fuzzy Nanoparticles) reduces side effects on healthy tissues; as a result, physician has enough authority to select the type and dosages of drugs. Nano medicine is a non-invasive method for the prevention and cancer treatment compared with other existing method switch nowadays is being considered. Hamzeh et al. [1] demonstrated that their designed targeted DENPs, 80 nm
Available online at www.ispacs.com/jfsva
Volume 2015, Issue 1, Year 2015 Article ID jfsva-00229, 11 Pages doi:10.5899/2015/jfsva-00229
the drugs and imaging agents in laboratory mice. Freitas et al. [2-3] proposed a theoretical basis using chemical and thermal gradients to navigate Nano robots towards the coronary occlusion. Generally, the previous works are classified into two categories: DENPs and Nano robots. The capabilities of a Nano robot is much higher than a DENP, while manufacturing of Nano robots is difficult by current nanotechnology.
control be achieved. The proposed model is a new one for the ADENP environment and the cells of cancerous tissue. This model is based on the popular models of cancer [13]. In the next section, the mathematical model is described and after that proposed controlling method is defined in section 3. The simulation results will be presented, in section 4.
2 Mathematical models
Here, a model based on aknown cancermodels fortheADENP withdrug-related and cells cancertissues
is presented [10,13]. In this model, the tumor growth has been affected in the condition of using the total cells number including population function of numerous immune cells diffiusion i.e thenumber ofthe active
antigensoftumorcellsCD8 + T,natural killercells(NK)and all lymphocytes of circulating blood in addition to theconcentration of IL-2andchemotherapy drug in theblood flow[14]. In theequationsbelow: T(t) is the total tumor cell population; N(t) is the concentration (cells/l) of NK cells per litter of blood; L(t) is the concentration (cells/l) of CD8+T-cells per litter of blood; C(t) indicates the concentration (cells/l) of
lymphocytes per litter of blood including NK cells and CD8+T-cells; M(t) shows the concentration (mg/l) of
chemotherapy drug per litter of blood; I(t) is the concentration (IU/l) of IL-2 per litter of blood; VL(t) defines
the number of tumor-activated CD8+T cells injected per day per litter of blood volume (in cells/l per day);
VM(t) is the amount of doxorubicin injected per day per litter of body volume (in mg/l per day); and VI(t)
indicates the amount of IL-2 injected per day per litter of body volume (in IU/l per day); CDenp is
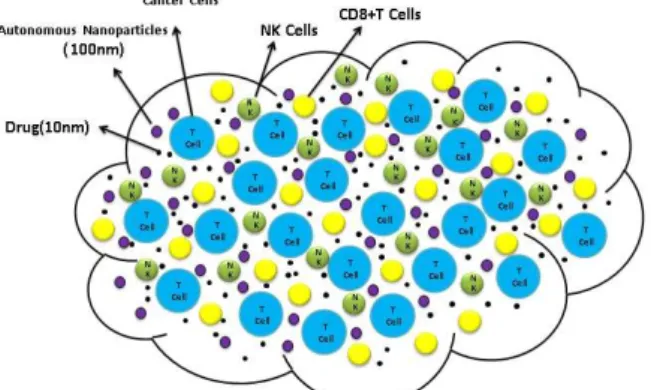
drug-encapsulated by Nanoparticles and UDenp, is the Controller. Figure 1, shows the cancer cells, immune cells,
drugsand autonomous nanoparticles.
Figure 1: cancer cells, immune cells, drugs and autonomous nanoparticles illustration.
The model equations are as follows [10-13]:
T e K DT cNT bT aT dt dT M T T ) 1 ( ) 1 (
(2.1)
In equation (2.1), a is the Growth rate of tumor, b is inverse of carrying capacity, c denotes the rate of NK-induced tumor death, Rate KT is rate of chemotherapy-induced tumor death and T is Medicine efficacy
coefficient. N e K I g NI p pNT N C f e f dt dN M N N
N (1 N )
) (
(2.2)
Where e / f is the ratio of NK cell synthesis with turnover rate,f indicates the rate of NK cell turnover, p shows the Rate of NK cell death due to tumor interaction. PN indicates the rate of IL-2 induced NK cell
) ( ) 1 ( ) ( 2 2 1 t I g LI p L e K I k CI uL T C r N r qLT L T k T j I mL dt dL L I I M L L (2.3)
In the equation (2.3), m is the rate of activated CD8+T-cell turnover. θ is the Concentration of IL-2 to halve
CD8T-cell turnover, Rate of q: CD8+T-cell death due to tumor interaction. r is the rate of NK −
lysed tumour cell debris activation of CD8 + T − cell r is the rate of CD8 production from circulating lymphocytes, pI is the rate of IL-2 induced CD8+T-cell activation, g
I is the concentration of IL-2 for
half-maximal CD8T-cell activation, u is the CD8+T-cell self-limitation feedback coefficient, K is the
concentration of IL-2 to halve magnitude of CD8T-cell self-regulation, j is the rate of CD8T-lysed tumor cell debris activation of CD8T cells, K is the tumor size for half-maximal CD8T-lysed debris CD8T activation,KLis the rate of CD8T depletion from medicine toxicity, δLis the medicine toxicity coefficient.
C e K C dt dC M C C ) 1 ( ) (
(2.4)
In the equation (2.4), is the ratio of rate of circulating lymphocyte production to turnover rate, is the
rate of lymphocyte turnover, �� is the rate of lymphocyte depletion form medicine toxicity,
� is the medicine toxicity coefficient.
) (t U C M dt dM denp denp
(2.5)
In the equation (2.5), is the rate of excretion and elimination of doxorubicin.
) (t I LI C dt dI I
I
(2.6)
In the equation (2.6), �� is the rate of excretion and elimination of IL-2, � is the rate of IL − 2 production from CD8T cells, � is the rate of IL-2 production from CD4/naive CD8T cells, � is the concentration of IL − 2 for half − maximal CD8T − cell IL − 2 production.
l l L s L d D ) / ( ) / (
(2.7)
The the equation (2.7), d is theimmune system strength coefficient
Ɩ is the immune strength scaling coefficient, � is the value of L
T l
necessary for half maximal CD8+T cell
toxicity.
In literatures it was shown that immunotherapy in combination with chemotherapy in patients with strong immune system is very useful. In contrast, in patients with impaired immune systems, immunotherapy may have relatively little effect on destroying cancerous tissue [15]. The proposed model shows that with a swarm of autonomous nanoparticle carrying drugs, tumor cells can be destroyed with minimum side effects. Parameter values in the model simulations have been considered according to [13].
3 The proposed controlling methods
3.1. Motivtion
Global drug delivery and local drug delivery by nanoparticles are two purposeful cases for cancer therapy. From the control engineering point of view, the global drug delivery is an open control loop and drug delivery by autonomous nanoparticle is targeted by a closed loop control. In overall drug delivery, the drug is released directly into the bloodstream. And only a small dose of drug can be taken to avoid drug toxicity. This method cannot identify unhealthy cells (abnormal) from healthy (normal). Thus the drug is released blindly (open loop control). Which leads to unwanted side effects in normal tissues. Although therapy by overall drug delivery is economically much cheaper. Accordingly, we proposed the proportional autonomous nanoparticles and Fuzzy autonomous nanoparticles. These autonomous nanoparticles release drugs locally and as a swarm so side effects on healthy tissues, is reduced. Autonomous nanoparticles have a sensor which recognizes cancer cells continuously at any moment, and accordingly decides about the amount of drug release at the site of cancerous tissues. In the following, we will explain these structure of the autonomous nanoparticles.
3.2. Structure of proposed autonomous Nanoparticles
Proposed autonomous nanoparticles utilizes three simple units, consisting of sensor unit, control unit and operator unit which in case of any disturbance enables us to feel the human body and independence of required medicines consistently. Figure 2 depicts the structure of autonomous nanoparticles. Nanoparticle sensor unit diagnoses the number of cancer cells in every moment, and then by the control unit, decides how much drugs must be released via operator unit which includes medicines control valve. So through this way drug consumption is more efficient. Nanoparticle control unit consist of linear controller or nonlinear controller.
In this paper, we consider the nanoparticles as a system, and our vision is mathematical. So Specific nanoparticles can be designed by specific medicines and chemical and physical structure. An autonomous nanoparticle does not include any propulsion unit, while it acts like a molecule in terms of emission or natural convection to the transport pharmaceuticals.
Figure2: Proposed Autonomous Nanoparticles (ADENP).
3.3. Applying proportional controller
Autonomous nanoparticle operates with proportional controller as a simple structure closed-loop control, which reduces the computational complexity. proposed controller is as follows:
KT
In equation (3.8), T is the number of cancer cells and is the release rate of drugs and K is the Adjustable parameters in the controller and which according to the desired performance, is designed like rate of drug delivery. Freitas works [2], [18] in the field of Nano-robotics is a base and hypothesis structure in medicine. Today, in nanotechnology, nanoparticle is easy to carry and its hardware architecture is of interest to be used reasonably. In recent years, much work has been done in the field of sensors and stimulants Nano scales. Different data from empirical studies shows the nanometer manufacturing of control valves which will be used for drug delivery [17], [19]. Particle size in many of these effects is less than 100 nm, which is promising enough to build an autonomous nanoparticle in near future.
3.4.Apply fuzzycontroller
Knowledge-based systems such as, Fuzzy logic have been successfully implemented in multifarious applications where the human expertise and dealing with uncertainty play a vital role in decision making process [8]. Fuzzy logic avoids arbitrary rigid boundaries by taking into account the continuous character of imprecise informations. A fuzzy system is characterized by the inference system that contains the rule base for the system, input membership functions that are used for the fuzzification of the input variables and de-fuzzification of the output variables.
Fuzzification is a process where crisp input values are transformed into membership values of the fuzzy sets. After the process of fuzzification, the inference engine calculates the fuzzy output using fuzzy rules which are linguistic in the form of if then rules. De-fuzzification is a mathematical process used to convert the fuzzy output to a crisp value. System the rules and the membership functions should be tuned according to the application. In the proposed fuzzy system, Mamdani minimum inference method [9] is used and de-fuzzification is carried out using centroid defuzzifer. Mamdani’s inference system can be mathematically written as,
)))
(
,
max(min(
wz
where,
w(
z
)
is the output membership function and
is the combined membership in the rule antecedent.3.4.1.Single input - Autonomous Fuzzy Nanoparticle (AFNP)
Single input -AFNP contains cancer cells as the input and includes the following Fuzzy rules:
If T is Very High Then����� is Very Large
If T is High Then����� is Large
If T is Medium Then�����is Avrage
If T is Low Then�����is Small
If T is Zero Then����� is Zero
3.4.2. Membership functions of AFNP
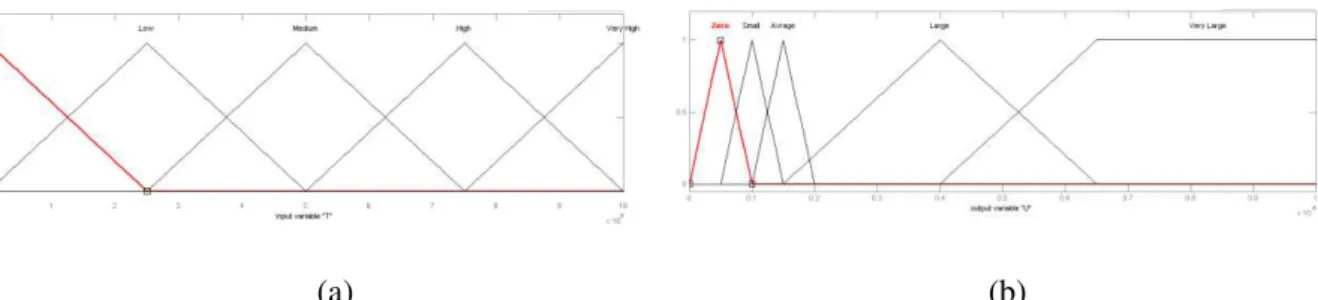
In Figure 3, input and output membership functions are displayed.
(a) (b)
Figure 3: (a) the membership function of the tumor cells (b) membership function of injected medication drug
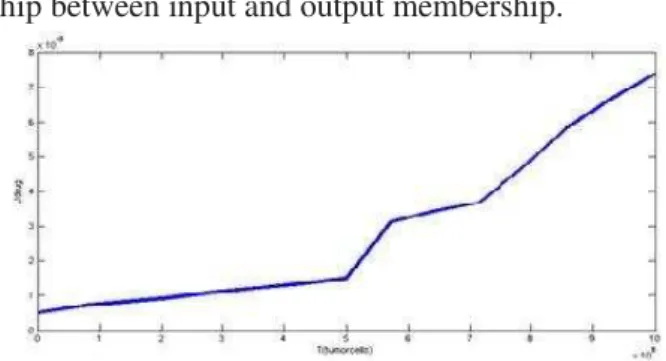
Figure 4shows therelationshipbetweeninput and outputmembership.
Figure 4: The relationship between input and output membership functions.
4Simulation Results
In this section, the proposed model for the control of cancer is simulated using MATLAB. Drug concentrations of proposed ADENP and the molecular weight of pharmaceuticals for cancer patients have been considered as follow [10].
L
Number
C
DENP^
6
.
693
10
13/
(4.9)g
m
4
.
98
10
20 (4.10)day mg
K1017 / (4.11) Maximum number of drug molecules to the encapsulated ADENP is 1500 and the mass of each nanoparticle is considered to be 60000 times larger than m [10].
4.1. Proportional controllerSimulation Results
(a) (b)
(c) (d)
(e) (f)
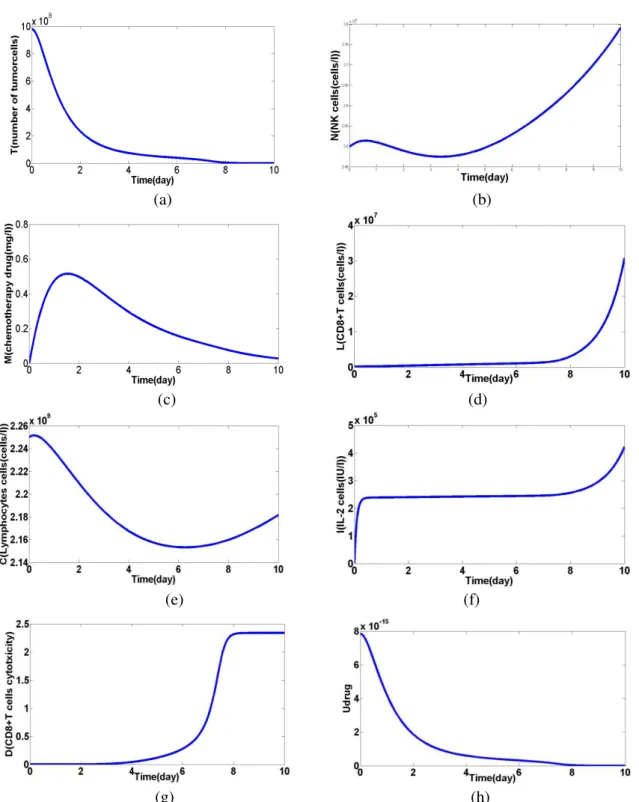
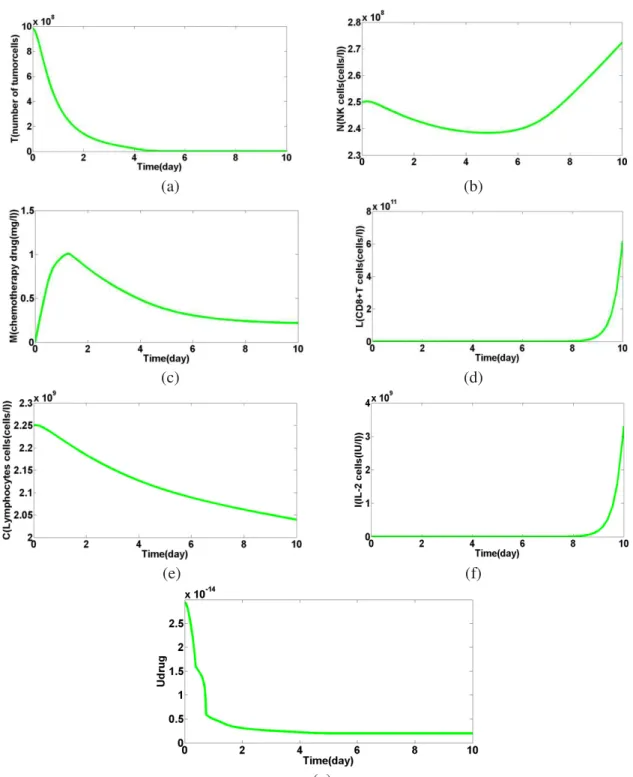
(g) (h)
Figure 5: the impact of proposed autonomous nanoparticles on tumor cells and Safety in 10 days. (a) Number of tumor within 10 days. (b) Number of decreased natural killer (NK) shows the duration of treatment. (c) Shows the chemical drugs. (d), (f), (g) the concentration of immune cells and cancer cells in drug use during the 10-days show.
(e) The concentration of emissions per liter ofbloodlymphocytesshow.(h) drug released by ADENP.
4.2. Simulation resultsof singleinput AFNP
(a) (b)
(c) (d)
(e) (f)
(g)
Figure 6: the impact of single-input Fuzzy autonomous nanoparticles (AFNP) on tumor cells and Safety in 10 days. (a) Number of tumor within 10 days for a single-input AFNP show. (b) Number of decreased natural killer (NK) shows the duration of treatment. (c) Shows the chemical drugs. (d), (f) the concentration of immune cells and cancer
cells in drug use during the 10-days show. (e) The concentration of emissions per liter of blood lymphocytes show. (g) Drug released by AFNP.
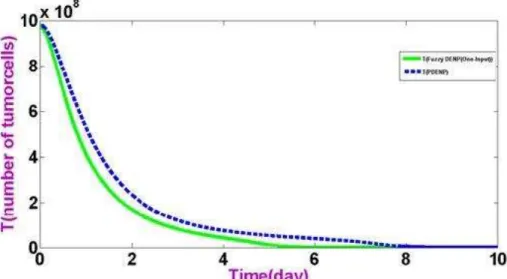
According to the results objecting applying proportional controller and fuzzy nanoparticles it can be observed. Well as single-input Fuzzy autonomous nanoparticles has better performance than Proportional
Figure 7: reduction of tumor cells by Proportional nanoparticles and Fuzzy nanoparticle single-input
5 Conclusion
In this paper a new approach for controlling tumor growth is proposed using single input fuzzy and proportional Nano particles. Fuzzy controller is a nonlinear robust one which has ability of controlling system considering to linguistic knowledge and whiteout any need for knowing the exact model of dynamical system. The proposed nanoparticles have the ability of cancerous tissue recognition and decreasing the side effects. Also we optimize the drug amount and time duration of treatment considering to simulation results. Related to simulation results it is clear that the fuzzy controller performs more effective results, in terms of time duration of treatment and consumption, compare to proportional controller.
Reference
[1] J. Hamzah et al; Specific penetration and accumulation of a homing peptide within atherosclerotic plaques of apolipoprotein E-deficient mice, Proc. Natl. Acad. Sci. USA, 108 (17) (2011) 7154-59.
http://dx.doi.org/10.1073/pnas.1104540108
[2] R. Freitas, Pharmacytes: An ideal vehicle for targeted drug delivery, J. Nanosci. Nanotechnol, 6 (2006) 2769-75.
http://dx.doi.org/10.1166/jnn.2006.413
[3] A. Cavalcanti, B. Shirinzadeh, M. Zhang, L. C. Kretly, Nanorobot hardware architecture for medical defense, Sensor, 8 (2008) 2932-58.
http://dx.doi.org/10.3390/s8052932
[4] H. Suraj, V. B. Reddy, QCA based navigation for nano robot for the treatment of coronary artery disease, in Proc. IEEE Int. Workshop Med. Meas. (2011) 131-36.
[5] S. S. Hossain, S. F. A. Hossainy, Y. Bazilevs, V. M. Calo, and T. J. R. Hughes, Mathematical modeling of coupled drug and drug-encapsulated nanoparticle transport in patient-specific coronary artery walls, in Computational Mechanics, Springer, 49 (2012) 213-242.
[6] S. S. Hossain, S. F. A. Hossainy, Y. Bazilevs, V. M. Calo, T. J. R.Hughes, Mathematical modeling of coupled drug and drug-encapsulated nanoparticle transport in patient-specific coronary artery walls, Institute for Computational Engineering and Sciences, University of Texas at Austin, ICES Rep, (2010) 10-41.
[7] S. Martel, Aggregates of synthetic microscale nanorobots versus swarms of computer-controlled flagellated bacterial robots for target therapies through the human vascular network, in Proc. 4th Int. Conf. Quantum, Nano, Micro Technol, (2010) 14-17.
http://dx.doi.org/10.1109/ICQNM.2010.28
[8] K. Nakano, K. Egashira, S. Masuda, K. Funakoshi, G. Zhao, S. Kimura, T. Matoba, K. Sueishi, Y. Endo, Y. Kawashima, K. Hara, H. Tsujimoto, R. Tominaga, K. Sunagawa, Formulation of nanoparticle-eluting stents by a Cationic electrodeposition coating technology: Efficient nano-drug delivery via bioabsorbable polymeric nanoparticle-elutingtents in Porcine coronary arteries, Cardiovascular Interventions, 2 (4) (2008) 277-83.
http://dx.doi.org/10.1016/j.jcin.2008.08.023
[9] Priyambada Parhi, Chandana Mohanty, Sanjeeb Kumar Sahoo, Nanotechnology-based combinational drug delivery:an emerging approach for cancer therapy, Elsevier Ltd, 17 (17/18) (2012) 1044-52.
[10] Alireza Rowhanimanesh, Mohammad-R. Akbarzadeh-T, Control of Low-Density Lipoprotein Concentration in the Arterial Wall by Proportional Drug-Encapsulated Nanoparticles, IEEE Transactions On Nanobioscience, 11 (4) (2012) 394-401.
http://dx.doi.org/10.1109/TNB.2012.2217382
[11] Lisette de Pillisa, K. Renee Fisterb, Weiqing Gua, Craig Collinsc, Michael Daubd, David Grosse, James Mooree, Benjamin Preskill, Mathematical model creation for cancer chemo-immunotherapy, Computational and Mathematical Methods in Medicine, 10 (3) (2009) 165-84.
[12] A. K. Abbas, A. H. Lichtman, Cellular and Molecular Immunology, 5th ed., Elsevier Saunders, St. Louis, MO (2005).
[13] J. Elbaz, O. Lioubashevski, F. Wang, F. Remacle, R. D. Levine, I. Willner, DNA computing circuits using libraries of DNAzyme subunits, Nature Nanotechnol, 5 (2010) 417-422.
http://dx.doi.org/10.1038/nnano.2010.88
[14] L. Ai, K. Vafai, A coupling model for macromolecule transport in a stenosed arterial wall, Int. J. Heat Mass Transf, 49 (2006) 1568-91.
http://dx.doi.org/10.1016/j.ijheatmasstransfer.2005.10.041
[15] A. Cavalcanti, R. A. Freitas, Jr, Nanorobotics control design: A collective behavior approach for medicine, IEEE Trans. NanoBiosci, 4 (2) (2005) 133-40.
http://dx.doi.org/10.1109/TNB.2005.850469
[16] X. Zhang, F. Dou, H. Liu, Molecular concentration sensor based on the diffraction resonance mode of gold nanowire gratings, Nanotechnology, 21 (33) (2010) 335-501.