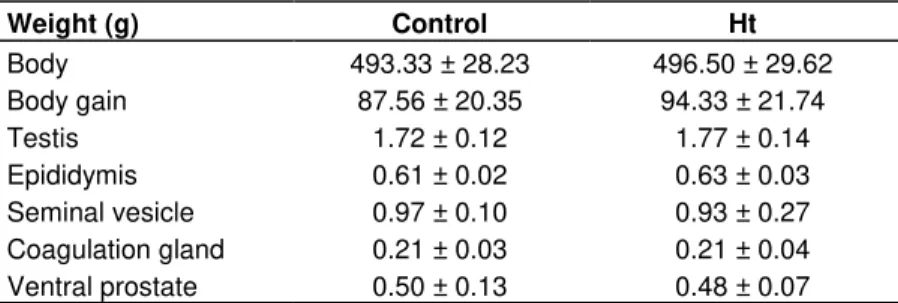
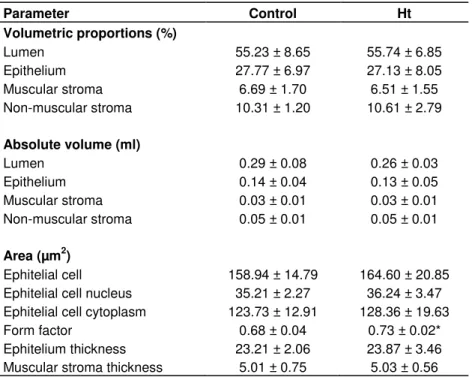
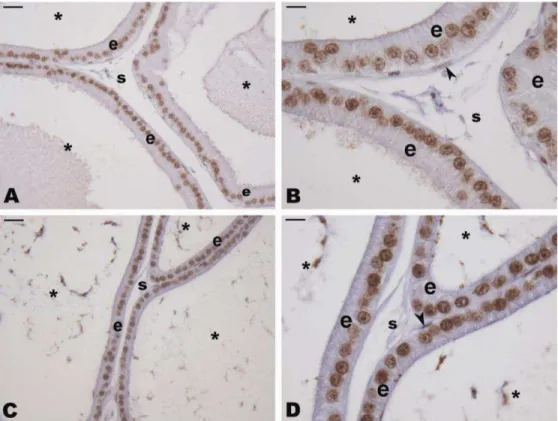
Study of the ventral prostate of Wistar rats treated with Heteropterys tomentosa (A. Juss.)
Texto
Imagem
Documentos relacionados
The probability of attending school four our group of interest in this region increased by 6.5 percentage points after the expansion of the Bolsa Família program in 2007 and
In the present study, we evaluated the effect of plasma and prostate caro- tenoid concentration on oxidative DNA damage in 12-week-old Wistar rats treated with intraperitoneal ( ip
The present study was carried out to investigate the effects of Rudgea viburnoides infusion on the body biometry and testicular morphometry and stereology of adult Wistar
Ousasse apontar algumas hipóteses para a solução desse problema público a partir do exposto dos autores usados como base para fundamentação teórica, da análise dos dados
Notavelmente, a coadministração única com uma dose sub-efetiva de guanosina (0,01 mg/kg) e de cetamina (0,1 mg/kg) foi eficaz em camundongos submetidos ao teste da
The era of building Japanese Buddhist temples in Brazilian society coincided with the beginning of the discovery of Buddhists among the immigrants even though only a few of
This study evaluated the effects of 5ARI on androgen receptor (AR) and proteins involved in prostate cell growth such as HOXB13 expression in human prostate tissue and LNCaP
Allen Dulles, ex-diretor-geral da Agência Central de Inteligência (CIA) norte- americana, sugere que inteligência seja o mesmo que clarividência ou ofício