Abst ract
Submitted: September 2, 2016 0RGL¿FDWLRQ1RYHPEHU Accepted: December 18, 2016
Effect iveness of a new non- hydr ogen
per oxide bleaching agent aft er
single use - a double- blind
placebo-cont r olled shor t - t er m st udy
Toot h whit ening represent s perhaps t he m ost com m on aest het ic procedure LQGHQWLVWU\ZRUOGZLGH7KHHI¿FDF\RIEOHDFKLQJGHSHQGVRQWKUHHDVSHFWV bleaching agent , bleaching m et hod, and t oot h color. Obj ect ive: This in vivo
st udy aim ed t o ex am ine w hit ening effect s on fr ont al t eet h of t he upper
and low er j aw s using an over- t he- count er ( OTC) non- hy dr ogen per ox ide
bleaching agent in com parison t o a placebo aft er one single use. Mat erial and
m et hods: Fort y subj ect s ( 25 fem ale; 15 m ale) part icipat ed in t his double- blind
random ized placebo- cont rolled t rial. The subj ect s w ere random ly allocat ed t o
t w o groups ( n= 20) . The t est group received t he OTC product ( iWhit e I nst ant )
and t he placebo group received an ident ically com posed product except for t he DFWLYHDJHQWV(DFKVXEMHFWZDVWUHDWHGZLWKDSUH¿OOHGWUD\FRQWDLQLQJL:KLWH I nst ant or t he placebo for 20 m inut es. The t oot h shade of t he front t eet h ( upper
and low er j aw s) was assessed before ( E_0) , im m ediat ely aft er ( E_1) and 24 h
aft er t reat m ent ( E_2) , using a shade guide ( VI TA classical) . St at ist ical t est ing
was accom plished using t he Mann- Whit ney U t est ( p< 0.001) . The dropout UDWHZDV5HVXOWV7KHUHZHUHQRVLJQL¿FDQWGLIIHUHQFHVDW(BEHWZHHQ placebo and t est groups regarding t he t oot h color. Differences in t oot h color FKDQJHVLPPHGLDWHO\DIWHUƩ(BDQGKDIWHUWUHDWPHQWƩ(BZHUH calculat ed for bot h groups. The m ean values ( st andard deviat ions) of t oot h FRORUFKDQJHVIRUƩ(BZHUHLQWKHWHVWJURXSDQG LQWKHSODFHERJURXS7KHFRORUFKDQJHVIRUƩ(BVKRZHGPHDQYDOXHVRI LQWKHWHVWJURXSDQGLQWKHSODFHERJURXS)RUƩ(B DQGƩ(BVLJQL¿FDQWGLIIHUHQFHVZHUHIRXQGEHWZHHQWKHJURXSV&RQFOXVLRQ I n t his short - t erm st udy, t he result s show ed t hat a non- hydrogen peroxide EOHDFKLQJDJHQWKDVVLJQL¿FDQWZKLWHQLQJHIIHFWVLPPHGLDWHO\DQGKDIWHU a single- use t reat m ent .
Ke y w o r d s: Ef f icacy. Hy d r og en p er ox id e. Dou b le- b lin d st u d y. Toot h
bleaching. Side effect s. Mozhgan BIZHANG1
Julia DOMIN1
Gholamreza DANESH2
Stefan ZIMMER1
http://dx.doi.org/10.1590/1678-7757-2016-0463
1Universität Witten/Herdecke, Department of Operative and Preventive Dentistry, Witten, Germany. 2Universität Witten/Herdecke, Department of Orthodontics, Witten, Germany.
I nt r oduct ion
Accor ding t o r epr esent at ive dat a fr om t he Unit ed
Ki n g d o m an d f r o m a st u d y w i t h Ch i n ese u r b an
populat ion, t he prevalence of t oot h discolorat ion in
13-65 year- olds is appr ox im at ely 50%3,27. Consequent ly,
t oot h whit ening is perhaps t he m ost frequent ly applied
aest het ic pr ocedur e in dent ist r y.
The et iology of t oot h discolorat ions is m ult i- causal
and can r esult fr om indiv idual behav ior, diseases,
i n j u r y a n d o t h e r e x p o su r e s a l o n g w i t h v a r i o u s
p h y siolog ical p r ocesses1. Pr of ession al clean in g of
discolor ed t eet h is a com m on pr ocedur e t o r em ove
t he m aj or it y of ex t r insic st rains. Var ious bleaching
t echniques and pr oduct s ar e used t o r em ove int r insic
VWDLQV,QRI¿FHRUSRZHUEOHDFKLQJKRPHEOHDFKLQJ
and over- t he- count er bleaching pr oduct s.
Most bleaching pr oduct s use hydr ogen per ox ide
as act iv e ag en t . How ev er, b leach in g t r eat m en t s
w it h per ox ide m ay cause local adver se effect s such
as oral m ucosa ir r it at ion, pulpal sensit iv it y, pulpit is
or alt erat ion of t he enam el sur face14. On t he ot her
hand, bleaching is a r elat ively safe pr ocedur e t hat
pr edom inant ly causes sev er e adv er se effect s only
at high hy dr ogen per ox ide concent rat ions on har d
t issue, soft t issue and r est orat iv e m at er ials15. The
(XURSHDQ6FLHQWL¿F&RPPLWWHHRQ&RQVXPHU3URGXFWV
( SCCP)4 r epor t ed t h at t h e u se of t oot h w h it en in g
pr oduct s cont aining > 0.1 t o 6.0% hydr ogen per ox ide
or equivalent hydrogen peroxide- releasing subst ances
is safe aft er consult at ion w it h a dent ist .
On 3 1 Oct ober 2 0 1 2 , t h e EU Cou n cil Dir ect iv e
2011/ 84/ EU ( am ending EU Council Dir ect ive 76/ 768/
((&VSHFL¿HGWKDW27&WRRWKZKLWHQLQJSURGXFWVDQG
k it s m ay only cont ain up t o 0.1% hydr ogen per ox ide.
How ever, t his concent rat ion is t oo low t o have any
not iceable effect on t he color of t eet h.
Legally, pr oduct s t hat cont ain m or e t han 0. 1%
hydr ogen per ox ide can only be sold t o a dent ist24. I n
Januar y 2008, t he SCCP r ecom m ended 6% hydr ogen
per ox ide as a safe lim it t o use for hom e bleaching
if super v ised by a dent ist24. Toot h bleaching based
on non- per ox ide sy st em s ar e available as over- t
he-count er pr oduct s in t he for m of gels, r inses, gum s,
GHQWLIULFHV ZKLWHQLQJ VWULSV RU SDLQWRQ ¿OPV10,15,24. Auschill, et al.5 H[DPLQHG WKH HI¿FDF\ DQG side effect s of t hr ee differ ent bleaching t r eat m ent s:
Whit est r ips ( OTC pr oduct , 5.3% hydr ogen per ox ide) ,
Opalescence PF 10% ( hom e bleaching, 10% carbam ide
p er ox id e) an d Op alescen ce Xt r a Boost ( in - of f ice
b l e a ch i n g , 3 8 % h y d r o g e n p e r o x i d e ) . Th e si d e
effect s w er e r ever sible and t he t est pr oduct s had no
har m ful effect on t he t oot h sur faces. Addit ionally, t he
t reat m ent s were effect ive in rem oving int rinsic st ains4,
but given t he act ual legislat ion, such pr oduct s ar e no
longer allow ed t o be sold as OTC pr oduct s w it hout
super v ision of a dent ist . Var ious st udies invest igat ed
t he effect iveness, safet y and possible r ebound effect
of at - hom e and OTC agent s. The m ost com m on side
effect s aft er bleaching were dent al hypersensit ivit y and
gingival ir r it at ion180RUHRYHUWKHHI¿FDF\RISURGXFWV con t ain in g h y d r og en p er ox id e is g en er ally b ased
on cum ulat ive and r epeat ed t r eat m ent s; no inst ant
RU RQHWUHDWPHQW HI¿FDF\ KDV EHHQ UHSRUWHG 1R
clinical st udies w er e found for non- per oxide bleaching
pr oduct s. Only one st udy ex am ined a non- per ox ide
at - hom e bleaching pr oduct based on sodium chlor ide
in vit r o and obser ved det er iorat ing effect s on dent al
enam el20.
A n ov el OTC b l each i n g ag en t b ased o n n o n
-peroxide gel was invest igat ed in t his st udy. I t cont ains
pht halim ido per ox y capr oic acid ( PAP) and calcium
lact at e gluconat e as act ive ingr edient s and has a high
pot ent ial of ox idat ion w it h r elease of act ive ox y gen.
The gel should be placed on a r eady- t o- use t ray. The
calcium - lact at e- gluconat e is an im plem ent ed com plex
t o r em ineralize and conser ve dent al har d t issue. A
com binat ion of bleaching and r em ineralizat ion agent s
ZDV HI¿FLHQW LQ UHGXFLQJ GHQWDO K\SHUVHQVLWLYLW\8,23. Thus, t he pur pose of t his random ized, double- blind,
an d p laceb o- con t r olled sh or t - t er m st u d y w as t o
GHWHUPLQH WKH HI¿FDF\ DQG VLGH HIIHFWV RI D QRYHO
OTC non- per ox ide bleaching agent aft er a single- use
applicat ion.
I n t his double- blind st udy, t he pr im ar y goal was
t o t est t h e h y p ot h esis t h at t h er e ar e sig n if ican t
differ ences in t oot h color unit s ( 1- 16 unit s) bet w een
a n ov el OTC n on - per ox ide bleach in g an d placebo
im m ediat ely aft er a single- use applicat ion r egar ding
t he effect iveness of bleaching. The secondar y goal
was t o det er m ine t he color change as w ell as t he side
effect s bet w een baseline and aft er 24 h plus bet w een
Mat er ial and Met hods
Et hical appr oval
Wr it t en infor m ed consent was obt ained fr om all
par t icipant s, and all pr ocedur es of t he st udy w er e
clearly explained in advance. The st udy was conduct ed
accor ding t o t he Declarat ion of Helsink i pr inciples for
m edical research involving hum an subj ect s. The st udy
was appr oved by t he et hical com m it t ee of Wit t en/
Her decke Univer sit y, Ger m any ( No. 26/ 2014) , w her e
t he full t r ial pr ot ocol can be accessed. The st udy has
t he follow ing Ger m an Clinical Tr ials Regist er num ber :
DRKS00007636, dat e: 7 Januar y 2015. The sam ple
size w as calcu lat ed u sin g G* Pow er ( v er sion 3 . 0 ;
ht t p: / / w w w. psy cho. uni- duesseldor f. de/ abt eilungen/
aap/ gpow er 3)12. The num ber of par t icipant s for t he
pr im ar y out com e was set at 20 per gr oup t o allow for
dr opout and t o ensur e an adequat e pow er of 80%
and an alpha er r or of 0 . 0 5 t o det ect a differ ence
of one- shade guide unit s and 0.5 unit s of st andar d
dev iat ion bet w een t he t w o gr oups w it h an effect size
of 1. Met hods w er e applied in accor dance w it h t he
appr oved guidelines.
St udy populat ion
For t y volunt eer s ( 15 m en and 25 w om en) w it h
a m ean age of 35.1± 14.5 year s par t icipat ed in t his
VWXG\7KH¿UVWVXEMHFWVIURPYROXQWHHUVIURP
scr een in g ex am in at ion w h o w er e in t er est ed an d
DJUHHG WR SDUWLFLSDWH DQG IXO¿OOHG WKH LQFOXVLRQ DQG
exclusion cr it er ia w er e asked t o sign t he consent for m
and enrolled in t he st udy. The subj ect s received dent al
pr ophy lax is, t hen a single average shade scor e was
calculat ed for each individual. This st udy was a
double-blind random ized parallel st udy, and subj ect s w er e
random ly assigned t o one of t he t wo groups, balancing
for gender and baseline t oot h color. Random izat ion was
done w it h a sealed envelope sy st em by a per son w ho
was not involved in t he t r ial. The random izat ion was
VWUDWL¿HGDFFRUGLQJWRIRXUFRPELQDWLRQVRIFRORUVKDGH
and gender ( fem ale & average shade color darker t han
A3/ fem ale & average shade color br ight er t han A3/
m ale & average shade color dar ker t han A3/ m ale &
average shade color bright er t han A3) . The part icipant s
w er e given random ly generat ed t r eat m ent allocat ions
w it hin sealed opaque envelopes.
Test gr oup ( n= 20) : iWhit e I nst ant ( Sy lphar N.V.,
Deur le, Belgium ) w it h bleaching agent s;
Placebo gr oup ( n= 20) : iWhit e I nst ant ( Sy lphar
N. V., Deu r le, Belg iu m ) w it h ou t b leach in g ag en t s
( in gr edien t of t h e placebo agen t : aqu a, gly cer in ,
sor bit ol, pot assiu m acesu lf am e, ch on dr u s cr ispu s
p ow d er, h y d r at ed silica, ar om a, m et h y l p ar ab en ,
ant i- ox idant ) .
The ingr edient s of t he pr oduct s used in t his st udy
ar e list ed in Figur e 1. The pr oduct s had t he sam e
pack aging.
The key list t hat docum ent ed t o w hich gr oup t he
subj ect s belonged was kept by a t hir d per son and
was not r evealed t o t he clinical ex am iner unt il t he
st udy w as com plet ed. No par t icipant indicat ed t he
pr esence of a sy st em ic disease in his/ her m edical
h ist or y. An in t r aor al ex am in at ion con f ir m ed t h at
each par t icipant had an age bet w een 18- 65 year s, at
least 22 nat ural t eet h w it h no cur r ent car ies act iv it y,
gingivit is, periodont al disease, or ot her oral pat hology.
The fr ont t eet h should not be light er t han A2 and
w it hout defect ive enam el st r uct ur e or r est orat ions.
Exclusion cr it er ia w er e t he pr esence of any sy st em ic
d isease, p r eg n an cy or b r east f eed in g , t h e u se of
¿[HGRUUHPRYDEOHRUWKRGRQWLFDSSOLDQFHVVPRNLQJ
alcohol abuse, aller gic r eact ion t o any com ponent of
t he agent s, t he use of any bleaching agent s w it hin
t he last year and dent al hy per sensit iv it y. Subj ect s
w er e in st r u ct ed t o u se t h e sam e n on - w h it en in g
t oot h past e ( Sen sody n e Flu or ide, Glax oSm it h Klin e
Consum er Healt hcar e Gm bH & Co. KG, Ham bur g,
Germ any) and a soft t oot hbrush ( Sensodyne Precision;
GlaxoSm it hKline Consum er Healt hcar e Gm bH & Co.
KG; Ham bur g, Ger m any ) t hr oughout t he ex per im ent .
Whit ening pr oduct s and t he consum pt ion of cur r y,
coffee, black t ea or r ed w ine w er e st r ict ly for bidden
dur ing t he st udy.
Calibrat ion of t he exam iner
All exam inat ion procedures and subj ect inst ruct ions
were perform ed by t he sam e experienced clinician ( JD)
t o avoid int er ex am iner differ ences. Befor e t he st udy,
t he clinician was calibrat ed on 20 subj ect s t o ensur e
t he validit y of t he t oot h color m easurem ent s using t he
VI TA shade guide ( Vit a Zahnfabr ik , Bad Säck ingen,
Ger m any ) . Th e secon d t oot h color m easu r em en t s
w er e per for m ed aft er a t w o- w eek br eak. The st r engt h
of agr eem en t w as assessed by calcu lat in g Kappa
YDOXHV7KHLQWUDH[DPLQHUFRUUHODWLRQFRHI¿FLHQWZDV
0.83; hence, t he st r engt h of agr eem ent can be rat ed
Exam inat ion
(I¿FDF\ WRRWK FRORU DQG VDIHW\ VLGH HIIHFWV
su ch as t oot h sen sit iv it y an d g in g iv al ir r it at ion )
w er e assessed at baseline ( E_0) , im m ediat ely aft er
t r eat m en t ( E_ 1 ) an d 2 4 h aft er t r eat m en t ( E _ 2 ) .
Toot h color was m easured using t he VI TA Lum in shade
guide ( Vit a Zahnfabr ik , Bad Säck ingen, Ger m any ) ,
w hich consist s of t he 16 m ost com m on t oot h color s
( 1- 16) . The exam iner assessed pat ient s’ condit ions by
deploy ing a blast of air t o t eet h isolat ed w it h cot t on
r olls, fr om a dist ance of 1 cm , for 1 s. The scale was
used as follow : 0 = absence of pain and 1 = pr esence
of pain ( sligh t , m oder at e an d sev er e sen sit iv it y ) .
*LQJLYDOLUULWDWLRQLHDQ\DEQRUPDO¿QGLQJVVXFKDV
r edness, edem a or epit helial ir r it at ion of soft t issues
was r ecor ded by t he ex am iner. The nex t param et er s
w er e self- r epor t s for t oot h sensit iv it y and gingival
LUULWDWLRQ DW WKH ¿QDO H[DPLQDWLRQ 7RRWK VHQVLWLYLW\
in clu d ed p ain af t er t h e con su m p t ion of cold an d
hot beverage and/ or food r espect ively and gingival
irrit at ion involved any discom fort or gingival irrit at ion.
The scale r ecor ded as 0 = absence of pain and 1 =
pr esence of pain.
Tr eat m en t s w er e p er f or m ed accor d in g t o t h e
m an u fact u r er s’ in st r u ct ion s. Th e t est an d placebo
gels w er e placed on a r eady- t o- use t ray for six upper
and six low er ant er ior t eet h. The exam iner super vised
t he subj ect s t o ensur e cor r ect applicat ion. They w er e
not allow ed t o r em ove t he t ray s dur ing t he t r eat m ent
session but t hey w er e allow ed t o t alk . Aft er 20 m in,
t he t ray s w er e r em oved and discar ded. Teet h w er e
cleaned fr om r em nant s of t he gel. Ther eaft er, t he
t oot h color, t oot h hyper sensit ivit y and t he condit ion of
WKHVRIWWLVVXHZHUHUHFRUGHG7KHÀXRULGHJHO(OPH[
Gelée, CP GABA, Ham bur g, Ger m any ) was applied
VXEVHTXHQWO\IRU¿YHPLQXWHV7KH¿QDOH[DPLQDWLRQ
was per for m ed aft er 24 h.
The individual color det erm inat ions were evaluat ed
by on e ex am in er u n der iden t ical con dit ion s u sin g
t he VI TA Lum in shade guide ( Vit a Zahnfabr ik , Bad
Säck ingen, Ger m any ) . The VI TA shade guide ser ved
as r efer ence t o t he clinical sit uat ion and consist s of
t he 16 m ost com m on t oot h color s sor t ed accor ding t o
shade5,18. The m iddle one- t hir d of t he facial sur face
of t he upper and low er ant er ior t eet h was m easur ed
t o select t he shade. A single average shade scor e of
Products Ingredients Function
Test gel $TXD Emulsion stabilizer
Hydrated Silica Abrasive
Glycerin Solvent
pthalimido peroxy capronacid (PAP) Bleaching agents
Sorbitol plasticizer
Chondrus crispus powder/Hydrated Silica Abrasive, thickener
PEG-40 Hydrogenated Castor Oil Emulsifying
Aroma Flavouring agent
Acrylates/Arcylamide Copolymer and Mineral Oil and Polysorbate 85
Stabilizes emulsions
Citric acid acid
Methyl Paraben Preservative
Calcium-lactate-gluconate remineralization
Xylitol Sweetener
Potasium Acesulfame Sweetener
Placebo gel $TXD Emulsion stabilizer
Glycerin Solvent
Sorbitol plasticizer
Potasium Acesulfame Sweetener
Chondrus crispus powder/Hydrated Silica Abrasive, thickener
Aroma Flavouring agent
Methyl Paraben Preservative
BHT Anti-oxidant, masking agent
upper and low er ant er ior t eet h w as calculat ed for
each indiv idual.
A clinical exam inat ion of t he oral and perioral region
was conduct ed t o r eveal any signs of adver se changes
t o t eet h or soft t issue aft er t r eat m ent , in addit ion t o
WKH TXHVWLRQQDLUHV DW WKH ¿QDO H[DPLQDWLRQ 7RRWK
hy per sen sit iv it y an d or al ir r it at ion w er e t h e m ost
im por t ant issues. These r epr esent t he m ost com m on
adver se event s associat ed w it h v it al bleaching9. The
severit y and durat ion of adverse event s were recorded.
St at ist ical analysis
St at ist ical analy ses w er e per for m ed by st at ist ical
soft w ar e ( SPSS for Window s v er sion 2 0 , Super ior
Perform ing Soft ware Syst em s, Chicago, USA) . For each
gr oup, m ean t eet h color shade at differ ent t im es of
ex am inat ion was calculat ed w it h st andar d dev iat ion
an d 9 5 % con f id en ce in t er v als. Th e Kolm og or ov
-Sm ir nov t est show ed an inhom ogeneous dist r ibut ion
of t he dat a. We applied non- param et r ic t est s due t o
t he asym m et r ic dat a dist r ibut ion. The non- param et r ic
Man n - Wh it n ey U t est w as u sed f or an aly sis. Th e
VLJQL¿FDQFHOHYHOZDVVHWDWp< 0.05. The analy sis of
t he secondar y dat a was car r ied out in a descr ipt ive
m anner.
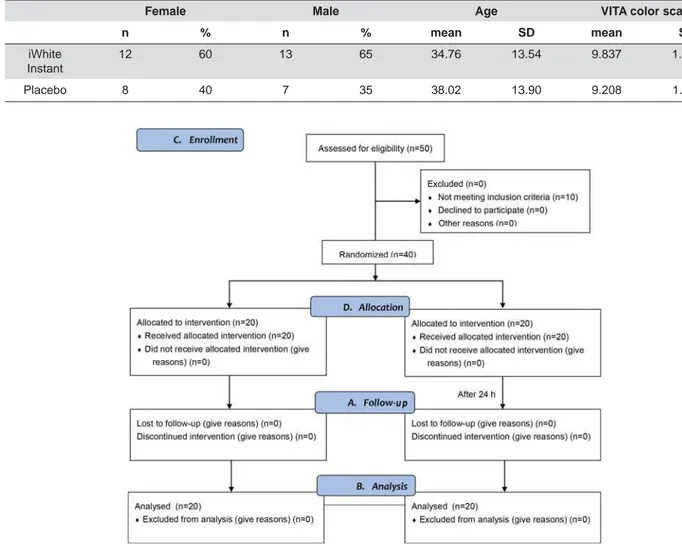
Result s
Fort y subj ect s bet ween 18 and 64 years part icipat ed
in t h is st u d y. Baselin e d em og r ap h ic an d clin ical
charact er ist ics for each gr oup ar e show n in Table 1.
7KHGURSRXWTXRWHZDV1RVLJQL¿FDQWGLIIHUHQFH
i n co l o r w as f o u n d b et w een g r o u p s at b asel i n e
ex am inat ion. Aft er a single- use bleaching, t he t oot h
FRORUVKDGHIRUEDVHOLQHVKRZHGDVLJQL¿FDQWGLIIHUHQFH
bet w een iWhit e and t he placebo im m ediat ely and 24
h aft er t r eat m ent ( Figur e 2) .
Th e m e a n ( ± s t a n d a r d d e v i a t i o n , a n d 9 5 %
FRQ¿GHQFHOHYHO&, t oot h color shade E_0 was 9.84 ( ± 1.93, 95% CI = 8.94- 10.74) unit for t he t est gr oup and 9.21 ( ± 1.11, 95% CI = 8.69- 9.73) unit for
Female Male Age VITA color scale
n % n % mean SD mean SD
iWhite Instant
12 60 13 65 34.76 13.54 9.837 1.928
Placebo 8 40 7 35 38.02 13.90 9.208 1.114
Table 1- Distribution of subjects by gender, age, and VITA color scale at baseline examination
Figure 3- Values of the tooth color of the bleaching product and the placebo for baseline examination, immediately and 24 h after EOHDFKLQJQVQRWVLJQL¿FDQWS
Figure 4- Distribution of shade improvement for the iWhite Instant between baseline and immediately after bleaching (a) per teeth (b) per
t he placebo gr oup. The m ean t oot h color shade E_1 was 7.58 ( ± 1.66, 95% CI = 6.80- 8.36) unit for t he t est gr oup and 9.22 ( ± 1.16, 95% CI = 8.68- 9.76) unit for t he placebo gr oup. The m ean of t oot h color shade E_2 was 7.79 ( ± 1.73, 95% CI = 6.78- 8.60) unit for t he t est gr oup and 9.31 ( ± 1.01, 95% CI = 8.84- 9.79) unit for t he placebo gr oup.
The t oot h color shade change for t he placebo group
VKRZHG QR VLJQL¿FDQW GLIIHUHQFHV EHWZHHQ EDVHOLQH DQG LPPHGLDWHO\ DIWHU EOHDFKLQJ Ʃ(B EHWZHHQ LPPHGLDWHO\DQGKDIWHUEOHDFKLQJƩ(BDQG EHWZHHQEDVHOLQHDQGKDIWHUEOHDFKLQJƩ(B
( Fi g u r e 3 ) . Th e t e st g r o u p sh o w e d si g n i f i ca n t
GLIIHUHQFHVIRUƩ(BDQGƩ(BEXWQRWIRUƩ(B
I n t he t est group, 39% of t he exam ined t eet h had a
shade im pr ovem ent of four shade values im m ediat ely
aft er t r eat m ent , 17% had an im pr ovem ent of t hr ee
shades, and 3% had an im provem ent of seven shades.
Am ong t he ex am ined t eet h, 41% show ed no shade
value im pr ovem ent ( Figur e 4a) . When assessed per
indiv idual, 5% of t he t est gr oup show ed an average
LPSURYHPHQW RI ¿YH VKDGHV KDG DQ DYHUDJH
im pr ovem ent of t hr ee shades, 50% an average of t w o
shades, and 15% an average of one shade ( Figure 4b) .
The exam iner obser ved t w o subj ect s ( im m ediat ely
aft er t r eat m ent ) and t hr ee ( 24 h aft er t r eat m ent )
w it h gingival ir r it at ion on soft t issue dur ing t he st udy
in t he t est gr oup. None of t he subj ect s had dent al
hy per sensit iv it y. Thr ee subj ect s in t he placebo gr oup
w er e diagnosed w it h hy per sensit iv it y and t hr ee w it h
gingival irrit at ion ( im m ediat ely aft er t reat m ent ) ( Table
2) .
Tw o subj ect s in t he t est gr oup r epor t ed gingival
ir r it at ion, one subj ect gingival edem a im m ediat ely
af t er t r eat m en t an d on e su b j ect r ep or t ed t oot h
hy per sensit iv it y im m ediat ely aft er t r eat m ent . These
adver se event s had disappear ed aft er 24 h ( Table
2 ) . I n t he placebo gr oup, t hr ee subj ect s r epor t ed
“ no com m ent ” inst ead of “ no gingival ir r it at ion” or
“ gingival ir r it at ion”.
Discussion
Bleaching is an ox idat ive pr ocess t hat alt er s t he
OLJKW DEVRUSWLRQ RU UHÀHFWLRQ RQ WKH WRRWK VXUIDFH
The bleaching t r eat m ent is int ended t o be par t of a
com plet e dent al pr ocedur e. The dent ist ’s exam inat ion
is n ecessar y t o en su r e t h e cor r ect in dicat ion s for
bleach in g, an d h e/ sh e m on it or s t h e t r eat m en t t o
DYRLGVLJQL¿FDQWVLGHHIIHFWV19. A double- blind placebo-cont r olled clinical st udy was conduct ed t o evaluat e
init ial color im provem ent aft er a single- use applicat ion
of t he newly m arket ed non- peroxide bleaching syst em .
The r esult s of our st udy dem onst rat ed t hat t he
non-per ox ide bleaching sy st em is effect ive.
Ger lach and Zhou13 ( 2001) show ed t hat t he m or e
yellow t he t eet h at t he baseline t he bet t er t he out com e
of t h e t oot h bleach in g1 3. Th er efor e, t h e bleach in g
gr oups w er e balanced for baseline t oot h color and
age. The t est bleaching product s m ay pot ent ially cause
ir r ever sible dam age if used on a long- t er m basis; so
w e decided t o per for m a single- use applicat ion under
t he super v ision of a dent ist .
The t oot h color alt erat ions w er e assessed using Examiner iWhite Instant (E_1) iWhite Instant (E_2) Placebo (E_1) Placebo (E_2)
n % n % n % n %
No hypersensitivity 20 100 20 100 17 85 20 100
hypersensitivity 0 0 0 0 3 15 0 0
No gingival irritation 18 90 17 85 17 85 18 90
Gingival irritation 2 10 3 15 3 15 2 10
Self-report iWhite Instant (E_2) Placebo (E_2)
n (%) n (%)
No hypersensitivity 19 95 20 100
Hypersensitivity 1 5 0 0
No comment 0 0 0 0
No gingiva irritation 17 85 17 85
Gingiva irritation 3 15 0 0
No comment 0 0 3 15
a shade guide and t he v isual evaluat ion fr om one
ex am iner, w hich is in accor dance w it h ot her clinical
st udies6,22. Visual inspect ion is a clinical subj ect ive
m et hod17 WKHUHIRUH WKH .DSSD FRHI¿FLHQW YDOXH IRU int raexam iner reliabilit y was assessed before t he st art
RIWKHVWXG\±HQVXULQJDKLJKUHOLDELOLW\RIWKH
evaluat ed t oot h color s25. I nt er fer ence by sur r ounding
light is a disadvant age for t he color det er m inat ion by
m eans of a shade guide. The color im pr ession of a
WRRWKGHSHQGVRQWKHWUDQVOXFHQF\WKHUHÀHFWLRQDQG
t he adsor pt ion of t he light on t he t oot h sur face. The
t oot h color was exam ined under st andardized light and
VXUURXQGLQJLQÀXHQFHVWRHOLPLQDWHDUWHIDFWV
The inv est igat ed nov el bleaching agent ( iWhit e
I nst ant ) consist s of pht halim ido per ox y capr oic acid
( PAP) , an or ganic peracid cont aining a high pot ent ial
of ox idat ion. Ox idat ion is necessar y for t he bleaching
pr ocedur e as it neut ralizes or ganic double bonds t hat
cause dent al discolorat ions. But t he act iv it y of PAP is
not based on t he release of hydrogen peroxide11. Risks
LQYROYHGLQDWKRPHDQGLQRI¿FHEOHDFKLQJSURFHGXUHV
ar e t oot h hy per sen sit iv it y an d gin gival ir r it at ion s.
These side effect s depend on t he concent rat ion of
t he bleaching agent and t he cont act bet w een t he
t ray and t he gingiva9. Ther efor e, a m ucosal ir r it at ion
or dent al hy per sensit iv it y m ay also occur aft er t he
u se of n on - per ox ide bleach in g agen t s. No den t al
h y p e r se n si t i v i t i e s a n d m u co sa l i r r i t a t i o n s w e r e
obser ved by t he ex am iner im m ediat ely and 24 h aft er
t he single use of t he new bleaching agent . Fluor ide
was applied aft er t he t reat m ent which m ay also have a
rem ineralizing effect on t he enam el7. But self- report ed
dent al hy per sensit iv it y and m ucosal ir r it at ion indicat e
t hat t hese side effect s have t o be ex am ined in
long-t er m slong-t udies.
7KHUH ZDV D VLJQL¿FDQW GLIIHUHQFH EHWZHHQ WKH
a ct i v e b l e a ch i n g a g e n t a n d t h e p l a ce b o . To o t h
w hit ening using at - hom e OTC bleaching agent s has
already been exam ined in several st udies6,16,26 and was
show n t o be effect ive for w eak t oot h discolorat ions16.
I n t his st udy, 59% of all ex am ined t eet h bet w een
baselin e an d im m ediat ely af t er bleach in g sh ow ed
an im pr oved color shade in t he t est gr oup ( Figur e
5) . Never t heless, not all t eet h w er e hom ogenously
bleached depending on t he posit ion and indiv idual
charact er ist ics like discolorat ion pat t er n or anat om ic
st r uct ur e. The r eady- t o- use t ray m ay also be a r eason
The color st abilit y aft er bleaching has been lar gely
FRQ¿QHGWRZHHNVRUPRQWKV2,6,28.
The bleaching effect is well known and depends not
only on t he bleaching m et hod but also on subj ect s’
food and lifest y le habit s21. We invest igat ed t he shor t
-t er m effec-t of -t he novel OTC non- per ox ide bleaching
DJHQWKDIWHUEOHDFKLQJ1RVLJQL¿FDQWFRORUFKDQJH
w as obser ved w it hin 24 h aft er bleaching. Fur t her
st udies about t he effect iveness aft er 14 day s of t he
applicat ion and a long- t erm color st abilit y aft er m ont hs
ar e needed for t he non- hydr ogen per ox ide bleaching
agent .
The single use and t he shor t - t er m obser vat ion for
t he effect of color st abilit y w er e t he lim it at ions of t his
st udy, w hich, on t he ot her hand, w er e necessar y t o
assess t he init ial effect iveness of t he new bleaching
agent . The single applicat ion of t he novel pr oduct
VKRZHGDVLJQL¿FDQWHIIHFWZKLFKZDVQRW\HWUHSRUWHG
for hydr ogen per ox ide pr oduct s.
Conclusions
Wit hin t he lim it at ions of t his st udy, t he r esult s
sh o w e d t h a t si n g l e - u se b l e a ch i n g co n t a i n i n g a
pht halim ido per ox y capr oic acid ( PAP) agent y ielded
VLJQL¿FDQWLQLWLDOZKLWHQLQJFRPSDUHGWREDVHOLQHDQG
placebo. These r esult s r em ained st able in t im e, e.g.
24 h aft er applicat ion.
Acknow ledgem ent s
We t hank all of t he part icipant s, w it hout w hom t his
st udy could not have been conduct ed. We t hank
Dipl.-Dolm Chr ist ina Wagner, Wit t en/ Her decke Univer sit y,
for pr oofr eading t he m anuscr ipt .
&RQÀLFWRILQWHUHVWDQGVRXUFHRIIXQGLQJVWDWHPHQW $OODXWKRUVGHFODUHWKDWWKH\KDYHQRFRQÀLFWRI
int erest s. The st udy was support ed by Sylphar ( Deurle,
Belgiu m ) . Th e com pany pr ov ided t h e f u n din g f or
m at erials and a com pensat ion for t he part icipant s. The
com pany was not involved in t he st udy design, t he
dat a collect ion and analy sis, t he decision t o publish,
or t he pr eparat ion of t he m anuscr ipt .
Refer ences
1 - Ad d y M, Mor an J. Ex t r in sic t oot h d iscolor at ion b y m et als an d chlor hex idine. I I . Clinical st aining pr oduced by chlor hex idine, ir on and
t ea. Br Dent J. 1985; 159: 331- 4.
2 - Al Sh et h r i S, Mat is BA, Coch r an MA, Zek on is R, St r opes M. A FOLQLFDO HYDOXDWLRQ RI WZR LQRI¿FH EOHDFKLQJ SURGXFWV 2SHU 'HQW 2003; 28: 488- 95.
3 - Alk h at ib MN, Holt R, Bedi R. Pr evalen ce of self- assessed t oot h discolourat ion in t he Unit ed Kingdom . J Dent . 2004; 32: 561- 6. $PHULFDQ 'HQWDO $VVRFLDWLRQ &RXQFLO RQ 6FLHQWL¿F $IIDLUV 7RRWK w hit ening/ bleaching: t r eat m ent considerat ions for dent ist s and t heir
pat ien t s [ in t er n et ] . 2 0 0 9 . [ cit ed 2 0 1 6 Nov. 1 1 ] . Av ailable f r om : KWWSVZZZDGDRUJaPHGLD$'$$ERXWWKH$'$)LOHV ada_house_of_delegat es_w hit ening_r epor t .ashx .
$XVFKLOO70+HOOZLJ(6FKPLGDOH66FXOHDQ$$UZHLOHU1%(I¿FDF\ side- effect s and pat ient s' accept ance of differ ent bleaching t echniques 27&LQRI¿FHDWKRPH2SHU'HQW
%L]KDQJ00OOHU03KDUN-+%DUNHU0/*HUODFK5:&OLQLFDOWULDO of long- t er m color st abilit y of hydr ogen per ox ide st r ips and sodium SHUFDUERQDWH¿OP$P-'HQW6SHF1R$$$
%L]KDQJ06HHPDQQ5'XYH*5|PKLOG*$OWHQEXUJHU-0-DKQ KR, et al. Dem ineralizat ion effect s of 2 bleaching procedures on enam el VXUIDFHVZLWKDQGZLWKRXWSRVWWUHDWPHQWÀXRULGHDSSOLFDWLRQ2SHU Dent . 2006; 31: 705- 9.
8- Borges AB, Torres CR, Souza PA, Caneppele TM, Sant os LF, Magalhães $&%OHDFKLQJJHOVFRQWDLQLQJFDOFLXPDQGÀXRULGHHIIHFWRQHQDPHO er osion suscept ibilit y. I nt J Dent . 2012; 2012: 347848.
9- Bruzell EM, Pallesen U, Thoresen NR, Wallm an C, Dahl JE. Side effect s of ex t er nal t oot h bleaching: a m ult i- cent r e pract ice- based pr ospect ive
st udy. Br Dent J. 2013; 215( 9) : E17.
10- Dem ar co FF, Meir eles SS, Masot t i AS. Over- t he- count er w hit ening agent s: a concise r ev iew. Braz Oral Res. 2009; 23 Suppl 1: 64- 70.
1 1 - Fässler M, Meissn er K, Sch n eider A, Lin de K. Fr equ en cy an d cir cum st ances of placebo use in clinical pract ice - a sy st em at ic r ev iew
of em pir ical st udies. BMC Med. 2010; 8: 15.
12- Faul F, Er dfelder E, Buchner A, Lang AG. St at ist ical pow er analy ses
using G* Pow er 3. 1: t est s for cor r elat ion and r egr ession analy ses. Behav ior Res Met hods. 2009; 41: 1149- 60.
13- Gerlach RW, Zhou X. Vit al bleaching wit h whit ening st rips: sum m ary of clinical r esear ch on effect iveness and t olerabilit y. J Cont em p Dent
Pract . 2001; 2( 3) : 1- 16.
1 4 - Goldber g M, Gr oot v eld M, Ly n ch E. Un desir able an d adv er se
effect s of t oot h- w hit ening pr oduct s: a r ev iew. Clin Oral I nv est ig. 2010; 14( 1) : 1- 10.
15- Hasson H, I sm ail AI , Neiva G. Hom e- based chem ically- induced w h i t e n i n g o f t e e t h i n a d u l t s . Co c h r a n e D a t a b a s e Sy s t Re v.
2006; 18( 4) : CD006202.
16- Hor n BA, Bit t encour t BF, Gom es OM, Far hat PA. Clinical evaluat ion
of t he w hit ening effect of over- t he- count er dent ifr ices on v it al t eet h. Braz Dent J. 2014; 25: 203- 6.
17-Judeh A, Al- Wahadni A. A com par ison bet w een convent ional v isual and spect r ophot om et r ic m et hods for shade select ion. Quint essence
I nt . 2009; 40( 9) : e69- 79.
18- Leonar d RH Jr, Bent ley C, Eagle JC, Gar land GE, Knight MC, Phillips &1LJKWJXDUGYLWDOEOHDFKLQJDORQJWHUPVWXG\RQHI¿FDF\VKDGH r et ent ion. side effect s, and pat ient s' per cept ions. J Est het Rest or Dent .
2001; 13: 357- 69.
19- Li Y. Safet y cont r over sies in t oot h bleaching. Dent Clin Nor t h Am .
2011; 55( 2) : 255- 63.
20- Maj eed A, Gr obler SR, Moola MH, Ober holzer TG. Effect of four
21- Mat is BA, Cochran MA, Franco M, Al- Am m ar W, Ecker t GJ, St r opes 0(LJKWLQRI¿FHWRRWKZKLWHQLQJV\VWHPVHYDOXDWHGin vivo: a pilot st udy. Oper Dent . 2007; 32: 322- 7.
22- O'Br ien WJ, Gr oh CL, Boenke KM. One- dim ensional color or der sy st em for dent al shade guides. Dent Mat er. 1989; 5( 6) : 371- 4. 3LQWDGR3DORPLQR.7LUDSHOOL&7KHHIIHFWRIKRPHXVHDQGLQRI¿FH bleaching t reat m ent s com bined wit h experim ent al desensit izing agent s
on enam el and dent in. Eur J Dent . 2015; 9( 1) : 66- 73.
6FLHQWL¿F&RPPLWWHHRQ&RQVXPHU3URGXFWV(XURSHDQ&RPPLVVLRQ Opinion on hydr ogen per ox ide in t oot h w hit ening pr oduct s [ int er net ] . 2005. [ cit ed 2016 Nov. 11] . Available fr om : ht t ps: / / ec.eur opa.eu/
healt h/ ph_r isk / com m it t ees/ 04_sccp/ docs/ sccp_o_022.pdf.
25- Sim J, Wr ight CC. The k appa st at ist ic in r eliabilit y st udies: use, int erpret at ion, and sam ple size requirem ent s. Phys Ther. 2005; 85:
257-68.
26- Sopar k ar P, Rust ogi K, Zhang YP, Pet r one ME, DeVizio W, Pr osk in
HM. Com parat ive t oot h w hit ening and ex t r insic t oot h st ain r em oval HI¿FDF\ RI WZR WRRWK ZKLWHQLQJ GHQWLIULFHV VL[ZHHN FOLQLFDO WULDO -Clin Dent . 2004; 15( 2) : 46- 51.
27-Xiao J, Zhou XD, Zhu WC, Zhang B, Li JY, Xu X. The pr evalence
of t oot h discolourat ion and t he self- sat isfact ion w it h t oot h colour in a Chinese ur ban populat ion. J Oral Rehabil. 2007; 34( 5) : 351- 60.