Appliations of the Shwinger Multihannel Method
with Pseudopotentials to Eletron Sattering from
Polyatomi Moleules I. Elasti Cross Setions
Alexandra P. P. Natalense,Mario T. doN. Varella,
InstitutodeFsiaGlebWataghin,
UniversidadeEstadual deCampinas,UNICAMP,
13083-970Campinas,S~aoPaulo,Brazil
Mario H. F. Bettega,
Departamento deFsia,UniversidadeFederaldoParana,
Universidade FederaldoParana
CaixaPostal19044, 81531-990Curitiba,Parana, Brazil
Luiz G. Ferreira,and Maro A. P. Lima
InstitutodeFsiaGlebWataghin,
UniversidadeEstadual deCampinas,UNICAMP,
13083-970Campinas,S~aoPaulo,Brazil
Reeivedon14Marh,2000
Thispaperis adatabasis whihinludes tables ofintegral, dierential,and momentumtransfer
rosssetionsforelastieletronsatteringfromCF
4 ,CCl
4 ,SiCl
4 ,SiBr
4 ,SiI
4 ,CH
3 FCH
2 F
2 ,CHF
3 ,
CH3Cl, CH2Cl2, CHCl3,CF3Cl, CF2Cl2,CFCl3,CH3Br,CH3I,SiH3Cl, SiH3Br, SiH3I,GeH3Cl,
GeH
3
Br, SnH
3 Br, C
2 H
6 , Si
2 H
6 , Ge
2 H
6 , B
2 H
6 , Ga
2 H
6 , H
2 O, H
2 S, H
2 Se, H
2
Te, trimethylarsine
(TMAs), N2O, and O3. These tables show our new results, along with some of our previously
publishedross setions, andan easilybeomparedto futureexperimentaldata andothernew
theoretial results. Our sattering amplitudes werealulated usingthe Shwinger multihannel
method with norm-onserving pseudopotentials. Our results are in good agreement with other
theoretialdataandexperimentalresultswhenavailable.
All tables are available in the eletroni version of the paper only [on the world wide web at
http://www.sbf.if.usp.br/bjp/Vol31/Num1/.℄
I Introdution
In this paper we show appliations of the Shwinger
multihannel methodwith norm-onserving
pseudopo-tentials[1℄toeletronsatteringfromCF
4 ,CCl
4 ,SiCl
4 ,
SiBr
4 , SiI
4 , CH
3 F CH
2 F
2 , CHF
3 , CH
3
Cl, CH
2 Cl
2 ,
CHCl
3 ,CF
3 Cl,CF
2 Cl
2 ,CFCl
3 ,CH
3 Br,CH
3 I,SiH
3 Cl,
SiH
3
Br, SiH
3
I, GeH
3
Cl, GeH
3
Br, SnH
3 Br, C
2 H
6 ,
Si
2 H
6 , Ge
2 H
6 , B
2 H
6 , Ga
2 H
6 , H
2 O, H
2 S, H
2 Se, H
2 Te,
trimethylarsine(TMAs),N
2
O,andO
3
. Manyofthese
moleules are plasmaproessing gases[2, 3℄and some
arealsoofenvironmental interestforbeinggreenhouse
gases or stratospheri ozonedepleting gases [3℄.
De-spite their industrial and environmental importane,
areverysare. Toourknowledge,thisistherst
ol-letion of eletron sattering ross setions for many
ofthe moleules ited above. We intend to presentin
this paper aomplete data base of our results, whih
aneasilybeomparedtofutureexperimentaldataand
othernewtheoretialresults.
InsetionIIwepresentabriefreviewofthetheory
anddesribethemain approximationsusedin our
al-ulations. In Setion III we present our results and
disussion. This setion is divided as follows:
Sub-setion III-1 shows our elasti dierential ross
se-tions(DCS)forCF
4 ,CCl
4 ,SiCl
4 ,SiBr
4
andSiI
4 from
Ref. [4℄, in addition to results for other impat
se-setions (MTCS) for these moleules. Subsetion
III-2 inludes tables with elasti DCS from Ref. [5℄ for
CH 3 FCH 2 F 2 ,CHF 3 , CH 3 Cl,CH 2 Cl 2 ,CHCl 3 ,CF 3 Cl, CF 2 Cl 2
, and CFCl
3
along with our results for other
eletron impat energies. Subsetion III-3 shows our
newresultsonelastidierentialandmomentum
trans-ferrosssetionsforthemoleulesXH
3
Y,withX=C,
Si, Ge, Sn; Y = F, Cl, Br, I. Results of our previous
studies[6℄onX
2 H
6
(X=C,Si,Ge)arepresentedin
Sub-setion III-4. In Subsetion III-5 we presentour new
elastieletronsatteringresultsforGa
2 H
6
andtables
withourDCSandourICSforB
2 H
6
fromRef.[6℄. Our
MTCSforB
2 H
6
,whihwerenotinludedinRef.[6℄are
alsoshown. SubsetionIII-6presentsourDCSforH
2 X
(X=O,S,Se,Te)fromRef. [7℄. InSubsetionIII-7we
showourresultsfortrimethylarsine(TMAs)[8℄.
Elas-tirosssetionsforN
2
O[9℄andO
3
[10℄arepresented
inSubsetionIII-8.
II Theory
The implementation of pseudopotentials in the
Shwinger multihannel method allows alulationsof
low-energyeletronsatteringbymoleulesontaining
atomswithmanyeletronswithreduedomputational
eort [1℄. The basi idea is to replae the ore
ele-trons andthenuleusofeah atomin themoleuleby
the orresponding soft norm-onserving
pseudopoten-tialandtodesribethevaleneeletronsinaquantum
hemistry framework (Hartree-Fok approximation in
the present implementation). The ross setions for
eletron sattering by moleules with dierent atoms
butwiththesamenumberofvaleneeletronsanthen
bealulatedwithaboutthesameomputationaleort.
To illustrate this idea, Table I shows the total
num-ber of eletrons for eah studied moleule ompared
to the number of valene eletrons. The method an
providesubstantialomputationalsaving,espeiallyfor
moleulesontainingmanyheavierthanHenters,suh
asCFCl
3
forexample.
The Shwinger multihannel method has been
de-sribed previously and we only review here some key
featuresforompleteness. Inthismethod,theworking
expressionforthesatteringamplitudeis
[f ~ k i ; ~ k f ℄= 1 2 X m;n hS ~ k f jVj m i(d 1 ) mn h n jVjS ~ k i i; (1) where d mn =h m jA (+) j n i (2) and A (+) = ^ H
N+1 (
^
HP+P ^
H)
2
+
(VP+PV)
2 VG (+) P V: (3)
IntheaboveequationsjS
~
k
i
iistheprodutofa
tar-get state and a plane wave, V is the interation
po-tential between the inident eletron and the target,
j
m
i is a (N+1)-eletron Slater determinant used in
theexpansionofthetrialsatteringwavefuntion, ^
His
thetotalenergyoftheollisionminusthefull
Hamilto-nianofthesystem,P isaprojetionoperatorontothe
open hannel spae dened by target eigenfuntions,
andG (+)
P
isthefree-partileGreen'sfuntionprojeted
ontheP-spae.
Inour formulation all thematrix elementsneeded
toevaluatethesatteringamplitudeareomputed
an-alytially,exept thoseinvolvingtheGreen's funtion,
i.e. h m jVG (+) P Vj n
i,whih arealulatedby
numer-ialquadrature [11℄.
We use the norm-onserving pseudopotentials of
BaheletHamannandShluter[12℄todesribethe
nu-lear potential and the ore eletrons of eah atom.
These pseudopotentialswereimplementedin theSMC
method asdesribed in Ref. [1℄. TheCartesian
Gaus-sianfuntionsusedtodesribethemoleularand
sat-tering orbitals were espeially designed to be used in
ourpseudopotentialalulations[13℄.
Ourrosssetionswereobtainedinthexed-nulei
stati-exhangeapproximation. Wedonotinlude the
desriptionofpolarizationeets,sinetheyareknown
to beof littleimportane for theimpatenergy range
westudy here(5{30 eV).Forwater moleule, wealso
present stati-exhange DCS in the (2-5)-eV energy
rangebeause,in thisase,polarizationeets arenot
soimportant,sinetheexisting long{rangepermanent
dipole moment potential is known to dominate
low-energy satteringfor thissystem [14℄. Wealsodo not
inludeinthisalulationanyorretiontoaountfor
the dipole potential ofthe polarmoleules, exeptfor
theH
2
X (X=O,S, Se, Te) moleules andTMAs,for
whih we have ombined the Shwinger multihannel
methodwithaBornlosureproedure[7,8℄. Themain
ontribution of this longrange potential to the
dier-ential ross setions is at very low sattering angles,
where the ontribution of high partial waves is more
important.
III Results and Disussion
III.1 CF 4 , CCl 4 , SiCl 4 , SiBr 4
, and SiI
4
Ourelastidierentialrosssetions(DCS)for
ele-tronsatteringfromCF
4 , CCl 4 ,SiCl 4 ,SiBr 4
, andSiI
4
fromRef.[4℄arepresentedinTablesIItoVI,alongwith
areinverygoodagreementwithavailableexperimental
data [15℄ (see Ref. [4℄). To ourknowledge, exept for
CF
4
,therearenootherDCSforthesemoleulesinthe
literatureforomparison.
InTablesVIIandVIIIweshowourintegralelasti
rosssetionsandourunpublishedmomentumtransfer
rosssetionsforCF
4 ,CCl
4 ,SiCl
4 ,SiBr
4
,andSiI
4 .
III.2 Fluoromethanes, Chloromethanes
and Chlorouoromethanes
Inthissubsetionwepresenttableswithour
dier-entialrosssetionsofRef.[5℄forCH
3 FCH
2 F
2 ,CHF
3 ,
CH
3 Cl, CH
2 Cl
2 , CHCl
3 , CF
3 Cl, CF
2 Cl
2
, and CFCl
3
and new results for many other eletronimpat
ener-gies. Asanillustration,Fig.1omparesourdierential
ross setionsfor CF
2 Cl
2 , CF
3 Cl, CH
3
Cl, andCH
2 F
2
at seleted impat energies with the experimental
re-sultsofRef.[16,17,18,19℄respetivelyandthe
agree-ment is very good even for impat energies as low as
5eV.Although notshownhere,ourresultsforCH
3 Cl
are also in good agreement with the results obtained
with the omplex Kohn variational method [20℄ (see
Ref. [5℄).
Figure 1. Our dierential ross setions for seleted
moleulesandenergiesomparedtoexperimentaldata.Full
lines: ourtheoretialresults;triangles: CF
2 Cl
2
experimen-taldata[16 ℄;diamonds: CF
3
Clexperimentaldata[17 ℄;
ir-les: CH
3
Clexperimentaldata[18 ℄;squares: CH
2 F
2
exper-imentaldata[19 ℄.
InFigs. 2and 3weompare dierential ross
se-tions for uoromethanes and hloromethanes
respe-tively,bothat10eVimpatenergy. Wealsoinludeour
resultsforCF
4 ,CCl
4
andCH
4
[1℄. Wehaveshown
pre-viously [5℄ that the moleules with largerouteratoms
present more osillations in the dierential ross
se-tions than the other ones espeially for high impat
energies. This behaviorindiates that the presene of
largerouteratomsinreasestherangeofthepotential,
andfavorstheouplingofhigherpartialwaves. Figs.2
and3showthatthis behaviorisalsopresentforlower
impatenergies,althoughlessevident.
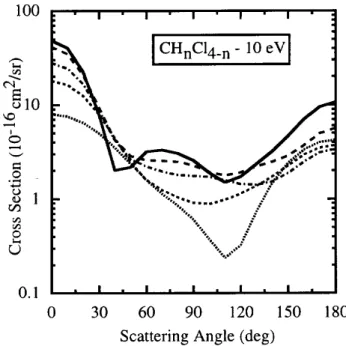
Figure2. DierentialrosssetionsforCHnF4
n
at10eV.
Fullline: CF4[4 ℄;longdashedline: CF3H;dot{dashedline:
CF2H2;shortdashedline: CFH3;dottedline: CH4 [1 ℄.
Figure3.DierentialrosssetionsforCH
n Cl
4 n
at10eV.
Full line: CCl
4
[4℄; long dashed line: CCl
3
H; dot{dashed
line: CCl
2 H
2
; short dashed line: CClH
3
; dotted line:
CH
4 [1 ℄.
TablesIX to XVII show our dierential ross
CF
3 Cl,CF
2 Cl
2
,andCFCl
3
respetively,forseveral
im-patenergies.
III.3 XH
3
Y (X = C, Si, Ge, Sn; Y = F,
Cl, Br, I)
In this subsetion we present aomparativestudy
ofdierentialandmomentumtransferrosssetionsfor
CH
3 F, CH
3 Cl, CH
3 Br, CH
3 I, SiH
3
Cl, SiH
3
Br, SiH
3 I,
GeH
3
Cl, GeH
3
Br, and SnH
3
Br. To our knowledge,
there are no theoretial nor experimental results for
these moleules in the literature for omparison,
ex-eptforCH
3
F[19℄andCH
3
Cl[18,20℄,asdisussedin
SubsetionIII-2above.
Fig. 4 ompares our dierential ross setions at
20eVfor CH
3
Brand CH
3
I to ourpreviousresultsfor
CH
3
F, and CH
3
Cl [5℄. The dierential ross setion
for CH
3
Fis signiantlydierent from the resultsfor
theothermoleules. CH
3 Cl,CH
3
Br,andCH
3
Ipresent
verysimilar dierential ross setions, with small
dif-ferenesonlyintheforwardandbakwarddiretions.
Figure4. Dierentialrosssetionsfor CH3Y (Y=F,Cl,
Br,I)at20eV.Fullline: CH3F[5 ℄;dottedline: CH3Cl [5 ℄;
dashedline: CH3Br;dot{dashedline: CH3I.
InFig.5weompareourdierentialrosssetions
at 20 eV for SiH
3
Cl, SiH
3
Br, and SiH
3
I (top graph)
and GeH
3
Cl, and GeH
3
Br(bottom graph). Forthese
twosets of moleules theperipheral atoms have little
inuene on the dierential ross setions, exept for
highsatteringangles.
Fig.6showstheinueneoftheentralatomonthe
dierentialrosssetions at 20eV. Thethree pitures
show that dierententral atoms produelittle
dier-enes in therosssetions,exept forCH
3
I andSiH
3 I
(bottomgraph). Inthisase,thepreseneofsilionin
themoleuleinsteadof arbonintroduesundulations
intherosssetions,whih areharateristiofhigher
Figure5. Dierentialross setionsat20eV. Top: results
for SiH3Y (Y=Cl, Br, I). Full line: SiH3Cl; dotted line:
SiH3Br;dashedline: SiH3I.Bottom: resultsforGeH3Y (Y
=Cl,Br). Fullline: GeH3Cl; dot{dashedline: GeH3Br.
Figure6. Dierentialrosssetionsat20eV.Topleft:
re-sultsforXH
3
Cl(X=C,Si,Ge). Fullline: CH
3
Cl; dashed
line: SiH3Cl; dot{dashed line: GeH3Cl. Topright: results
for XH3Br (X = C, Si, Ge, Sn). Full line: CH3Br;
dot-ted line: SiH3Br; dashed line: GeH3Br; dot-dashed line:
SnH3Br. Bottom: resultsforXH3Cl(X=C,Si). Fullline:
CH3Cl;dashedline: SiH3Cl.
TablesXVIIIto XXV presentourdierentialross
setions for all the XH
3
Y{type of moleules we have
Ta-rosssetions.
III.4 X
2 H
6
(X=C, Si, Ge)
Elasti integral and dierential ross setions for
this family are presented in Tables XXVII, XXVIII
and XXIX. These moleules were subjet of previous
studiesbyourgroupusingtheSMCPPmethod[6℄.
III.5 B
2 H
6
and Ga
2 H
6
B
2 H
6
isusedasgaspreursorinproessesof
hemi-alvapordeposition[21℄andwasthesubjetofprevious
studies[6℄. Inthissubsetionwepresenttableswithour
previousresultsfor B
2 H
6
[6℄ andelasti eletron
sat-teringresultsforGa
2 H
6
forthersttime. Fig.7shows
elastiintegralrosssetionforB
2 H
6
andGa
2 H
6 from
5eV to30eV. Therosssetionfor Ga
2 H
6
liesabove
the result for B
2 H
6
, and presentsno struture in this
energyrange. TheintegralrosssetionforB
2 H
6 shows
averybroadfeaturearound10eV.
Figure 7. Elasti integral ross setions for B
2 H
6 and
Ga
2 H
6
. Solidline: Ga
2 H
6
;dashedline: B
2 H
6 .
Fig.8omparesdierentialelastirosssetionsfor
these two moleules for seleted energies. These DCS
arealsoshowninTablesXXXandXXXIalongwithour
results for other eletron impat energies. The DCS
for these two moleules are dissimilar, the results for
Ga
2 H
6
beingrihin osillationsdue tohigher angular
momentum ouplingespeially at higherimpat
ener-Figure 8. Dierential elasti ross setions for B2H6 and
Ga2H6at5eV,10eV,25eV,and30eV.Solidlines:Ga2H6;
dashedlines: B
2 H
6 .
III.6 H
2
X (X = O, S, Se, Te)
Inthissubsetion wepresentourelastidierential
rosssetions (DCS)forH
2
X(X=O,S, Se,Te)from
Ref.[7℄(TablesXXXIItoXXXV).Thelong{range
po-tentialduetothepermanentdipolemomentofthe
tar-gets(H
2
OandH
2
S)wasdesribedthroughaBorn
lo-sureproedure.OurDCSforH
2
OandH
2
Sareingood
agreementwithavailableexperimentaldataand
previ-ousalulations(seeRef.[7℄). Tableswithourintegral
rosssetionsandmomentumtransferrosssetionsfor
thesemoleulesareshownin Ref.[7℄.
III.7 As(CH
3 )
3
{ Trimethylarsine
(TMAs)
Thetrimethylarsinemoleuleanbefoundin
innu-merable dierent onformations, sine the three CH
3
groupsanrotatearoundtheAs{Chemialbond. In
our previous work on elasti eletron sattering from
TMAs [8℄wehaveshownthat, althoughthedierene
betweenthetwoseletedonformations(referene
on-formation(RC)andlowestenergyonformation(LEC))
issimplytherelativepositionsof thehydrogenatoms,
theeletronsatteringrosssetionsaresensitivetothe
onformationofthetargetforimpatenergiesbetween
4eV and15eV. Inthisenergy range,one should
per-formanaverageoverallpossibletarget onformations
in order to ompare alulated ross setions and
ex-perimentaldata. Above15eV,however,thereseemsto
benorelevantonformationaleet.
TableXXXVI presentsourdierentialelastiross
setionsand Table XXXVII showsour elastiintegral
III.8 N
2
O, and O
3
In this subsetion we present elasti rosssetions
for N
2
O [9℄ and O
3
[10℄ moleules. These moleules
were subjetofpreviousstudies. ForO
3
wehaveused
the rst Born approximation to orret the
dieren-tialrosssetionsatsmallsatteringanglesduetothe
inuene of the moleular permanent dipole moment.
For N
2
O we have not used this proedure. In
Ta-blesXXXVIII and XXXIXwepresentourdierential,
integralandmomentumtransferrosssetionsforN
2 O
andO
3
respetivelyatseletedenergies.
Tables
All tables are available only in the eletroni
version of the paper on the world wide web at
http://www.sbf.if.usp.br/bjp/Vol31/Num1/.
Aknowledgments
A.P.P.N.aknowledgessupportfromFunda~aode
AmparoaPesquisadoEstadodeS~aoPaulo(FAPESP).
M.H.F.B.,L.G.F.andM.A.P.L.aknowledge
par-tial support from Brazilian agenyConselho Naional
de Desenvolvimento Ciento e Tenologio (CNPq).
M. T. N. V. aknoledges both FAPESP and CNPq.
OuralulationswereperformedatCENAPAD-SP,at
CENAPAD-NEandat CCE-UFPR.
Referenes
[1℄ M. H. F. Bettega, L. G. Ferreira, and M. A. P.Lima,
Phys.Rev.A47,1111 (1993).
[2℄ M. Hayashi \Swarm Studies and Inelasti
Eletron-Moleule Collisions", edited by L. C. Pithford,B. V.
MKoy,A.Chutjian,andS.Trajmar,NewYork,p.167
(1987).
[3℄ See, for example, L. G. Christophorou, J. K. Oltho,
and M. V. V. S.Rao, J. Phys. Chem. Ref. Data, 25,
1341(1996);L.G.Christophorou,J.K.Oltho,andM.
V.V.S.Rao,J.Phys.Chem.Ref.Data,26,1(1997);L.
G.Christophorou,J.K.Oltho,andY.Wang,J.Phys.
Chem.Ref.Data,26,1205 (1997).
[4℄ M. T.doN.Varella,A.P.P.Natalense, M.H. F.
Bet-tega,L. G.Ferreira,andM.A.P.Lima,Phys.Rev.A,
aeptedforpubliation(1999).
[5℄ A. P. P. Natalense, M. H. F. Bettega, L. G.Ferreira,
andM.A.P.Lima,Phys.Rev.A,59,879(1999).
[6℄ M. H.F.Bettega,A.J.S.Oliveira,A.P.P.Natalense,
M. A.P.Lima,L. G.Ferreira,Eur.Phys.J.D. 3,291
(1998).
[7℄ M. T.doN.Varella,M.H.F.Bettega,M. A.P.Lima,
and L.G.Ferreira, J.Chem.Phys.aeptedfor
publi-ation(1999).
[8℄ M.T.doN.Varella,L.G.Ferreira,andM.A.P.Lima,
J.Phys.B32,2031 (1999).
[9℄ S.M.S.daCosta andM.H. F.Bettega, Eur.Phys.J.
D3,67(1998).
[10℄ M.H.F.Bettega,M.T.doN.Varella,M.A.P.Lima,
andL.G.Ferreira,J.Phys.B31,4419 (1998).
[11℄ M.A.P.Lima,L.M.Bresansin,A.J.R.daSilva,C.
Winstead,andV.MKoy,Phys.Rev.A41,327(1990).
[12℄ G.B.Bahelet,D.R.Hamann,andM.Shluter,Phys.
Rev.B26,4199(1982).
[13℄ M. H.F.Bettega,A. P.P.Natalense, M.A.P.Lima,
and L. G. Ferreira, Int. J. Quantum Chem., 60, 821
(1996).
[14℄ T. N. Resignoand B. H. Lengseld,Z. Phys.D 24,
117(1992).
[15℄ L. Boesten, H. Tanaka, A. Kobayashi, M. A. Dillon,
andM.Kimura,J.Phys.B25,1607(1992).
[16℄ A.Mann,andF.Linder,J.Phys.B,25,1633(1992).
[17℄ A.Mann,andF.Linder,J.Phys.B,25,1621(1992).
[18℄ X.Shi,V. K.Chan,G.A.Gallup, andP.D. Burrow,
J.Chem.Phys.,104,1855(1996).
[19℄ H. Tanaka,T.Masai, M. Kimura,T.Nishimura,and
Y.Itikawa,Phys.Rev.A56,R3338(1997).
[20℄ T.N.Resigno,A.E.Orel,andC.W.MCurdy,Phys.
Rev.A,56,2855,(1997).
[21℄ I.Konyashin,J.Bill,andF.Aldinger,Chem.Vap.