rev bras hematol hemoter. 2015;37(3):211–214
w w w . r b h h . o r g
Revista
Brasileira
de
Hematologia
e
Hemoterapia
Brazilian
Journal
of
Hematology
and
Hemotherapy
Letter
to
the
Editor
Somatic
mutations
of
calreticulin
in
a
Brazilian
cohort
of
patients
with
myeloproliferative
neoplasms
DearEditor,
Essentialthrombocythemia(ET),polycythemiavera(PV),and primarymyelofibrosis (PMF) are Philadelphia chromosome-negativemyeloproliferativeneoplasms(MPNs)characterized by increased myeloid proliferation. The gain of function inducedbythe Januskinase2mutation,JAK2V617F,hasbeen
reportedinmostPV andinmorethan halfofETand PMF cases.1However,thepresenceofdifferentdiseasephenotypes
andtheabsenceoftheJAK2mutationinsomeMPNssuggests thatadditionalgeneticlesionsor/andaberrantsignaling path-waysmaybeinvolvedinthepathogenesisofthesediseases.1,2
In December 2013, somatic mutations inthe calreticulin
(CALR)genewere identifiedinETand PMFpatients bytwo independent groups3,4 and confirmed by others.5–9 CALR
mutations have been reported as mutually exclusive with
JAK2and MPLmutationsandmay bepresent in56–88%of
JAK2/MPL-negativecases.3,4Arecentpaperreportedapatient
thathadbothmutations,JAK2V617FandaCALRexon9
muta-tion,simultaneously.10OverthirtydifferentCALRmutationsin
exon9havebeendescribed,butthemostfrequentmutations (about80%)maybeclassifiedastype-1[L367fs*46;deletionof 52basepairs(bp)]andtype-2(K385fs*47;insertionof5bp).3,4
Thefunctionalchangesinducedbythemutationsarestillnot completelyelucidated,buttheoverexpressionofthetype1
CALRmutationinBa/F3cells(anIL3dependentcellline)leads tocytokine-independentcellgrowthand STAT5activation.4
GeneexpressionsignaturestudiesalsoindicatethatJAK2and
CALR mutations share mechanisms ofmalignant transfor-mation,reaffirmingacentralroleoftheJAK/STATsignaling pathwayinthepathogenesisofMPN.11
The aim of the present study was to characterize the prevalenceofCALR mutationsand the clinical and labora-torialcharacteristicsofCALR-mutatedpatientsinaBrazilian cohortofMPNs.Seventy-threeMPNpatientswereincludedin thestudy(ET=32,PV=20,PMF=21).Patients’characteristics aredescribedinTable1.Peripheralbloodsampleswere col-lected,submittedtohemolysis,andDNAwasextractedbythe
phenol/chloroformmethod.Allsampleswereinvestigatedfor
JAK2andCALRmutations.TheJAK2V617FmutationandCALR
exon9mutationswereverifiedaspreviouslydescribed.12,13 CALRmutationswereclassifiedastype-1(deletionof52bp), type-2(insertionof5bp)orothers(Figure1).
Inourcohort,CALRmutationswerefoundin20patients
(13ETand7PMF;Table1)andweremutuallyexclusivewith
JAK2V617F:20/73(27%)oftotalMPNpatientsand20/28(71%)
oftheJAK2WTpatients. AmongtheCALR-mutatedpatients,
Type-1CALRmutationswerefoundin50%(10/20;8ETand2 PMF),type-2in40%(8/20;4ETand4PMF),andothersin10% (1ETand1PMF)oftheindividuals.CALRmutationswerenot detectedinPVpatients(allJAK2V617Fpositive).InET,CALRMUT
patientsshowedreduced hemoglobinlevelscomparedwith
JAK2V617Fpatients(p-value<0.01;Table2);nodifferenceswere observedinwhitebloodcell,neutrophilandplateletcounts, thromboticevents,hepatomegaly,splenomegalyand consti-tutionalsymptoms.InPMFpatients,CALRmutationalstatus wasnotassociatedwithclinicalfeatures(Table2).The fre-quencyofCALRmutationsinthisBraziliancohortwassimilar to previously described frequencies.3,4,7 Furthermore,CALR
andJAK2V617Fmutationsweremutuallyexclusive.Knowledge
regardingtheclinicalimpactofCALRmutationsinMPNsis stillunderconstruction,butsomestudiesindicatethatCALR -mutated patients have lower ages at disease onset, lower hemoglobinandplateletcounts,andhavebetteroverall sur-vivalthaneitherJAK2-mutatedorCALR/JAK2/MPLwild-type patients.5,9,14Inourcohort, CALRMUTETpatientspresented
lowerhemoglobinlevels,comparedwithJAK2V617FETpatients,
even though in both groups hemoglobin values remained withinthereferencerange.
212
revbrashematolhemoter.2 0 1 5;37(3):211–214Table1–Patientcharacteristics.
MPN(n=73) ET(n=32) PV(n=20) PMF(n=21)
Age(years)–median(range): 57(19–87) 53(19–83) 70(41–88) 62(27–87)
Gender–male/female 30/43 10/22 9/11 11/10
Hemoglobin(g/dL)–median(range) 13.9(8.1–22.7) 13.6(9.7–17) 18.4(12.8–22.7) 12.5(8.1–19.9) WBCcount(×109L–1)–median(range) 9.8(2.4–43.2) 9(3.0–17.8) 11.2(6.3–25.5) 11.8(2.4–43.2)
Plateletcount(×109L–1)–median(range) 764(108–2065) 914(541–2065) 609(203–1070) 661(108–1716)
JAK2mutationstatus–n(%)
JAK2WT 28(38) 20(63) 0(0) 8(38)
JAK2V617F 45(62) 12(37) 20(100) 13(62)
CALRmutationstatus–n(%)
CALRWT 53(73) 19(59) 20(100) 14(67)
CALRMUT 20(27) 13(41) 0(0) 7(33)
MPN:myeloproliferativeneoplasms;ET:essentialthrombocythemia;PV:polycythemiavera;PMF:primarymyelofibrosis;WBC:whitebloodcell count;JAK2:Januskinase2gene;WT:wild-type;CALR:Calreticulingene;MUT:exon9mutations.
20000
245 255 265 275 285 295 305 315 230 240 250 260 270 280290300310 250 260 270 280 290 300 240 260 280 300
302 bp 297 bp 291 bp 245 bp
16000
12000
Intensity
Wild-type
B
A
Wild-type
M NTC #1 #2 #3 #4 #5 #6 #7
Type-1
Type-1 Type-2 Other
Type-2
MPN patients
MPN patients
Calreticulin
mutations
Calreticulin mutations
Other
bp
8000
4000
0
20000
16000
12000
8000
4000
0
20000
16000
12000
8000
4000
0 16000
12000
8000
4000
0
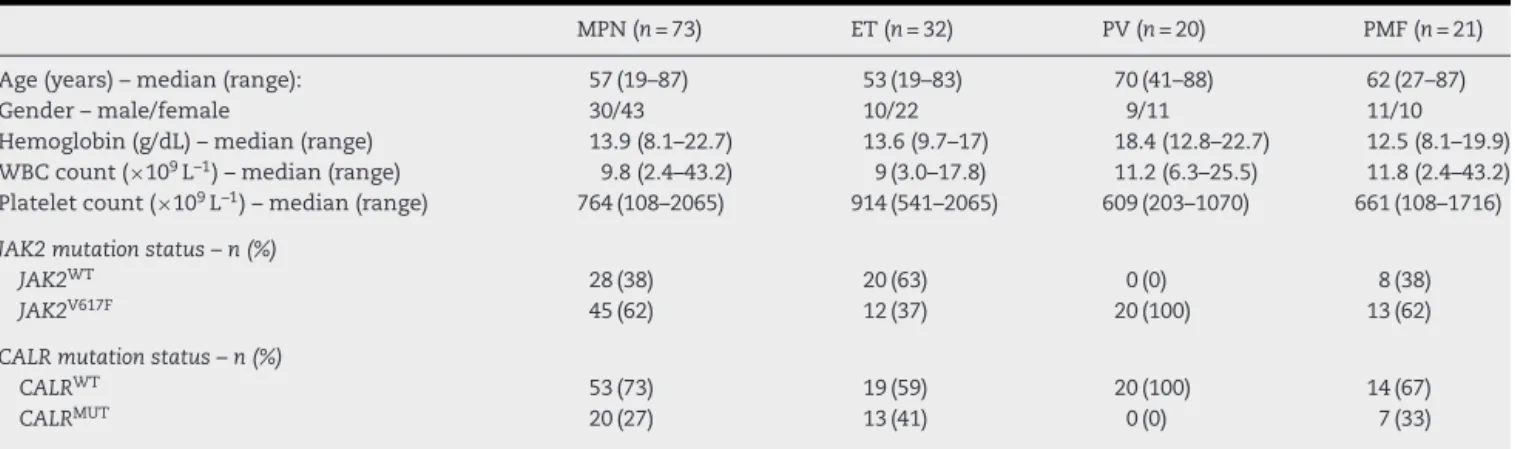
Figure1–Calreticulin(CALR)indelmutationsinmyeloproliferativeneoplasm(MPN)patients.(A)PCRamplificationofexon9 ofCALRgeneloadedon4%agarosegel;lane1:100bpmarker(M);lane2:notemplatecontrol(NTC);MPNpatients#1and#2:
CALRWT;#3and#4:heterozygotesfortype-1CALRmutation(CALRWTamplicons:297bp;type-1CALRMUTamplicons:245bp);
#5and#6:heterozygotesfortype-2CALRmutation(CALRWTamplicons:297;type-2CALRMUTamplicons:302bp);#7:
heterozygotefornon-type-1or-2CALRmutation(CALRWTamplicons:297bp;CALRMUTamplicons:291bp).(B)
revbrashematolhemoter.2 0 1 5;37(3):211–214
213
T able 2 – Clinical and labor a tory fea tur es of essential thr omboc ythemia and pr imary m y elofibr osis pa tients, str a tified accor ding to JAK2 and C ALR m uta tional sta tus. Essential thr omboc ythemia Primar y m y elofibr osis b C ALR MUT JAK2 V617F C ALR WT / JAK2 WT p -v alue a , c p -v alue a , d C ALR MUT JAK2 V617F p -v alue a , c Gender: male/female – n (%) 4 (31)/9 (69) 5 (42)/7 (58) 1 (14)/6 (86) 0.69 0.61 4 (57)/3 (43) 6 (46)/7 (54) 1.00 Ag e – y ears (r ang e) 57 (20–82) 55 (44–83) 43 (19–53) 0.61 0.13 53 (27–80) 65 (43–87) 0.30 Hemo globin – g/L (r ang e) 13 (10–15) 14 (12–17) 14 (12–16) 0.007 0.12 12 (9–13) 14 (8–20) 0.08 White b lood cells – × 10 9L –1 (r ang e) 7.1 (3–17.8) 9.2 (4.5–16.7) 9 (5–11.8) 0.64 0.69 9.4 (2.4–35.7) 11.8 (8.4–43.2) 0.38 Neutr ophils – × 10 9/L (r ang e) 4.8 (2.1–15.1) 6 (2.5–12) 5.6 (3.2–8.5) 0.43 0.48 6.2 (1–22.5) 7.1 (0.5–38.3) 0.48 Platelets – × 10 9/L (r ang e) 938 (593–2065) 880 (541–1340) 984 (617–1274) 0.37 0.87 567 (108–1716) 661 (129–1606) 0.69 Thr ombotic ev ents – n (%) 0 (0) 2 (17) 0 (0) 0.50 1.00 0 (0) 1 (8) 1.00 He patome g al y – n (%) 1 (8) 0 (0) 0 (0) 1.00 1.00 0 (0) 2 (17) 1.00 Splenome g al y – n (%) 2 (15) 1 (8) 1 (14) 1.00 1.00 5 (71) 7 (54) 0.64 Constitutional symptoms – n (%) 1 (8) 3 (25) 2 (29) 0.32 0.27 1 (14) 3 (23) 1.00 a F isher’ s e xact test wa s used for cate gorical factors with 2 le v els; Mann–Whitne y test for measur ed factors. b Onl y one patient with primar y m y elofibr osis pr esented C ALR WT / JAK2 WT . c C ALR MUT vs. JAK2 V617F . d C ALR MUT vs. C ALR WT / JAK2 WT .Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1.BaxterEJ,ScottLM,CampbellPJ,EastC,FourouclasN,
SwantonS,etal.Acquiredmutationofthetyrosinekinase
JAK2inhumanmyeloproliferativedisorders.Lancet.
2005;365(9464):1054–61.
2.ThoennissenNH,KrugUO,LeeDH,KawamataN,IwanskiGB,
LashoT,etal.Prevalenceandprognosticimpactofallelic
imbalancesassociatedwithleukemictransformationof
Philadelphiachromosome-negativemyeloproliferative
neoplasms.Blood.2010;115(14):2882–90.
3.NangaliaJ,MassieCE,BaxterEJ,NiceFL,GundemG,Wedge
DC,etal.SomaticCALRmutationsinmyeloproliferative
neoplasmswithnonmutatedJAK2.NEnglJMed.
2013;369(25):2391–405.
4.KlampflT,GisslingerH,HarutyunyanAS,NivarthiH,RumiE,
MilosevicJD,etal.Somaticmutationsofcalreticulinin
myeloproliferativeneoplasms.NEnglJMed.
2013;369(25):2379–90.
5.TefferiA,LashoTL,FinkeCM,KnudsonRA,KetterlingR,
HansonCH,etal.CALRvsJAK2vsMPL-mutatedor
triple-negativemyelofibrosis:clinical,cytogeneticand
molecularcomparisons.Leukemia.2014;28(7):1472–7.
6.RotunnoG,MannarelliC,GuglielmelliP,PacilliA,PancrazziA,
PieriL,etal.Impactofcalreticulinmutationsonclinicaland
hematologicalphenotypeandoutcomeinessential
thrombocythemia.Blood.2014;123(10):1552–5.
7.WuZ,ZhangX,XuX,ChenY,HuT,KangZ,etal.The
mutationprofileofJAK2andCALRinChineseHanpatients
withPhiladelphiachromosome-negativemyeloproliferative
neoplasms.JHematolOncol.2014;7:48.
8.ShenH,ChaoH,DingZ,FengY,CenJ,PanJ,etal.CALRand
ASXL1mutationanalysisin190patientswithessential
thrombocythemia.LeukLymphoma.2014:1–9.
9.LiB,XuJ,WangJ,GaleRP,XuZ,CuiY,etal.Calreticulin
mutationsinChinesewithprimarymyelofibrosis.
Haematologica.2014;99(11):1697–700.
10.McGaffinG,HarperK,StirlingD,McLintockL.JAK2V617Fand
CALRmutationsarenotmutuallyexclusive;findingsfrom
retrospectiveanalysisofasmallpatientcohort.BrJ
Haematol.2014;167(2):276–8.
11.RampalR,Al-ShahrourF,Abdel-WahabO,PatelJP,BrunelJP,
MermelCH,etal.Integratedgenomicanalysisillustratesthe
centralroleofJAK-STATpathwayactivationin
myeloproliferativeneoplasmpathogenesis.Blood.
2014;123(22):e123–33.
12.daSilvaRR,DominguesHatzlhoferBL,MachadoCG,LimaAS,
deAlbuquerqueDM,dosSantosMN,etal.JAK2V617F
mutationprevalenceinmyeloproliferativeneoplasmsin
Pernambuco,Brazil.GenetTestMolBiomarkers.
2012;16(7):802–5.
13.ChiJ,NicolaouKA,NicolaidouV,KoumasL,MitsidouA,
PieridesC,etal.Calreticulingeneexon9frameshift
mutationsinpatientswiththrombocytosis.Leukemia.
2014;28(5):1152–4.
14.ChenCC,GauJP,ChouHJ,YouJY,HuangCE,ChenYY,etal.
Frequencies,clinicalcharacteristics,andoutcomeofsomatic
CALRmutationsinJAK2-unmutatedessential
214
revbrashematolhemoter.2 0 1 5;37(3):211–214JoãoAgostinhoMachado-Neto1,PauladeMeloCampos1,
DulcinéiaMartinsdeAlbuquerque,FernandoFerreiraCosta, IreneLorand-Metze,SaraTerezinhaOlallaSaad,
FabiolaTraina∗
UniversidadeEstadualdeCampinas(UNICAMP),Campinas,SP, Brazil
∗Correspondingauthorat: Rua CarlosChagas, 480,13083-878
Campinas,SP,Brazil.
E-mailaddresses:[email protected],
[email protected](F.Traina).
1Bothauthorscontributedequallytothiswork.
Received19September2014 Accepted25February2015 Availableonline15April2015
http://dx.doi.org/10.1016/j.bjhh.2015.03.012