J. Braz. Chem. Soc. vol.17 número5
Texto
Imagem
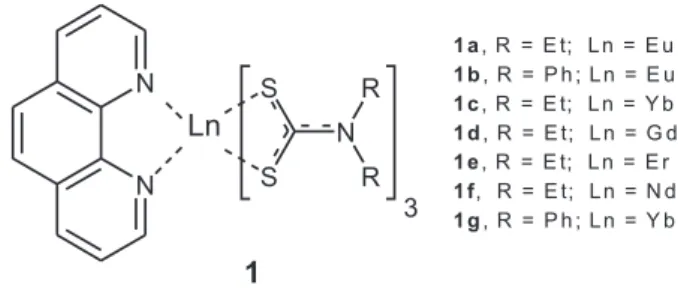
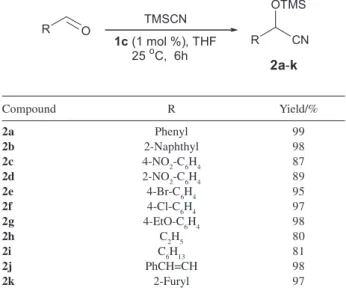
Documentos relacionados
Ao Dr Oliver Duenisch pelos contatos feitos e orientação de língua estrangeira Ao Dr Agenor Maccari pela ajuda na viabilização da área do experimento de campo Ao Dr Rudi Arno
Ousasse apontar algumas hipóteses para a solução desse problema público a partir do exposto dos autores usados como base para fundamentação teórica, da análise dos dados
i) A condutividade da matriz vítrea diminui com o aumento do tempo de tratamento térmico (Fig.. 241 pequena quantidade de cristais existentes na amostra já provoca um efeito
didático e resolva as listas de exercícios (disponíveis no Classroom) referentes às obras de Carlos Drummond de Andrade, João Guimarães Rosa, Machado de Assis,
O credor que não requerer perante o Juízo da execução a(s) adjudicação(ões) do(s) bem(ns) a ser(em) leiloado(s) antes da publicação deste Edital, só poderá adquiri- lo(s)
Extinction with social support is blocked by the protein synthesis inhibitors anisomycin and rapamycin and by the inhibitor of gene expression 5,6-dichloro-1- β-
(Eear 2018) Em alguns parques de diversão há um brinquedo em que as pessoas se surpreendem ao ver um bloco aparentemente subir uma rampa que está no piso de uma casa sem a
É uma ramificação da ginástica que possui infinitas possibilidades de movimentos corporais combinados aos elementos de balé e dança teatral, realizados fluentemente em