Revista
Brasileira
de
Hematologia
e
Hemoterapia
Brazilian
Journal
of
Hematology
and
Hemotherapy
w w w . r b h h . o r g
Original
article
Association
between
maternal
and
fetal
factors
and
quality
of
cord
blood
as
a
source
of
stem
cells
Rodrigo
Dias
Nunes
∗,
Flávia
Maria
Zandavalli
UniversidadedoSuldeSantaCatarina(UNISUL),Florianópolis,SC,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received16February2014 Accepted3July2014
Availableonline26November2014
Keywords:
Stemcells Fetalblood Transplantation
Cordbloodstemcelltransplantation Bloodbanks
a
b
s
t
r
a
c
t
Objectives:Tocomparativelyanalyzematernalandfetalfactorsandqualitymarkersofblood samplesinapublicumbilicalcordbloodbank.
Method:Thisisacross-sectionaldescriptivestudythatrevisited458recordsofdonations fromSeptember2009toMarch2013attheHemocentrodeSantaCatarina.Themeansof markerswereusedtodefinecutoffpointsforthequalityofcordblood.
Results:Mostdonationscamefromwomen withagesbetween18 and29years (62.8%), gestationalage≥40weeks(55.2%),vaginaldelivery(51.3%),primiparous(41.4%),andwith malenewborns(54.4%)weighingbetween3000and3499g(41.8%).Thevolumeofthe dona-tionsrangedfrom71.6to275.2mL,thetotalnucleatedcellcountrangedfrom4.77×108to
31.0×108cellsandCD34+cellsrangedfrom0.05to1.23%.Therewerestatisticallysignificant
differencesinthevolumewithrespecttogestationage>38weeks(p-value=0.001),cesarean section(p-value<0.001)andbirthweight>3500g(p-value<0.001).Thetotalnucleatedcell countwaspositivelyaffectedbycesareansection(p-value=0.022)andbirthweight>3500g (p-value<0.001).Therewasnostatisticallysignificantdifferencebetweenthevariablesand thepercentageofCD34+cells.
Conclusions:Deliveryrouteandbirthweightinfluencethevolumeofcordbloodandthetotal nucleatedcellcount.Gestationalageinfluencesonlythevolumeofcordblood.
©2014Associac¸ãoBrasileiradeHematologia,HemoterapiaeTerapiaCelular.Published byElsevierEditoraLtda.Allrightsreserved.
Introduction
Umbilicalcordblood,whichpreviouslyusedtobediscarded, canbecollectedshortlyafterbirthandhasgreatcapacityto reconstitutethehematopoieticsystem.1Severalstudieshave
shown the simplicityof umbilicalcordblood collection, in additiontothelackofriskforbothmotherandnewborn,low
∗ Correspondingauthorat:Av.PedraBrancan◦25,CidadeUniversitáriaPedraBranca,88137-270Palhoc¸a,SC,Brazil.
E-mailaddress:[email protected](R.D.Nunes).
riskofgraft-versus-hostdiseaseandlowriskoftransmitting infectious-contagiousdiseases.2,3Furthermore,itisauseful
alternativeofhematopoieticstemcellsfortransplantationto treatdiseasesoftheblood,immunesystemandforgenetic disorders.3,4
One of the limitations of this type of transplant is the volumeandcontentsofthebloodcollectedfromthe umbil-ical cord which is an obstacle to hematopoietic stem cell
http://dx.doi.org/10.1016/j.bjhh.2014.07.023
grafting.Themainparametersusedinumbilicalcordblood banksincludethetotalnucleatedcell(TNC)count,percentage ofCD34+cells,andthevolumeofblood.5
Transplantationsusingumbilicalcordbloodarestillinthe researchphaseandvariablesthatmightimprovethequalityof bloodarecurrentlythefocusofresearch,inasmuchas know-ingthesefactorsmayresultinlowercostsandlesswasteof timeintheevaluation,processingandstorageofmaterial.6,7
Recentstudiesreportthatsomevariablesaffectthequality oftheumbilicalcordblood,especiallythoserelatedto mater-nalandfetalfeaturessuchasplacentalweight,birthweight, gestationalage(GA),routeofdelivery,genderofthenewborn, amongotherthings;thusresearchisbeingdevelopedinthis areatoattempttoimprovecelllevels,whichisessentialto increasegraftingsuccessrates.
Objectives
Theaimsofthisstudyweretodeterminematernalandfetal characteristics of umbilical cord blood donors, to evaluate qualitymarkersofumbilicalcordblood,aswellasto deter-mineassociationsbetweenmaternalandfetalcharacteristics andthesequalitymarkers.
Methods
This is an observational, cross-sectional, epidemiological study that revisited 458 charts of patients who, between September2009andMarch2013,donatedumbilicalcordblood attheBloodUmbilicalCordandPlacentalBank(BSCUP)ofthe HemocentrodeSantaCatarina(HEMOSC),locatedinthecity ofSãoJosé,SantaCatarina.Ninemedicalrecordsofpatients whoperformedallogeneicdonationswereexcluded.Data col-lectionbeganafterthestudywasapprovedbytheResearch EthicsCommitteeofHEMOSC,anddatawerecollectedfrom computerizedregistersintheCryobiologySectorofHEMOSC, locatedinFlorianópolis,SantaCatarina.
Donationcandidatesweresubmittedtothestandard sur-veyofHEMOSCbeforecollectioninordertoassesspersonal history,familyhistory,and laboratoryresultswithstandard questionsusedforallkinds ofblood donation.Allpatients whoagreedtodonatesignedaninformedconsentformbefore thecollection.
Adatacollectioninstrumentwasdevelopedexclusivelyfor thisstudy whichinvestigatedthematernalage,gestational age,routeofdelivery,numberofpreviouspregnancies, gen-derandnewbornweight.Moreover,thepreprocessing(blood volume)andpost-processingdata(TNCandCD34+counts)of
theumbilicalcordbloodwerestudied.Themeansofmarkers wereusedtodefinecutoffpointsforthequalityofcordblood. Blood collection was performed extra-utero and carried outbyatrainedprofessionalinaseparateroom.Assoonas placental deliveryoccurs,the placenta istaken toan area reservedforcollection wheretheumbilical cordiscleaned withanantisepticsolutionandthebloodisdrainedbygravity throughthemostdistalpuncturesite.Thebloodisstoredin astandardbloodbag containingcitrate-phosphate-dextrose anticoagulant.
Oncecollected,thesesamplesarelabeledandsenttoblood banks, where theyundergo safety testing,HLAtyping and cryopreservation.Typically,collectionsarekeptforatimenot exceeding 24–28h at22±2◦C before processing, and then
cryopreserved inliquid nitrogen, under controlledfreezing ratesandstoredlongterminaccordancewithinternational criteriaatatemperaturelowerthan−150◦C.1,8,9The
umbil-ical cord blood was processed within a maximum of 48h afterbeingcollectedasisdeterminedbytheNationalHealth SurveillanceAgency(ANVISA).2
CollecteddatawerestoredintheMicrosoftExcelcomputer programandlaterexportedtotheStatisticalPackageforthe SocialSciences(Version16.0)foranalysis.
Qualitative variables aredescribed asabsoluteand rela-tivefrequencies,whilequantitativevariablesaredescribedas means±standarddeviations.
TheChi-squaredtest(2)orFisher’sexacttestwasused
totestthehomogeneityofproportions.Prevalenceratios(PR) and 95%confidenceintervals(95%CI)were calculated.The levelofsignificancewassetforap-value<0.05.
Results
Mostdonationscamefromwomenwithagesfrom18to29 years (62.8%) and from 30 to 34 years (24.4%), gestational age≥40weeks(55.2%),vaginaldelivery(51.3%),primiparous (41.4%),andwithmalenewborns(54.4%)weighingbetween 3000and3499g(41.8%).
Comparisonsbetweenmaternalandfetalcharacteristics andcordbloodvolumeareshowninTable1.
Statisticallysignificantdifferenceswerefoundinrespect to cord blood volume for gestational age>38 weeks (p -value=0.001), cesarean delivery (p-value<0.001) and birth weight>3500g(p-value<0.001).
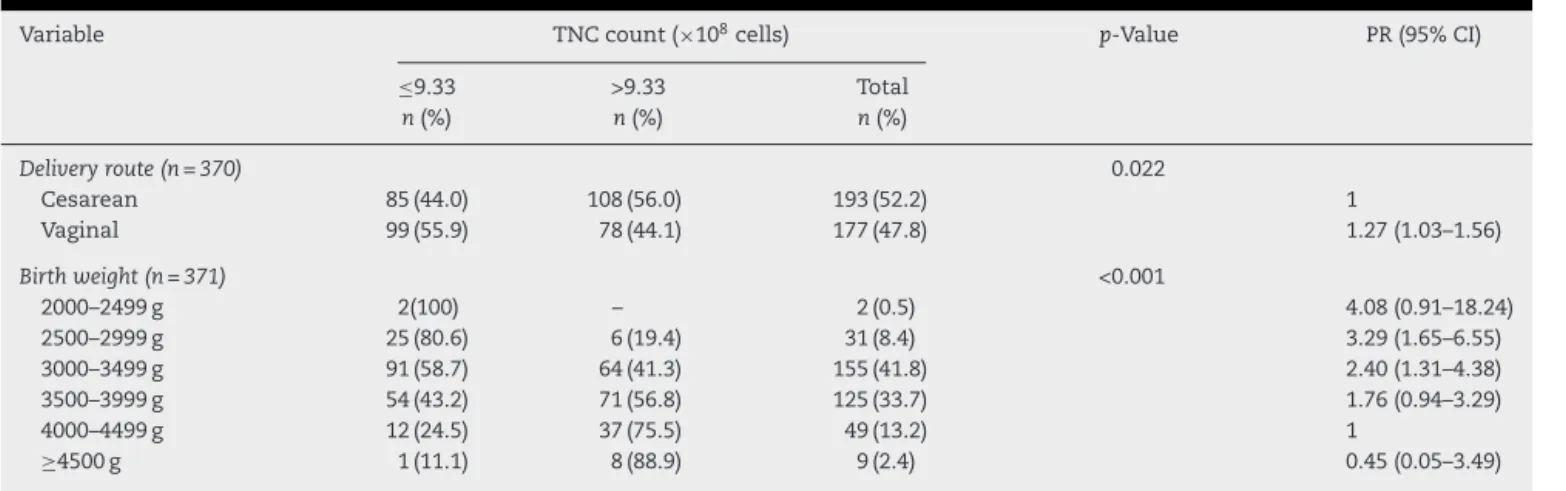
A smallervolumeofumbilical cordblood wascollected whenthegestationalagewasbelow37weeksand6days com-paredwithgestationalagesbetween38and39weeksand6 days.Vaginalrouteofdeliverywasassociatedwithasmaller volumeofcordbloodwhencomparedtocesareandelivery. Newbornweightbetween2500and3499gwasassociatedwith lower volumeofumbilicalcordblood whencomparedwith infantsweighing4000–4999g.Theprevalenceoflowervolume wasapproximatelytwotimeshigherthanthereferencevalue. Thecomparisonbetweenmaternalandfetal characteris-ticsandTNCcountispresentedinTable2.
VaginalrouteofdeliverywasassociatedwithalowerTNC countthancesareandelivery.Birthweightsbetween2500and 3499gwere associatedwithlower TNCcountscomparedto thosebetween4000and4499g,whichwereatleast2.4times greater.
Therewerenostatisticallysignificantdifferencesbetween qualitymarkers–bloodvolume,TNCcountandpercentageof CD34+.
Discussion
Table1–Maternalandfetalcharacteristicsandcordbloodvolume.
Variable Volume(mL) p-Value PR(95%CI)
≤115.20 >115.20 Total
n(%) n(%) n(%)
GA 0.001
36to37/6weeks 24(92.3) 2(7.7) 26(6.0) 1.70(1.08–2.67)
38to39/6weeks 90(54.2) 76(45.8) 166(38.8) 1.0
≥40weeks 132(55.9) 104(44.1) 236(55.2) 1.03(0.79–1.35)
Deliveryroute <0.001
Cesarean 107(49.1) 111(50.9) 218(48.7) 1.0
Vaginal 151(65.7) 79(34.3) 230(51.3) 1.34(1.04–1.71)
Birthweight <0.001
2000–2499g 3(75.0) 1(25.0) 4(0.9) 2.39(0.69–8.20)
2500–2999g 46(88.5) 6(11.5) 52(11.6) 2.82(1.60–4.98)
3000–3499g 121(64.7) 66(35.3) 187(41.8) 2.06(1.22–3.47)
3500–3999g 71(49.3) 73(50.7) 144(32.1) 1.57(0.91–2.70)
4000–4499g 16(31.4) 35(68.6) 51(11.4) 1.0
≥4500g 1(10.0) 9(90.0) 10(2.2) 0.32(0.04–2.40)
GA:gestationalage;PR:prevalenceratio;95%CI:95%confidenceinterval.
forreconstitution.Thesecellsareessentialforthetreatment ofsomehematologicneoplasias.
Inthisanalysis,otherfactorsconsideredimportant,such asalcohol intakeand smoking during pregnancy were not studiedbecausethisinformationdidnotexistinthepatient’s records.
Inthisstudy,vaginalroutewas themostcommonform ofdelivery,similartothestudybyRosenauetal.4that
inves-tigated1549units ofumbilicalcordblood,ofwhich 75.27% were collected after vaginal delivery; again similar to this study52.03%ofthenewbornsweremale.4AccordingtoOmori
etal.,1069.8%ofthedonorswereprimiparous.Thisfactmight
indicateatendencyofyoungpeopletodonateumbilicalcord bloodasmostofthesewomenwereyoungand41.4%hadno otherchild. Thisprovestheimportanceoffurtherresearch involvingumbilicalcordbloodandthefactorsthatinfluence thequality.
Themean birth weightof the newborn, according to a retrospectiveanalysisbyMancinellietal.,11 was3390gand
thereforesimilartothisstudy.
Accordingtotheresults,thegestationalageonlypositively influenced thevolumeofblood;thegreater thegestational age, the larger the volume ofumbilical cord blood.Askari etal.,inordertodemonstratewhichvariablesaffectthethree mainqualityparametersofumbilicalcordblood,showedthat agestationalageofover40weekswasapredictorforalarger volumeofblood,similartothecurrentstudy.6Shu-HuiWen
etal.demonstratedthatgestationalagewasinversely propor-tionaltothevolume.4Alongwithincreasinggestationalage,
thereisaphysiologicalplacentalsenescence,especiallyfrom 36weeksofgestationonwards.Thisplacentalagingcreates areducedoxygensupplytothefetus,releasing vasoconstric-torsubstances that,onreachingthe placenta,couldreflect ina reducedblood volumeavailableforcollectionatbirth. Inthisstudynosignificantdifferenceswerefoundbetween gestationalageandotherqualityparameters(TNCandCD34+
cellcounts).However,someauthorsfoundapositive relation-shipbetweengestationalageandtheCD34+ cellcount.12–15
Thisinformationcastsdoubtonthehypothesisthat placen-tal aging andconsequent reductionin blood supplyto the
Table2–Maternalandfetalcharacteristicsandtotalnucleatedcellcount.
Variable TNCcount(×108cells) p-Value PR(95%CI)
≤9.33 >9.33 Total
n(%) n(%) n(%)
Deliveryroute(n=370) 0.022
Cesarean 85(44.0) 108(56.0) 193(52.2) 1
Vaginal 99(55.9) 78(44.1) 177(47.8) 1.27(1.03–1.56)
Birthweight(n=371) <0.001
2000–2499g 2(100) – 2(0.5) 4.08(0.91–18.24)
2500–2999g 25(80.6) 6(19.4) 31(8.4) 3.29(1.65–6.55)
3000–3499g 91(58.7) 64(41.3) 155(41.8) 2.40(1.31–4.38)
3500–3999g 54(43.2) 71(56.8) 125(33.7) 1.76(0.94–3.29)
4000–4499g 12(24.5) 37(75.5) 49(13.2) 1
≥4500g 1(11.1) 8(88.9) 9(2.4) 0.45(0.05–3.49)
fetuscouldprovidesmallernumbersofumbilicalcordblood hematopoieticcells.Someauthorsbelievethatwith advanc-inggestational ageand consequentlywith placentalaging, thefetus would becomeprogressivelyhypoxic, resulting in defensemechanisms. Thesemechanismswouldbe respon-siblefortheincreasesinthenumberofhematopoieticcellsin umbilicalcordblood.11
When the route of delivery was analyzed, it was seen thatcesareansectionwasassociatedwithhigherTNCcounts andgreaterbloodvolumes.Thisresulthasbeenreportedby severalstudies.5,11,13,14,16 However,other authorsassociated
vaginaldeliverywithhigherTNCcounts.4,5,17Onehypothesis
forcesareansectionsbeingresponsibleforagreatervolume ofblood collectedfrom theumbilicalcordbloodisthefact thatthe newbornisplacedabove the placentabefore cord clamping,possiblycausingadownwardflowofbloodintothe umbilicalcordandconsequentlyintotheplacental compart-ment.Anotherhypothesisisthatcesareandeliverypermitsa fastmanualextractionoftheplacentatherebyreducingthe chanceofbloodclotformation.11
As expected, the weight of the newborn at birth pre-sentedapositiverelationshipwithtwolaboratoryparameters analyzed:theTNCcountandbloodvolume.Inthisstudy, sig-nificantly higherTNCcountsand volumeswere associated withbirthweightsabove3500g.Toreinforcethesefindings, other studies reported that greater birth weight positively influenced these parameters.5,11,12,17,18 Mancinelli et al., in
ordertodeterminewhichobstetricsfactorsinfluencethe qual-ityofumbilicalcordblood,showedthatahigherbirthweight wasassociatedwithbetterqualityofumbilicalcordbloodin relationtothethreemainvariables:TNCandCD34+ counts
and the blood volume.11 Other authors also reported that
thisfactorwasassociatedwithincreasedCD34+counts4,13,19;
thiswasnotconfirmedinthecurrentstudy.Thusthegreater birthweight,withaconsequentincreaseinplacentalvolume, somehowleadstoincreasedblood volumeinthe placenta. However,unlikethe resultsintheliterature,thisreasoning doesnotexplainthe correlationbetweenplacentalvolume accordingtobirthweightandthenumberofCD34+cells.
Possibly,thedeterminantofthequalityofthestemcells willbedefinedaftertheir use inpatientsand willthus be evaluatedintheclinicalpractice.Theincreasing applicabil-ityanddevelopmentoftechnologyshouldservetohighlight theresultsfoundinthisstudyandinmanyotherstudiesthat dealwiththissubject.
Conclusions
Agestationalagegreaterthanorequalto38weeks,cesarean deliveryandbirthweightgreaterthanorequalto3500g posi-tivelyinfluencethebloodvolumecollectedfromtheumbilical cord.Cesareandeliveryandbirthweightgreaterthanorequal to3500galsohavepositivecorrelationswiththeTNCcount. Noneofthematernal-fetalcharacteristicsinterferewiththe CD34+cellcount.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1.ArmsonBA.Umbilicalcordbloodbanking:implicationsfor perinatalcareproviders.JObstetGynaecolCan.
2005;27(3):263–90.
2.BariniR,FerrazUC,AcácioGL,MachadoIN.Doesthetime betweencollectingandprocessingumbilicalcordblood samplesaffectthequalityofthesample?Einstein.2011;92Pt 1:207–11.
3.GomesTL,PrankeP.Comparac¸ãoentreascélulas-troncode sanguedecordãoumbilicaldeneonatosprematurose nascidosatermo:umarevisão.RBAC.2008;40(1): 25–30.
4.WenSH,ZhaoWL,LinPY,YangKL.Associationsamongbirth weight,placentalweight,gestationalperiodandproduct qualityindicatorsofumbilicalcordbloodunits.Transfus ApherSci.2012;46(1):39–45.
5.JanRH,WenSH,ShyrMH,ChiangBR.Impactofmaternaland neonatalfactorsonCD34+cellcount,totalnucleatedcells, andvolumeofcordblood.PediatrTransplant.
2008;12(8):868–73.
6.AskariS,MillerJ,ChryslerG,McCulloughJ.Impactofdonor andcollection-relatedvariablesonproductqualityinexutero cordbloodbanking.Transfusion.2005;45(2):
189–94.
7.RosenauE,SugrueM,HallerM,FiskD,KellySS,ChangM, etal.Characteristicsofthawedautologousumbilicalcord blood.Transfusion.2012;52(10):2234–42.
8.PedrassaP,HamerschlakN.Transplantedecélulas-troncode sanguedocordãoumbilical.PratHosp.2008;55:
98–102.
9.ColdwellK,LeeS,KeanJ,KhooCP,TsaknakisG,SmytheJ, etal.Effectsofobstetricfactorsandstoragetemperatureson theyieldofendothelialcolonyformingcellsfromumbilical cordblood.Angiogenesis.2011;14(3):81–92.
10.OmoriA,HiraiM,ChibaT,TakahashiK,YamaguchiS, TakahashiTA,etal.Quality-assessmentsofcharacteristicsof placental/umbilicalcordbloodassociatedwithmaternal age-andparity-relatedfactor.TransfusApherSci.2012;46(1): 7–13.
11.MancinelliF,TamburiniA,SpagnoliA,MalerbaC,SuppoG, LasorellaR,etal.Optimizingumbilicalcordbloodcollection: impactofobstetricfactorsversusqualityofcordbloodunits. TransplantProc.2006;38(4):1174–6.
12.NakagawaR,WatanabeT,KawanoY,KanaiS,SuzuyaH, KanekoM,etal.Analysisofmaternalandneonatalfactors thatinfluencethenucleatedandCD34+cellyieldforcord bloodbanking.Transfusion.2004;44(2):262–7.
13.WuJY,LiaoC,ChenJS,XuZP,GuSL,WuSQ,etal.Analysisof maternalandneonatalfactorsassociatedwithhematopoietic reconstructionpotentialinumbilicalcordbloodunits. ZhongguoShiYanXueYeXueZaZhi.2010;18(6): 1535–41.
14.YamadaT,OkamotoY,KasamatsuH,HorieY,YamashitaN, MatsumotoK.Factorsaffectingthevolumeofumbilicalcord bloodcollections.ActaObstetGynecolScand.
2000;79(10):830–3.
15.AtanassovaV,AtanassovaM,NikolovA,ZlatkovV,
MihaylovaA,NaumovaE.Influenceofobstetricfactorsonthe qualityofcordbloodunitscollectedforallogeneic
transplantation.AkushGinekol(Sofiia).2012;51(2):11–6.
16.SparrowRL,CauchiJA,RamadiLT,WaughCM,KirklandMA. InfluenceofmodeofbirthandcollectiononWBCyieldsof umbilicalcordbloodunits.Transfusion.2002;42(2):210–5.
18.KeersmaekersCL,MasonBA,KeersmaekersJ,PonziniM, MlynarekRA.Factorsaffectingumbilicalcordbloodstemcell suitabilityfortransplantationinaninuterocollection program.Transfusion.2014;54(3):545–9.