w w w . r b o . o r g . b r
Review
Article
Viscosupplementation
for
treating
knee
osteoarthrosis:
review
of
the
literature
夽
Tiago
Youssef
Ammar,
Tomas
Araujo
Prado
Pereira,
Saulo
Luís
Lopes
Mistura,
André
Kuhn,
José
Idilio
Saggin,
Osmar
Valadão
Lopes
Júnior
∗InstitutodeOrtopediaeTraumatologiadePassoFundo,PassoFundo,RS,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received3September2014 Accepted18September2014 Availableonline5August2015
Keywords:
Knee Osteoarthritis Hyaluronicacid Viscosupplementation
a
b
s
t
r
a
c
t
Theaimherewastoevaluatetheevidencethatmightsupportorrefutetheuseof intra-articularviscosupplementationintreatingpatientswithsymptomatickneeosteoarthrosis. AreviewoftheliteraturewasconductedusingtheMedline,PubMedandCochrane Con-trolled Trial Register databases and Cochrane database systematic reviews (Cochrane Library).Onlystudiespresentingahighlevelofevidenceweretakenintoconsideration.This studyincludedanalysisonrandomizedclinicaltrialsthatincludedatleast100patientsin eachinterventiongroup,meta-analysesandsystematicreviews.Twometa-analyses,five systematicreviewsandsixrandomizedclinicaltrialsfulfilledtheinclusioncriteriaforthis review.Inthelightofthebestevidenceavailablesofar,thereisnoconsensusforindicating orevenforcontraindicatingtheuseofintra-articularviscosupplementationamongpatients withsymptomatickneeosteoarthrosis(levelofevidenceIanddegreeofrecommendation A).Furtherstudieswithappropriatemethodologyareneededtoelucidatethismatter.
©2014SociedadeBrasileiradeOrtopediaeTraumatologia.PublishedbyElsevierEditora Ltda.Allrightsreserved.
Viscossuplementac¸ão
no
tratamento
da
osteoartrose
do
joelho:
uma
revisão
da
literatura
Palavras-chave:
Joelho Osteoartrite Ácidohialurônico Viscossuplementac¸ão
r
e
s
u
m
o
Avaliarevidênciasqueapoiemourefutemousodeviscossuplementac¸ãointra-articularno tratamentodepacientescomosteoartrosesintomáticadejoelho.Foifeitaumarevisãoda literaturacomousodosbancosdedadosMedline,PubmedeCochraneControlledTrial RegistereCochraneDatabasesSystematicReviews(CochraneLibrary).Foramconsiderados apenasestudoscomelevadoníveldeevidências.Oestudoincluiuaanálisedeensaios clíni-cosrandomizadosqueincluírampelomenos100pacientesemcadagrupodeintervenc¸ão, metanáliseserevisõessistemáticas.Duasmetanálises,cincorevisõessistemáticaseseis ensaiosclínicosrandomizadospreencheramoscritériosdeinclusãodestarevisão.Frenteàs
夽
WorkperformedattheInstitutodeOrtopediaeTraumatologiadePassoFundo,PassoFundo,RS,Brazil.
∗ Correspondingauthor.
E-mails:[email protected],[email protected](O.V.LopesJúnior).
http://dx.doi.org/10.1016/j.rboe.2015.07.007
melhoresevidênciasexistentesatéomomento,nãoexisteconsensoparaindicac¸ãoeaté mesmocontraindicac¸ãodousodaviscossuplementac¸ãointra-articularempacientescom osteoartrosesintomáticadojoelho(níveldeevidênciaIegrauderecomendac¸ãoA).Futuros estudoscommetodologiaadequadasãonecessáriosparaelucidac¸ãodessaquestão.
©2014SociedadeBrasileiradeOrtopediaeTraumatologia.PublicadoporElsevier EditoraLtda.Todososdireitosreservados.
Introduction
Osteoarthrosisischaracterizedbypainandprogressivejoint dysfunctionresultingfrom destruction ofthecartilage and subchondralbone,withoccurrencesofreductionofthejoint space,inflammation/synovitisandformationofperiarticular osteophytes.1–4 Amongthe majorjoints,the knees are the
onesmostaffected,suchthatkneeosteoarthrosisgivesrise tofunctionaldeficitsin10%ofindividualsovertheageof55 yearsand in25% incasesofadvanced disease.5 Currently,
therearenoepidemiologicalstudiesinBrazilthathave pre-ciselyelucidatedtheprevalenceofosteoarthrosisorthepublic expenditurerelatedtothisdisease.Inviewofthesignificant increaseinlifeexpectancythathasoccurredintheBrazilian populationandthe increasingproportionofelderlypeople, osteoarthrosisneedstobeconsideredtobeamatterofpublic healthinterest.6
Severaloptionsforconservativetreatmentof osteoarthro-sis exist. These include weight reduction, physiotherapy, physicalexercise and extra-articular devices forfunctional assistance.Theoptionsforpharmacologicaltherapy(ordinary analgesics, opioids, non-steroidal anti-inflammatory drugs andcorticoids)areaimedtowardspainrelief.Glucosamines, diacereinandhyaluronicacidare drugsknowntobe modi-fiersofthenaturalhistoryofthedisease,andthesepromote improvementoffunctionalandpainlevelsovertheshortterm. Nonetheless,furtherstudiesareneededinordertoelucidate theefficiencyofdrugsforavoidingdiseaseprogression.There isstillnoeffectivemedicationforchangingthecourseofthis disease.7,8
Synovial fluid is composed of polysaccharides, among otherelements.Thesecontainglucosamine,glucuronicacid and hyaluronicacid, and this last substance isconsidered to be a key molecule in joint biomechanics. Hyaluronic acid is a biopolymer formed by glucuronic acid and N-acetylglucosamine.Ithasaviscoustextureandisfoundinthe synovialfluid,vitreoushumourandcollagenousconnective tissue of numerous organisms and is an important gly-cosaminoglycan(GAG)inconstitutingthejoint.Thismolecule istheonlynon-sulfatedGAG.Ithasthecapacitytobecome associatedwithproteinsinorderto formmolecular aggre-gates,butitdoesnotformproteoglycans.Injointsaffected byosteoarthrosis,theconcentrationandmolecularweightof hyaluronicacidinthesynovialfluidbecomereduced,which alters its properties through diminishing its viscosity and reducingitscapacitytoabsorbshockandprovidelubrication, andleadstodamagetocartilageandincreasedsymptoms.9–13
Itisbelievedthatthemechanismofactionofhyaluronic acidinjointsisrelatedtoinhibitionofinflammatory medi-ators and cartilage degeneration enzymes. This reduces
cartilage degradation and increases the production of cartilaginous matrix.7,14 Hyaluronic acid preparations for
intra-articular usecanbefurtherdivided betweenthoseof low and high molecular weight. According to some stud-ies,thereareadvantagesinusingthehighmolecularweight presentation.7,8 Despitethepossiblebenefitsof
viscosupple-mentation,itsuseremainscontroversial.
The present review had the objective of assessing the current evidence supportingor contraindicating theuse of intra-articularviscosupplementationwithhyaluronicacidfor treatingkneeosteoarthrosis.
Materials
and
methods
AreviewoftheliteraturewasconductedusingtheMedline, PubMed, Cochrane Controlled Trial Register and Cochrane SystematicReview(CochraneLibrary)databases.This inves-tigation used the keywords viscosupplementation, hyaluronic acid,osteoarthritis,randomized,reviewandmeta-analysis.Only studies defined as presenting high-quality evidence (level A, according to the Oxford Centre for Evidence-Based Medicine),15suchassystematicreviews,meta-analysesand
randomized clinical trials (RCTs),were included. The pop-ulation of interest included patients with symptomatic osteoarthrosisofthekneewhowereundergoingnon-surgical treatmentforpainfulosteoarthrosis.
Theinclusioncriteriaforarticleswereasfollows:
- Systematic reviews or meta-analyses on randomized clinicaltrialsthatassessedtheuseofintra-articular visco-supplementationfortreatingosteoarthrosisofthekneein humans;
- Randomizedcontrolledclinicaltrials(RCTs)thatcompared theuseofviscosupplementationwithplaceboorother med-ication,wereadequatelydesignedandincludedatleast100 patientsineachintervention(viscosupplementationor vis-cosupplementationandplacebo).
Thecriteriaforexcludingarticleswereasfollows:
- Studiesonanimals;
- Studies withfewerthan 100patients ineach armofthe intervention.
Results
Out of the 239 potentially eligible studies that were investigated through Medline and PubMed (keywords:
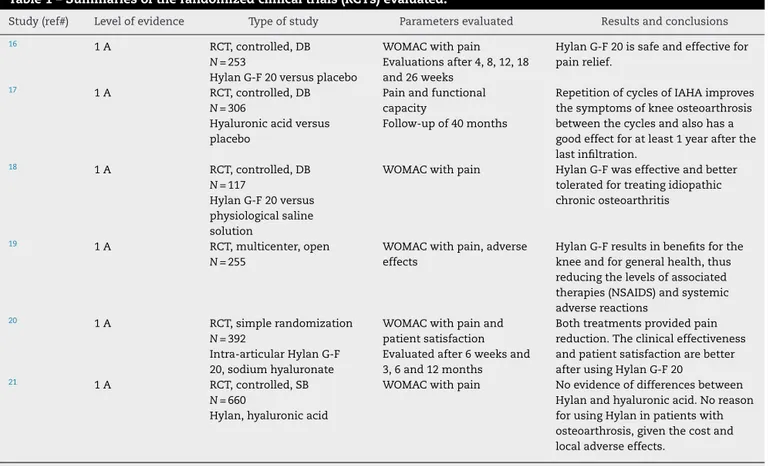
Table1–Summariesoftherandomizedclinicaltrials(RCTs)evaluated.
Study(ref#) Levelofevidence Typeofstudy Parametersevaluated Resultsandconclusions
16 1A RCT,controlled,DB
N=253
HylanG-F20versusplacebo
WOMACwithpain Evaluationsafter4,8,12,18 and26weeks
HylanG-F20issafeandeffectivefor painrelief.
17 1A RCT,controlled,DB
N=306
Hyaluronicacidversus placebo
Painandfunctional capacity
Follow-upof40months
RepetitionofcyclesofIAHAimproves thesymptomsofkneeosteoarthrosis betweenthecyclesandalsohasa goodeffectforatleast1yearafterthe lastinfiltration.
18 1A RCT,controlled,DB
N=117
HylanG-F20versus physiologicalsaline solution
WOMACwithpain HylanG-Fwaseffectiveandbetter toleratedfortreatingidiopathic chronicosteoarthritis
19 1A RCT,multicenter,open
N=255
WOMACwithpain,adverse effects
HylanG-Fresultsinbenefitsforthe kneeandforgeneralhealth,thus reducingthelevelsofassociated therapies(NSAIDS)andsystemic adversereactions
20 1A RCT,simplerandomization
N=392
Intra-articularHylanG-F 20,sodiumhyaluronate
WOMACwithpainand patientsatisfaction Evaluatedafter6weeksand 3,6and12months
Bothtreatmentsprovidedpain reduction.Theclinicaleffectiveness andpatientsatisfactionarebetter afterusingHylanG-F20
21 1A RCT,controlled,SB
N=660
Hylan,hyaluronicacid
WOMACwithpain Noevidenceofdifferencesbetween Hylanandhyaluronicacid.Noreason forusingHylaninpatientswith osteoarthrosis,giventhecostand localadverseeffects.
RCT,randomizedclinicaltrial;DB,doubleblinding;HA,hyaluronicacid;IAHA,intra-articularhyaluronicacid;SB,single-blinding.
fivewere systematic reviewsand twowere meta-analyses. Summariesandcommentsrelatingtothestudiesevaluated arepresentedinTables1and2.16–28
Discussion
Osteoarthrosisisthecommonestformofarthritisinpatients overtheageof50yearsandthekneesareamongthejoints mostcommonlyaffected.Becausethekneesareload-bearing joints,alterations totheir biomechanics lead tosignificant morbidityandfunctionallimitation.6Withtheincreaseinlife
expectancyoftheBrazilianpopulation,osteoarthrosisis tend-ingtobecomeapublichealthproblem.Nostudiesdirected towardsevaluating the prevalenceofosteoarthrosis or the publicexpenditureinvolvedintreatingithavebeenconducted inBrazil.6IntheUnitedStates,satesofmedicationsfor
treat-ingthisdiseasehadaturnoverofUS$760millionin2004.29
Thepharmacologicaltherapeuticoptionsforknee arthro-siscurrentlyavailablehavetheaimofpromotingpainrelief andfunctionalimprovement.Therearestillnomedications availableonthemarketwithproveninfluenceonthe progres-sionofthedisease.7,8
In individuals with osteoarthrosis, hyaluronic acid in the synovial fluid undergoes reductions in concentration and molecular weight, which lead to loss of viscosity and,consequently, lossofthe functionsoflubrication and shockabsorption.Thisprocesscontributestowards progres-sion of joint degeneration and activation of inflammatory pathways.13,30 Viscosupplementation with hyaluronic acid
wasdevelopedinordertopromotelonger-lastingpainrelief andfunctionalrecovery,andtodelaydiseaseprogression.17
Different mechanisms have been proposed for explaining its effect, such as stimulation of production of endoge-nous hyaluronic acid, suppression of degradation of the cartilaginous matrix and suppression of the inflammatory responsetointerleukin-1.Tofurtherincreasetheviscosityof hyaluronicacidanddiminishjointclearance,chemically mod-ifiedhyaluronicacidcompoundswerecreatedsuchthatthey wouldhavehighermolecularweights(around23×107Da)and alsoalongerhalf-life,whichwouldtheoreticallyincreasethe potentialanddurationofitseffect.31–35
Inamulticenterrandomizedcontrolledclinicaltrialwith 40monthsoffollow-up,namedtheAmeliaProject, Navarro-Sarabia et al.17 evaluated 306 patients over the age of 45
yearswhopresentedkneeosteoarthrosis(Kellgren–Lawrence gradesIIandIII,withaminimumjointspaceof2mm).Four cyclesofintra-articularinjectionofhyaluronicacidorplacebo wereperformed.Thepatientswereevaluatedwithregardto clinicalandfunctionalimprovementandsideeffects.These authorsconcludedthatthetreatmentwassafeandthatthere were significant improvements in functional capacity and symptoms,inrelationtothecontrolgroup,withaneffectthat wasmaintainedeven1yearafterthelastapplication.17
Chevalier et al.16 evaluated253patients overthe ageof
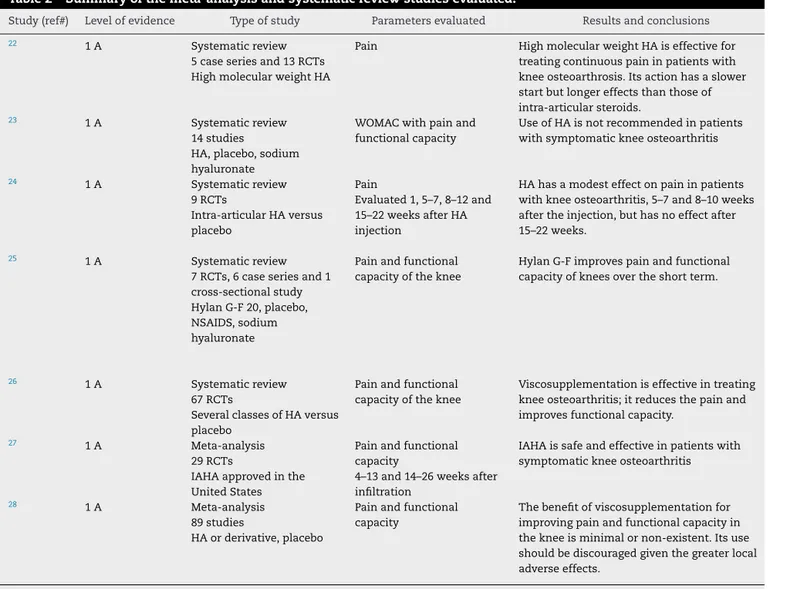
Table2–Summaryofthemeta-analysisandsystematicreviewstudiesevaluated.
Study(ref#) Levelofevidence Typeofstudy Parametersevaluated Resultsandconclusions
22 1A Systematicreview
5caseseriesand13RCTs HighmolecularweightHA
Pain HighmolecularweightHAiseffectivefor treatingcontinuouspaininpatientswith kneeosteoarthrosis.Itsactionhasaslower startbutlongereffectsthanthoseof intra-articularsteroids.
23 1A Systematicreview
14studies
HA,placebo,sodium hyaluronate
WOMACwithpainand functionalcapacity
UseofHAisnotrecommendedinpatients withsymptomatickneeosteoarthritis
24 1A Systematicreview
9RCTs
Intra-articularHAversus placebo
Pain
Evaluated1,5–7,8–12and 15–22weeksafterHA injection
HAhasamodesteffectonpaininpatients withkneeosteoarthritis,5–7and8–10weeks aftertheinjection,buthasnoeffectafter 15–22weeks.
25 1A Systematicreview
7RCTs,6caseseriesand1 cross-sectionalstudy HylanG-F20,placebo, NSAIDS,sodium hyaluronate
Painandfunctional capacityoftheknee
HylanG-Fimprovespainandfunctional capacityofkneesovertheshortterm.
26 1A Systematicreview
67RCTs
SeveralclassesofHAversus placebo
Painandfunctional capacityoftheknee
Viscosupplementationiseffectiveintreating kneeosteoarthritis;itreducesthepainand improvesfunctionalcapacity.
27 1A Meta-analysis
29RCTs
IAHAapprovedinthe UnitedStates
Painandfunctional capacity
4–13and14–26weeksafter infiltration
IAHAissafeandeffectiveinpatientswith symptomatickneeosteoarthritis
28 1A Meta-analysis
89studies
HAorderivative,placebo
Painandfunctional capacity
Thebenefitofviscosupplementationfor improvingpainandfunctionalcapacityin thekneeisminimalornon-existent.Itsuse shouldbediscouragedgiventhegreaterlocal adverseeffects.
RCT,randomizedclinicaltrial;DB,doubleblinding;HA,hyaluronicacid;IAHA,intra-articularhyaluronicacid.
repercussion(WOMACindex)wereevaluatedandtheauthors concludedthatthe treatmentwas safeand thattherewas asignificantclinicalimprovementamongthepatientswho underwentviscosupplementation.
Likeintheabovementionedstudy,inamulticenter ran-domizedstudy conductedbyRaynauldetal.,19 255patients
who received high molecular weight hyaluronic acid or placebowereevaluatedovera1-yearperiod.Theseauthors found a significant difference (greater than 20% in the WOMAC score) between their groups, which demonstrates thattherewerebenefitsfromviscosupplementation.Inother randomizedcontrolledtrialsthatusedhighmolecularweight hyaluronicacid(whicharelistedinTable1),itwasalso con-cludedthatthereweresignificantclinicalimprovements.18
Withregardtothemolecularweightofthehyaluronicacid tobeused,two studiescomparedthe useofhigh andlow molecularweighthyaluronicacidfortreatingosteoarthrosis. AccordingtoRaman etal.,20 use ofhigh-weighthyaluronic
acid(HylanG-F20)hastheadvantageofamorelong-lasting effect,butwithclinicalefficacyandtolerabilitysimilartoother presentations.Inaclinicaltrialthatcomparedthree presenta-tionsofhyaluronicacidfortreatingosteoarthrosisoftheknee, Jünietal.21 concludedthatthe differentmolecularweights
ofhyaluronicaciddidnotgiverisetoanysignificant differ-ences.
In asystematicreviewof76studies ofmediumquality, Bellamy et al.26 came to the conclusion that
viscosup-plementation was safe and led to significant clinical and functionalimprovements,incomparisonwithplacebo.They also reportedthat the effect ofthis treatment was longer-lastingthanthatofintra-articularcorticosteroids.Manyofthe studiesincludedintheirreviewpresenteddesign inadequa-cies.
AggarwalandSempowski22reviewedfivecaseseriesand13
randomizedcontrolledtrialsandconcludedthatuseof visco-supplementationfortreatingmildtomoderateosteoarthrosis of the knee, with high molecular weight hyaluronic acid, showed significant benefitsinrelation to clinical improve-ment anddurabilityofeffect.Theyalsodemonstrated that thepatientshadgoodtolerabilitytowardsthetreatmentand, incomparisonwithuseofintra-articularcorticoids,thepeak actionoccurredlaterandtheeffectwaslonger-lasting.Miller etal.27analyzedtheeffectandsafetyof
samplesizewereexcluded.Theseauthorsconcludedthat vis-cosupplementationwas effective forachieving clinical and functionalimprovements,aswellasbeingsafe.
Inameta-analysis,Rutjesetal.28evaluated89studiesand
12,667participantswith knee osteoarthrosis.Their conclu-sionwasthat,becauseoflackofevidenceofanysignificant clinicalandfunctionalimprovement,alongwiththepotential riskofsevereadverseeffectsviscosupplementationshouldbe discouragedfortreatingkneearthrosis.
Accordingtothe2013guidelinesfortreating osteoarthro-sis ofthe knee,23 published by the American Academy of
Orthopaedic Surgeons (AAOS) after a meta-analysis that involved 14 randomized controlled trials, there is strong evidence for not recommending the use of intra-articular hyaluronicacid,becauseallthestudiesthat made compar-isons with a control group showed uncertainty regarding practical clinical application of the treatment. Five of the sevenstudiesevaluatedthatrelatedtomolecularweight pre-sentedpatientswhoperhapswouldnotrepresentthegeneral conditionof the populationwith knee osteoarthrosis. This meta-analysiswas criticized inthe study byMiller et al.27
becauseofconfusioninthe dataanalysisand useof com-poundsthathadnotbeenapprovedintheUnitedStates.
Final
remarks
Thepatternofosteoarthrosistreatmentusinghyaluronicacid isextremelyvariablebetweenstudies.Therearedifferencesin thepreparationsused,numberofapplications,doseinjected perapplicationandnumberofcyclesused,inadditiontotime differencesbetweenthem.Theprofileofthepatientsanalyzed ineach studyalsovaried,suchthatsomepresentedyoung patientswithmildarthrosisandothers,elderlypatientswith severearthrosis.Theparameters foranalyzingclinical and functionalimprovementsalsochangedbetweenthestudies. Manyofthemdidnothaveacontrolgroupandthereisalso alackofstudiescomparingviscosupplementationwithother treatments.Mostofthestudiesareofpoorqualitywith inad-equatedesigns.
Conclusion
Inthelightoftheevidencethatcurrentlyexists,thereisstillno solidbasisforindicatingorevenforcontraindicatingtheuseof intra-articularviscosupplementationwithhyaluronicacidor itsderivativesfortreatingsymptomatickneeosteoarthrosis.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1. DieppePA,LohmanderLS.Pathogenesisandmanagementof paininosteoarthritis.Lancet.2005;365(9463):965–73.
2. HolbrookA.M.(Chair)fortheOntarioMusculoskeletal TherapyReviewPanel.In:Ontariotreatmentguidelinesfor osteoarthritis,rheumatoidarthritis,andacute
musculoskeletalinjury.Toronto:Queen’sPrinterofOntario; 2000.
3.BadleyE,DesMeulesM.ArthritisinCanada:anongoing challenge.Ottawa:HealthCanadá;2003.
4.FelsonDT.Anupdateonthepathogenesisandepidemiology ofosteoarthritis.RadiolClinNorthAm.2002;42(1):1–9.
5.PeatG,McCarneyR,CroftP.Kneepainandosteoarthritisin olderadults:areviewofcommunityburdenandcurrentuse ofprimaryhealthcare.AnnRheumDis.2001;60(2):91–7.
6.MinistériodaSaúdedoBrasil.RedeInteragencialde Informac¸õesparaaSaúde.Availableat:
http://tabnet.datasus.gov.br/cgi/idb2009/matriz.htm. 7.AmericanCollegeofRheumatologySubcommitteeon
OsteoarthritisGuidelines.Recommendationsforthemedical managementofosteoarthritisofthehipandknee:2000 update.ArthritisRheum.2000;43(9):1905–15.
8.ManekNJ,LaneNE.Osteoarthritis:currentconceptsin diagnosisandmanagement.AmFamPhysician. 2000;61(6):1795–804.
9.AyralX.Injectionsinthetreatmentofosteoarthritis.Best PractResClinRheumatol.2001;15(4):609–26.
10.GeorgeE.Intra-articularhyaluronantreatmentfor osteoarthritis.AnnRheumDis.1998;57(11):637–40.
11.SimonLS.Viscosupplementationtherapywithintra-articular hyaluronicacid.Factorfantasy?RheumDisClinNorthAm. 1999;25(2):345–57.
12.FamH,BryantJT,KontopoulouM.Rheologicalpropertiesof synovialfluids.Biorheology.2007;44(2):59–74.
13.DahlLB,DahlIM,Engström-LaurentA,GranathK.
Concentrationandmolecularweightofsodiumhyaluronate insynovialfluidfrompatientswithrheumatoidarthritisand otherarthropathies.AnnRheumDis.1985;44(12):817–22.
14.WenDY.Intra-articularhyaluronicacidinjectionsforknee osteoarthritis.AmFamPhysician.2000;62(3):565–70.
15.OxfordCentreforEvidence-BasedMedicine.Availableat:
http://www.cebm.net/index.aspx?o=5653.
16.ChevalierX,JeroschJ,GoupilleP,VanDijkN,LuytenFP,Scott DL,etal.Single,intra-articulartreatmentwith6mlhylanG-F 20inpatientswithsymptomaticprimaryosteoarthritisofthe knee:arandomised,multicentre,double-blind,placebo controlledtrial.AnnRheumDis.2010;69(1):113–9.
17.Navarro-SarabiaF,CoronelP,CollantesE,NavarroFJ,Dela SernaAR,NaranjoA,etal.A40-monthmulticentre, randomisedplacebo-controlledstudytoassesstheefficacy andcarry-overeffectofrepeatedintra-articularinjectionsof hyaluronicacidinkneeosteoarthritis:theAmeliaproject. AnnRheumDis.2011;70(11):1957–62.
18.WobigM,DickhutA,MaierR,VetterG.Viscosupplementation withhylanG-F20:a26-weekcontrolledtrialofefficacyand safetyintheosteoarthriticknee.ClinTher.1998;20(3):410–23.
19.RaynauldJP,TorranceGW,BandPA,GoldsmithCH,TugwellP, WalkerV,etal.Aprospective,randomized,pragmatic,health outcomestrialevaluatingtheincorporationofhylanG-F20 intothetreatmentparadigmforpatientswithknee osteoarthritis(part1of2):clinicalresults.Osteoarthritis Cartilage.2002;10(7):506–17.
20.RamanR,DuttaA,DayN,SharmaHK,ShawCJ,JohnsonGV. EfficacyofhylanG-F20andsodiumhyaluronateinthe treatmentofosteoarthritisoftheknee–aprospective randomizedclinicaltrial.Knee.2008;15(4):318–24.
21.JüniP,ReichenbachS,TrelleS,TschannenB,WandelS,Jordi B,etal.ViscosupplementationTrialGroup.Efficacyandsafety ofintraarticularhylanorhyaluronicacidsforosteoarthritisof theknee:arandomizedcontrolledtrial.ArthritisRheum. 2007;56(11):3610–9.
23.Treatmentofosteoarthritisoftheknee–evidence-based guideline.2nded.Rosemont:AmericanAcademyof OrthopaedicSurgeons;2013.
24.ModawalA,FerrerM,ChoiHK,CastleJA.Hyaluronicacid injectionsrelievekneepain.JFamPract.2005;54(9): 758–67.
25.EspallarguesM,PonsJM.Efficacyandsafetyof
viscosupplementationwithHylanG-F20forthetreatmentof kneeosteoarthritis:asystematicreview.IntJTechnolAssess HealthCare.2003;19(1):41–56.
26.BellamyN,CampbellJ,RobinsonV,GeeT,BourneR,WellsG. Viscosupplementationforthetreatmentofosteoarthritisof theknee.CochraneDatabaseSystRev.2006;(2):CD005321.
27.MillerLE,BlockJE.US-approvedintra-articularhyaluronic acidinjectionsaresafeandeffectiveinpatientswithknee osteoarthritis:systematicreviewandmeta-analysisof randomized,saline-controlledtrials.ClinMedInsights ArthritisMusculoskeletDisord.2013;6:57–63.
28.RutjesAW,JüniP,DaCostaBR,TrelleS,NüeschE, ReichenbachS.Viscosupplementationforosteoarthritisof theknee:asystematicreviewandmeta-analysis.AdvIntern Med.2012;157(3):180–91.
29.UnitedStatesCommeteeonHealth,Education,Labor,and Pensions.SubcommitteeonAging.CentreofDiseaseControl’s
roleincombatingtheburdenofarthritis.Washington: DepartmentofHealthandHumanServices;2004.
30.BalazsEA,DenlingerJL.Viscosupplementation:anew conceptinthetreatmentofosteoarthritis.JRheumatolSuppl. 1993;39:3–9.
31.SmithMM,GhoshP.Thesynthesisofhyaluronicacidby humansynovialfibroblastsisinfluencedbythenatureofthe hyaluronateintheextracellularenvironment.RheumatolInt. 1987;7(3):113–22.
32.BaggaH,BurkhardtD,SambrookP,MarchL.Longtermeffects ofintraarticularhyaluronanonsynovialfluidinosteoarthritis oftheknee.JRheumatol.2006:946–50.
33.GhoshP.Theroleofhyaluronicacid(hyaluronan)inhealth anddisease:interactionswithcells,cartilage,and componentsofsynovialfluid.ClinExpRheumatol. 1994;12(1):75–82.
34.YasuiT,AkatsukaM,TobettoK,HayaishiM,AndoT.The effectofhyaluronanoninterleukin-1alpha-induced
prostaglandinE2productioninhumanosteoarthriticsynovial cells.AgentsActions.1992;37(1–2):155–6.
35.MonfortJ,NacherM,MontellE,VilaJ,VergesJ,BenitoP. Chondroitinsulfateandhyaluronicacid(500–730kDa)inhibit stromelysin-1synthesisinhumanosteoarthritic