rev bras hematol hemoter. 2017;39(3):278–280
w w w . r b h h . o r g
Revista
Brasileira
de
Hematologia
e
Hemoterapia
Brazilian
Journal
of
Hematology
and
Hemotherapy
Case
Report
Erythrovirus
B19
induced
persistent
bicytopenia
in
a
healthy
child
Mili
Jain
∗,
Gurleen
Oberoi,
Rashmi
Kumar,
Ashutosh
Kumar
KingGeorge’sMedicalUniversity,Lucknow,India
a
r
t
i
c
l
e
i
n
f
o
Articlehistory:
Received24March2017 Accepted4April2017 Availableonline9May2017
Introduction
Immunethrombocytopenia(ITP),withanincidenceof3–5per 100,000individuals,isdiagnosedbyexclusion.Erythrovirus B19 (EV19) infection has been reported to be associated withITPinfrom13%to50%ofcases.1–3 Combined chronic
red cell hypoplasia and thrombocytopenia hasbeen rarely reportedinotherwisehealthyindividuals.Wereportacase ofEV19-inducederythroidhypoplasiaandthrombocytopenia diagnosedonbonemarrowexamination.
Case
report
Anotherwisehealthy4-year-oldmalechildinthemonthof June presented to us with a history of multiple petechiae andpurpura allover hisbody forthreemonthsalong with twoepisodesofmelenaeightdayspreviously.Hehadneither history ofother bleeding manifestationssuchas epistaxis, gum bleeding, hematemesis, hematuria, hemoptysis and hemarthrosis, nor were there reports of fever, rash, jaun-dice,diarrhea,acuteabdomenorarthralgia.Inaddition,no
∗ Correspondingauthorat:DepartmentofPathology,KingGeorge’sMedicalUniversity,226003Lucknow,India.
E-mails:[email protected],[email protected](M.Jain).
historyofrecentimmunization,drugintake,ortransfusions wasdescribed.Hispasthistoryandfamilyhistorywerenot significant.Onexamination,thepatienthadpallor,petechiae andpurpura alloverhisbody.Nolymphnodeenlargement orhepatosplenomegalywaspresent.Hisprevious investiga-tions from anotherinstitution showedagradual declinein hemoglobinfrom12.8to8.0g/dLoveraperiodofonemonth. Hisplateletcountwaslowovertheentireperiodrangingfrom 30×109/Lto50×109/L.
Onroutinebloodinvestigation,hemoglobinwas6.6g/dL. Generalbloodpicturerevealednormocyticandnormochromic redbloodcellswithnoevidenceofspherocytes,fragmented cells,schistocytes,polychromatophilsorimmaturecells.The whitebloodcellcountwasnormal(6.0×109/L)alongwith rel-ativelymphocytosis(58%).Theplateletcountwasmarkedly reducedto15.0×109cells/L.Hisbleedingtimewasraisedto 14min (by Ivy’s method); however, prothrombin time and thromboplastin time were within the normal ranges. His biochemical investigations were also within normal limits includingserumurea,creatinine,bilirubin,vitaminB12and folic acid. Serological tests for human immunodeficiency virus (HIV), hepatitisBand Cand Epstein BarrVirus(EBV)
http://dx.doi.org/10.1016/j.bjhh.2017.04.002
revbrashematolhemoter.2017;39(3):278–280
279
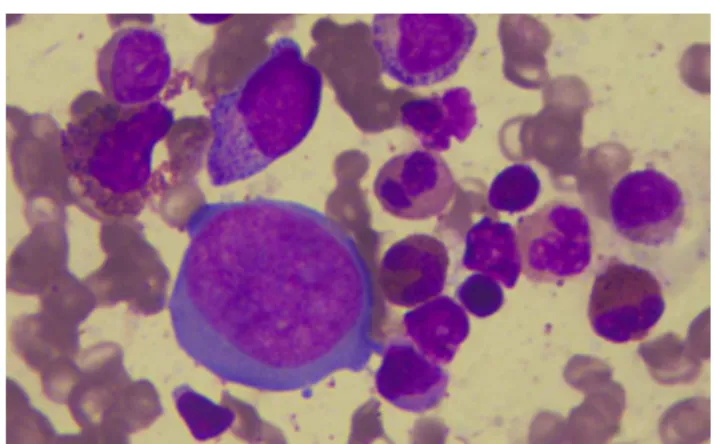
Figure1–Bonemarrowaspirate(magnification400×:
Leishmanstain)giantproerythroblastwithintranuclear
inclusionandabundantcytoplasmandnormalgranulocytic
precursors.
cameout to benegative. Direct antiglobulin test was neg-ative.Reticulocytecountwas <1%.Bone marrowaspiration smears were cellular with absolute reduction in erythroid precursors.Granulocyticprecursorsshowednormal morphol-ogyand maturation.Giant proerythroblastwith prominent intranuclearinclusionsandcytoplasmicprojections(dogear) suggestederythrovirus infection (Figure1). Megakaryocytes wereincreasedinnumberwithnumerousclusterformations. Multilobatedandhypolobated,andgranularand hypogranu-larformswereseen.However,allofthemwerenon-functional (consistentwithITP).Positiveserum immunoglobulin(Ig)M and IgG antibodies against EV19 confirmed our suspicion. Afinal diagnosis oferythrovirus induced red cell hypopla-siaand thrombocytopenia was rendered after detection of erythrovirusDNAbyqualitativerealtimepolymerasechain reaction(RT-PCR)usingprimersforthevirionstructural pro-tein(VP1).Theamplificationproductsweredetectedonthe basis of fluorescent dye labeled probes.4 The patient was
treatedwithintravenousimmunoglobulin(IVIG,1mg/kgbody weight)approvedforuseinerythrovirusinfections.
Discussion
YvonneCossartdiscoveredEV19(previouslycalledparvovirus B19)in1974inLondon.5Infectionismostcommoninlate
win-terandearlyspring.Thevirusistransmittedthroughexposure torespiratorydropletsorbloodproductsaswellasthrough vertical transmission from mother to fetus. It is a single strandedDNAviruswithmarkedtropismforerythroidcells viaattachmenttotheglobosidePantigen.Theantigenisalso foundonplatelets,endothelialcells,cardiacmyocytes,and synovium,liver,lung,andkidneytissue.Specificitytoinfect erythroidlineagemaybeattributedtopreferentialexpression ofthePantigeninerythrocytesandhighactivityoftheviral P6promoterinthesecells.6
Theclinicalmanifestationsarestronglyinfluencedbythe immunologicalandhematologicalstatusofthehost.In non-immunocompromised individuals,the infection may range fromasymptomatictomildflu-likeillness.Theotherprimary manifestations include erythema infectiosum, arthropathy
and hydropsfetalis.7 Viremiaisusuallydetected5–10days
afterexposure.Inahealthyindividual,atransientdecreaseof hemoglobinofupto1g/dLmaybeseenwithmarked reticulo-cytopeniaduringthephaseofviremia.Clinicallyinsignificant leucopenia,thrombocytopeniamayalsobeseen.All hemato-logicalparametersusuallynormalizewithintwoweeks.Thus, subclinicalerythroidhypoplasiafollowedbyrashor arthral-giaisthemostcommonclinicalpictureinimmunocompetent hosts.Inimmunocompromisedhosts,theinfectionmay man-ifestwithaplasticcrisis,chronicanemiaorITP.8Inpatients
with hemolytic anemia such as sickle cell anemia, EV19 may cause a transient aplastic crisis with sudden drop in hemoglobin.Althoughthemajorityofpatientsrecoverwithin twoweeks,thereisincreasedriskofcongestiveheartfailure, stroke,andacutesplenicsequestration.9
Ourcasewasanotherwisehealthychildpresentingwith symptomaticthrombocytopeniaandseverenormocytic nor-mochromicanemia.Bonemarrowexaminationrevealedclues forEV19infection(Lanterncells)alongwithmegakaryocytic hyperplasia(consistentwithITP).Thiswasfurtherconfirmed by positive IgM antibodies (indicating immunocompetency andacuteinfection).Ourcaseisuniqueduetopersistenceof erythroid suppressionandimmunemediated thrombocyto-peniainanotherwisehealthychild.Cytokinemediatedtoxic effectsofviralNS-1(centraltype)mayexplainthebi-lineage involvementinthiscase.10,11Thesecondmechanismmaybe
immune-mediateddestruction(peripheraltype).12
SerumIgMantibodytestinghas89%sensitivityand99% specificity.13AnelevatedlevelofIgMantibodiesappears10–12
daysandremainsdetectablefor3–6monthsafteracute infec-tion.IgGantibodiespresumablypersistforlife.Thediagnostic antibodies are detected against VP1 and VP2 antigens. In immunocompromisedhosts,asantibodyproductionis min-imal or absent, viral DNA testing by PCR is necessary for diagnosis. PCR is moresensitive than in situ hybridization assays.
Ourcasehighlightstheimportanceoftheidentificationof morphologicalfeaturesofEV19infection.Thecasealso illus-tratesthevariableclinicalmanifestationofEV19infection.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1.HeegaardED,RosthojS,PetersenBL,NielsenS,Karup PedersenF,HornslethA.RoleofparvovirusB19infectionin childhoodidiopathicthrombocytopenicpurpura.Acta Paediatr.1999;88(6):614–7.
2.MurrayJC,KelleyPK,HogrefeWR,McClainKL.Childhood idiopathicthrombocytopenicpurpura:associationwith humanparvovirusB19infection.AmJPediatrHematolOncol. 1994;16(4):314–9.
3.AktepeOC,YetginS,OlcayL,OzbekN.HumanparvovirusB19 associatedwithidiopathicthrombocytopenia.Pediatr HematolOncol.2004;21(5):421–6.
4.ClewleyJP.PolymerasechainreactionassayofparvovirusB19 DNAinclinicalspecimens.JClinMicrobiol.
280
revbrashematolhemoter.2017;39(3):278–2805. CossartYE,FieldAM,CantB,WiddowsD.Parvovirus-like particlesinhumansera.Lancet.1975;1(7898):72–3.
6. GareusR,GiglerA,HemauerA,Leruez-VilleM,MorinetF, WolfH,etal.Characterizationofcis-actingandNS1
protein-responsiveelementsinthep6promoterofparvovirus B19.JVirol.1998;72(1):609–16.
7. AndersonMJ,HigginsPG,DavisLR,WillmanJS,JonesSE,Kidd IM,etal.Experimentalparvoviralinfectioninhumans.J InfectDis.1985;152(2):257–65.
8. FloreaAV,IonescuDN,MelhemMF.ParvovirusB19infection inimmunocompromisedhost.ArchPatholLabMed. 2007;131(5):799–804.
9. Smith-WhitleyK,ZhaoH,HodkinkaRL,KwiatkowskiJ,Cecil R,CecilT,etal.EpidemiologyofhumanparvovirusB19in childrenwithsicklecelldisease.Blood.2004;103(2):422–7.
10.MoffattS,YaegashiN,TadaK,TanakaN,SugamuraK.Human parvovirusB19nonstructuralproteinNS1inducesapoptosis inerythroidlineagecells.JVirol.1998;72(4):3018–28.
11.NagaiK,MorohoshiT,KudohT,YotoY,SuzukiN,Matsunaga Y.Transienterythroblastopeniaofchildhoodwith
megakaryocytopeniaassociatedwithhumanparvovirusB19 infection.BrJHaematol.1992;80(1):131–2.
12.InoueS,KinraNK,MukkamalaSR,GordonR.ParvovirusB-19 infection:aplasticcrisis,erythemainfectiosumand
idiopathicthrombocytopenicpurpura.PediatrInfectDisJ. 1991;10(3):251–3.