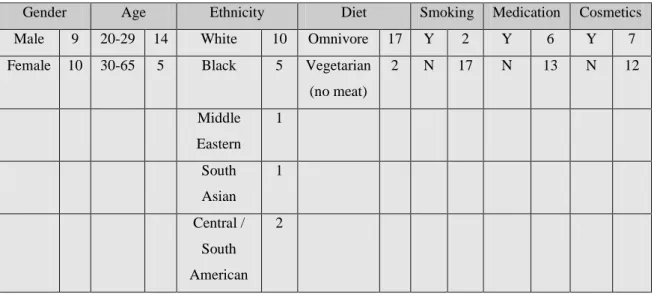
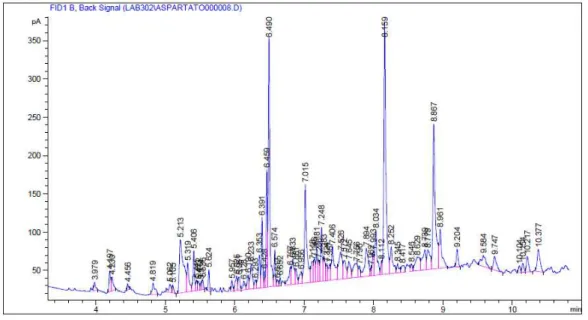
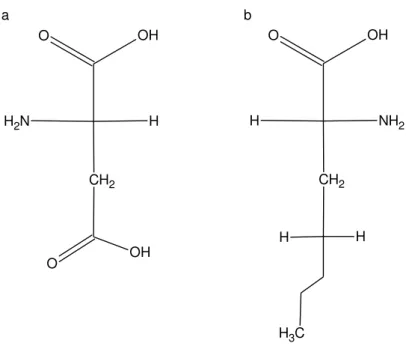
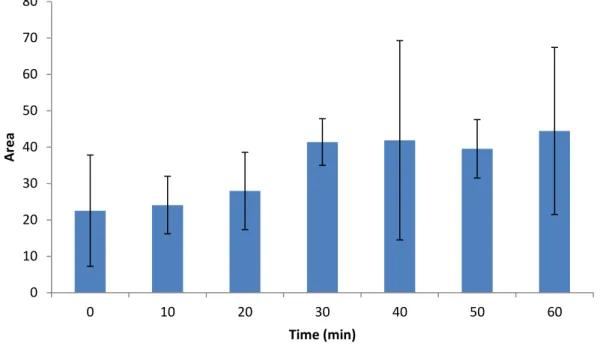
Adelantado Sánchez, Carlos
Texto
Imagem

Documentos relacionados
Para completar a minha missão, recorro aos estudos de Carl Gustav Jung, Claude Lévi-Strauss, Joseph Campbell e outros especialistas reputados..
Samples from lung and liver of Balb-C isogenic allergic pneumonitis bearing mice, treated with the same extract, were analyzed through GC-FID, and the fatty acid content
So, the aim of this work is to perform HPLC-DAD fingerprint analyses in “chapéu- de-couro” samples collected in different sites from Mato Grosso and Mato Grosso do Sul states, and
Samples were taken for rotavirus identification on day zero (recruitment to study), and monthly thereafter for one year (in addition to samples taken during acute diarrhea episodes
Os contributos de Slade, Way, Heathcote e de outros autores viriam a fundamentar substancialmente a criação de companhias de teatro para a infância completamente
portanto está bastante equilibrado (12/13) O texto escrito aparece quando, de facto, faz sentido, e se adequa aquilo que estamos a fazer e a produção oral que, no fundo, acaba
The results of this study contribute to the literature of entrepreneurship in education, and other related fields, by providing rigorous evidence on the reliability and validity of
Pattern recognition of the GC×GC-FID chromatograms was performed by parallel factor analysis (PARAFAC) and it was successfully applied for the differentiation of these