w w w . j c o l . o r g . b r
Journal
of
Coloproctology
Review
Article
A
review
of
experimental
models
in
colorectal
carcinogenesis
夽
Vanessa
Foresto
Machado,
Marley
Ribeiro
Feitosa
∗,
Jose
Joaquim
Ribeiro
da
Rocha,
Omar
Féres
DivisionofColoproctology,DepartmentofSurgeryandAnatomy,FaculdadedeMedicinadeRibeirãoPreto(FMRP),UniversidadedeSão Paulo(USP),RibeirãoPreto,SP,Brazil
a
r
t
i
c
l
e
i
n
f
o
Articlehistory: Received28April2015 Accepted4September2015 Availableonline20December2015
Keywords: Animalmodels Colorectalneoplasms Diagnosis
a
b
s
t
r
a
c
t
Colorectalcanceristheleadingcauseofmalignancyofthegastrointestinaltract.Abetter understandingofthemolecularandcellularchangesthatleadtothediseaseisnecessaryto developearlydiagnosisandoptimaltreatmentmodalities.Rodentmodelsarerapid, repro-ducibleandexhibitanadenoma-carcinomasequencesimilartothatfoundinhumans.The objectiveofthismanuscriptistoreviewthemostcommonchemicalcarcinogensusedto induceexperimentaltumorsandtheusualmethodsofevaluation.
©2015SociedadeBrasileiradeColoproctologia.PublishedbyElsevierEditoraLtda.All rightsreserved.
Modelos
experimentais
de
carcinogênese
colorretal
Palavras-chave: Modelosanimais Neoplasiascolorretais Diagnóstico
r
e
s
u
m
o
Ocâncercolorretaléaprincipalneoplasiamalignadotratogastrointestinal.Ummelhor entendimento dos processos moleculares e celulares é necessário para o desenvolvi-mentodeestratégiasquepermitamumdiagnósticoprecoceeumtratamentomaiseficaz. Modelosqueutilizamroedoressãorápidos,reprodutíveisepermitemoestudoda sequen-cia adenoma-carcinomadeforma similaraencontradaem humanos.Oobjetivodesse manuscritoérevisarosprincipaismodelosdecarcinogênesequímicaeosmétodosmais usuaisparaavaliac¸ãodosresultados.
©2015SociedadeBrasileiradeColoproctologia.PublicadoporElsevierEditoraLtda. Todososdireitosreservados.
夽
StudyperformedattheDepartmentofSurgeryandAnatomy,FaculdadedeMedicinadeRibeirãoPreto(FMRP),UniversidadedeSão Paulo(USP),RibeirãoPreto,SP,Brazil.
∗ Correspondingauthor.
E-mail:[email protected](M.R.Feitosa).
http://dx.doi.org/10.1016/j.jcol.2015.09.001
Introduction
Colorectalcancer(CRC)isthemostcommongastrointestinal malignancy.Worldwide,CRCisthethirdmostcommoncancer inmenandthesecondinwomen.InBrazil,atotalof32,000 newcasesofCRCareexpectedin2015.1,2
Colorectalmalignant neoplasmsarecharacterizedbyan excessiveanduncontrolledgrowthofabnormalcells originat-ingfromanypartofthecolon.Unlikebenignlesions,cancer ismorphologicallydifferentfromthenormaltissueoforigin, havetheabilitytoinvadeand destroy surroundingtissues, metastasistodistantorgansand,ifleftuntreated,willleadto death.
Thefactorsinvolvedintheoriginandprogressionofcolon tumorshavebeenanareaofincreasinginterest.Sincethefirst experimentofcolontumorsinduction,moreaccurate mod-els and various substanceswith carcinogenic activity have beenstudied.Carcinogen-inducedcoloncancer,haveproved toshare many similaritieswithhumantumorsand signif-icantly contributed to our actual understanding regarding cancerpathogenesis.3
The ideal experimental carcinogen should induce neo-plasmsinthecolon,however,mostagentslackspecificity(e.g. 1,2-dimethylhydrazine,thatmayinducelivertumors). Repro-ducibilityisanotherimportantfeaturethatmaybehampered bytheheterogeneousspeciesandlineagesofanimals,each withdifferentpatternsofdrugresponse.Moreover,thereis nostandardizationofdoseandtimeoftumordevelopment, tothevariousstudieddrugs.
Rodentsarewidelyacceptedmodelsofcolorectal carcino-genesisbecauseoftheirsimilaritywithhumans.Advantages includerapid,reproducibletumorinductionandthe possibil-itytostudytheadenoma-carcinomasequence.4
Naturalcolorectalcarcinogenesisfactorsmaybedivided intotwomaincategories:thoserelatedtogeneticor environ-mentalfactors.
Geneticfactors
The importance of oncogenes and suppressive genes are widelyrecognized.Earlygeneticchangesincludechromosome 5(APC)andrasmutationswhilealleliclossesinchromosomes 17(p53)and18(DCC)happenlateintheadenoma-carcinoma sequence.5 A schematic genetic model of the
adenoma-carcinomasequenceisshowninFig.1.
Environmentalfactors
Dietplaysanimportantroleoncarcinogenesisand,insome countrieswithhigherCRCprevalence,anattributedriskof 50%isestimated.6GeographicvariationsinCRCincidences
andstudieswithimmigrantpopulationssuggestthatlifestyle factors including poor diet, physicalinactivity and alcohol consumptionareassociatedtoanincreasedriskofCRC.While increasedreadmeatconsumptionmaybeharmful, omega-3,vitaminD,phenoliccompoundsandafiber-richdietmay lowertheriskofCRC.Regularphysicalactivitymaylowerthe riskin24%;ontheotherhand,obesitymayincreaseitin19%. Althoughmoderatetohighdosesofalcoholhaveprovento
bedeleterious,somestudieshaveobservedaprotectiveeffect whenlightdosesareconsumed.7,8
Methods
of
chemical
carcinogenesis
There are two typesof chemical agents: (1) direct agents, that donot need metabolization by the organism to have thedeleteriouseffectand(2)indirectagents,whicharenot activeunlessenzymaticreactionsconvertthemtoanactive form.9
1,2-Dimetilhidrazine(DMH)
DMH is the metabolic precursor of methylazoxymethanol (MAM).Itistheoldestandthemostusedcarcinogentoinduce tumorsinrats.UptakeofDMHisthreetimesgreaterinthe colon cells compared tothe enterocytes. The carcinogenic effectmaybeobtainedafterasingleinjectionorviaaseries ofweeklyinjections.10,11Themalignantlesionoriginatesfrom
the non-dysplasticmucosaand becomesevidentafter4–30 weeksafteradministrationofthedrug.Evenafterthe admin-istrationofsmalldosesofthedrug,upto80%ofthetreated micecandevelopadenocarcinoma.12
Azoxymethane(AOM)
AOMisametaboliteofDMH.Itscarcinogenesismechanism isattributedtoc-fosoverexpression,reduced expressionof c-myc and k-ras mutation. These changes are similar to thoseobservedinspontaneouscarcinogenesisinhumans.13
ComparedtoDMH,AOMismorepotentandrequiresfewer reactionstoitsactivation,whichmakesabetteroption.Itis activatedintheliverbyN-oxidation,andproducesessential reactive compounds forchemical carcinogenesis (methyla-zoxymethanol andmethyl-diazoxide),whicharebroughtto thecolonthroughthebloodstreamorbileasconjugated glu-curonide.
Heterocyclicamines(HAs)
AmongtheHAs,2-amino-3-methylimidazo[4,5-f]quinolone (IQ)and2-amino-1-methyl-6-phenylimidazo[4,5-b]pyridine (PhIP)havegainedattentionafterprovedtobehighly muta-genic and tumorigenic in rodents. They appear to have target-organspecificityandcaninducemalignanciesofthe colon,prostateandmammaryglands.Creatinine,sugarsand aminoacidsfromredmeatandfisharetheprecursorsofIQ. Thefinalactivecompoundisformedhepaticmetabolization. Incidenceofinducedcoloncancermayreach28%after admin-isteringadietrichinIQandPhIP,for52weeks.Theybecame notonlyaninterestingmodelofcarcinogenesisbutcanalso helpinvestigationofchemoprotectioneffectagainstcancer, ofsomesubstances.14
Aromaticamines
5q mutation/loss APC gene
Normal epitelium
Hyperproliferative
epitelium Carcinoma
18q loss DCC gene
12p mutation K-ras gene
Adenoma
Methylation abnormalities
17p loss p53 gene
Fig.1–Theadenoma-carcinomasequence.AdaptedfromVogelstein.5
etal.whonoticedcolontumorinductioninmice,after sub-cutaneousadministrationof3,2′-dimethyl-4-aminobiphenyl
(DMAB).
After 20 weeks of 50mg/kg subcutaneous injections of DMABinmale F344rats multipleadenomasand adenocar-cinomaswereinducedin30%ofanimalsfedwithlow-fatdiet andin74%oftheanimalswithhigh-fatdiet.
Thismodelhastwodisadvantages:(1)theneedfor mul-tipleinjectionsofDMABand(2)lowspecificity,sincetumors maybeinducedinothertissuessuchasadenocarcinomaof mammaryglands,sarcomaofsalivaryglands,squamouscell carcinomaoftheearandskin,gastricsquamouscell papil-loma,sarcoma, lymphomaand urothelialcarcinomaofthe bladder.15,16
Alkylnitrosamines
Alkylnitrosaminessuchasmethylnitrosourea(MNU)and N-methyl-N′-nitro-N-nitrosoguanidine(MNNG)aredirect-acting
carcinogens withhigh affinityand methylationcapacity of specificDNAregions.Rectalinstillationofsuchcompounds induce sessile and polypoid colorectal tumors in rodents. Dosesof1–3mg/weekofMNNGinstilledperrectum, for20 weeks,inducedtumorsin100%ofF344femalerats.Themain disadvantageofthismodelisthedifficultytoinstillaprecise amountofthedrugperrectum.17
Evaluation
of
experimental
models
Histologicalmethod
Optical microscopy can assess cellular changes including the development of aberrant crypt foci (ACF) that may be considered apreneoplasticevent.ThedevelopmentofACF is considered an early marker of a future CRC.18 Tumors
maybeclassifiedasadenomasoradenocarcinomas.Optical microscopymayalsoevaluatethedifferentiationgradeand thelevelofpenetrationthroughtheintestinalwall(Tis,T1, T2,T3eT4).
Immunohistochemistry
The method consists in associating specific histochemical dyestoanantigen-antibodyreaction,whichcanincreasethe identificationrateoftumors.Differentantigenscanbeused inthismethod:
Metallothionein(MT):MTsarelow-molecular-weight pro-teins,richincysteineresidues,mainlyexpressedintheliver, kidney,intestineandbrain.They playanimportantrole in protectingagainstoxidativestressbybindingmetalssuchas zincandcopper.ElevatedMTlevelscanbefoundincancerous tissueswithhighproliferationrates,sincetheycanprovide zincandcopperionsforthemetabolismofnucleicacids, pro-teinsynthesis,andothermetabolicprocesses.MThasbecome animportantbiomarkerofpreneoplasticlesionsofthecolon, whereitispredominantlyexpressedbymutatedcryptstem cells.19
Gamma-H2AX: histones are proteins that compact and
order DNA into nucleosomes. They playan importantrole onDNAtranscription,replicationandrepair.H2AXoriginates fromH2AphosphorylationafterDNAdamageandis respon-sibleforrecruitmentofrepairproteins.H2AXis,therefore,an importantDNAdamagebiomarker.20
Vascularendothelialgrowthfactor(VEGF):angiogenesisis anessentialmechanismforthegrowthanddevelopmentof CRC.VEGFiscloselyassociatedwithneovascularformation incolorectalneoplasia.Ahypoxictumormicroenvironment iscriticaltotheneoplasticprocessandastrongstimulusfor theproductionofVEGFviahypoxia-induciblefactor1␣
(HIF-1␣).21,22
Hypoxia-induciblefactor1␣:HIF-1␣isadimeric
transcrip-tion factor,whichis thekey tocellularhomeostasisunder hypoxia condition by regulating the expression of various genesinvolvedincancerbiology.23
Anti-COX2:earlyindicationsofCOX-2roleinCRC develop-mentcamefromtheobservationthatpatientswithGardner’s syndrome treated with non-steroidal anti-inflammatory drugshad reducednumbersofadenomas.Indeed,elevated levels of COX-2 may be found in some adenomas and in adenocarcinomas. ThetumorigeniceffectofCOX-2maybe attributed to the production of prostaglandin E (PGE2). A disruptionintheCOX-2/PGE2cascadeappearstoaffect colo-rectal tumorigenesis by promoting the maintenance and progressionoftumor,increasedmetastasisrateandeven par-ticipatingintumorinitiation.24
Cytokineassay
The immuneresponse may contributeto neoplastic trans-formation. Some studies have shown that inhibition of proinflammatory cytokines, such as tumor necrosis factor alpha(TNF-␣),mayleadtoreductionofpreneoplasticlesions.
Ontheotherhand,productionofproinflammatorycytokines andchemokinesbymacrophagesandmastcells,forexample, may stimulate other cells to produce cytokines with anti-inflammatory activity,suchasinterleukin-10 (IL-10),which maycontributetotumor-associatedimmunosuppression.25
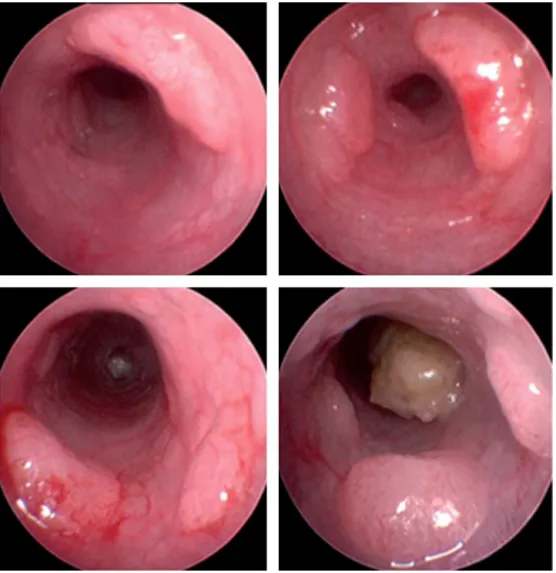
Endoscopy
High-resolution endoscopic devices can assess mucosal changes consistent with preneoplastic lesions or cancer. Endoscopic aspects are similar to humans. Vegetating and ulcerated lesions can be found (Fig. 2). Serial biopsies are requiredtodiagnosethenatureofthelesions.Scoresthattake theamountandsizeoftumorsintoaccountshouldbeuseto obtainuniformevaluations.26,27
Conclusion
Experimentalmodelsallowustostudytheearlystagesof car-cinogenesisincluding the adenoma-carcinomasequence. A betterunderstandingofthebiochemicalandcellularchanges thatdeterminecarcinogenesiscanhelpthedevelopmentof newmethodsofdiagnosis,aswellasbetterandmoreeffective treatment.
Conflicts
of
interest
Theauthorsdeclarenoconflictsofinterest.
r
e
f
e
r
e
n
c
e
s
1.Estimativa2014:IncidênciadeCâncernoBrasil.Riode Janeiro:INCA;2014.
2.StewartBW,WildCP.Worldcancerreport2014.Lyon;2014.
3.TaketoMM,EdelmannW.Mousemodelsofcoloncancer. Gastroenterology.2009;136:780–98.
4.BalmainA,HarrisCC.Carcinogenesisinmouseandhuman cells:parallelsandparadoxes.Carcinogenesis.2000;21:371–7.
6. KuneGA,BannermanS,WatsonLF.Attributableriskfordiet, alcohol,andfamilyhistoryintheMelbourneColorectal CancerStudy.NutrCancer.1992;18:231–5.
7. CardosoJF,CohenC,JordaoAAJr,VannucchiH,GarciaSB, ZucolotoS.Lightandmoderatedosesofethanolinchemical carcinogenesisofthecoloninrats.NutrCancer.
2011;63:1029–35.
8. WengCJ,YenGC.Chemopreventiveeffectsofdietary phytochemicalsagainstcancerinvasionandmetastasis: phenolicacids,monophenol,polyphenol,andtheir derivatives.CancerTreatRev.2012;38:76–87.
9. TanakaT.Colorectalcarcinogenesis:reviewofhumanand experimentalanimalstudies.JCarcinog.2009;8:5.
10.OliveiraEC,LeiteMS,MirandaJA,AndradeAL,GarciaSB, LuquettiAO,etal.ChronicTrypanosomacruziinfection associatedwithlowincidenceof
1,2-dimethylhydrazine-inducedcoloncancerinrats. Carcinogenesis.2001;22:737–40.
11.GoisEJr,DanielRA,ParraRS,AlmeidaAL,RochaJJ,GarciaSB, etal.HyperbaricoxygentherapyreducesCOX-2expressionin adimethylhydrazine-inducedratmodelofcolorectal carcinogenesis.UnderseaHyperbMed.2012;39:693–8.
12.RodriguesMA,SilvaLA,SalvadoriDM,DeCamargoJL, MontenegroMR.Aberrantcryptfociandcoloncancer: comparisonbetweenashort-andmedium-termbioassayfor coloncarcinogenesisusingdimethylhydrazineinWistarrats. BrazJMedBiolRes.2002;35:351–5.
13.CaderniG,FemiaAP,GianniniA,FavuzzaA,LuceriC, SalvadoriM,etal.Identificationofmucin-depletedfociinthe unsectionedcolonofazoxymethane-treatedrats:correlation withcarcinogenesis.CancerRes.2003;63:2388–92.
14.RosenbergDW,GiardinaC,TanakaT.Mousemodelsforthe studyofcoloncarcinogenesis.Carcinogenesis.
2009;30:183–96.
15.WalpoleAL,WilliamsMH,RobertsDC.Thecarcinogenic actionof4-aminodiphenyland
3:2′-dimethyl-4-amino-diphenyl.BrJIndMed.1952;9:255–63.
16.LorenzE,StewartHL.Intestinalcarcinomaandotherlesions inmicefollowingoraladministrationof
1,2,5,6-dibenzanthraceneand20-methylcholanthrene.JNatl CancerInst.1940;1:17–40.
17.MaurinN,Forgue-LafitteME,LevyP,ZimberA,BaraJ. ProgressionoftumorsarisingfromlargeACFisassociated withtheMUC5ACexpressionduringratcolonMNNG carcinogenis.IntJCancer.2007;120:477–83.
18.PretlowTP,BarrowBJ,AshtonWS,O’RiordanMA,PretlowTG, JurcisekJA,etal.Aberrantcrypts:putativepreneoplasticfoci inhumancolonicmucosa.CancerRes.1991;51:1564–7.
19.BruewerM,SchmidKW,KrieglsteinCF,SenningerN, SchuermannG.Metallothionein:earlymarkerinthe carcinogenesisofulcerativecolitis-associatedcolorectal carcinoma.WorldJSurg.2002;26:726–31.
20.KuoLJ,YangLX.Gamma-H2AX–anovelbiomarkerforDNA double-strandbreaks.Invivo(Athens,Greece).2008;22:305–9.
21.StackerSA,CaesarC,BaldwinME,ThorntonGE,WilliamsRA, PrevoR,etal.VEGF-Dpromotesthemetastaticspreadof tumorcellsviathelymphatics.NatMed.2001;7:186–91.
22.DanielRA,CardosoVK,GoisEJr,ParraRS,GarciaSB,RochaJJ, etal.Effectofhyperbaricoxygentherapyontheintestinal ischemiareperfusioninjury.ActaCirBras.2011;26:463–9.
23.ChenH,FengJ,ZhangY,ShenA,ChenY,LinJ,etal.PienTze Huanginhibitshypoxia-inducedangiogenesisvia
HIF-1␣/VEGF-Apathwayincolorectalcancer.EvidBased
ComplementAlternatMed.2015;2015:454279.
24.GreenhoughA,SmarttHJ,MooreAE,RobertsHR,Williams AC,ParaskevaC,etal.TheCOX-2/PGE2pathway:keyrolesin thehallmarksofcancerandadaptationtothetumour microenvironment.Carcinogenesis.2009;30:377–86.
25.KlampferL.Cytokines,inflammationandcoloncancer.Curr CancerDrugTargets.2011;11:451–64.
26.BeckerC,FantiniMC,WirtzS,NikolaevA,KiesslichR,Lehr HA,etal.Invivoimagingofcolitisandcoloncancer developmentinmiceusinghighresolution chromoendoscopy.Gut.2005;54:950–4.